| Journal of Neurology Research, ISSN 1923-2845 print, 1923-2853 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Neurol Res and Elmer Press Inc |
| Journal website https://jnr.elmerpub.com |
Review
Volume 15, Number 2, March 2025, pages 65-70
Transcranial Direct Current Stimulation in Motor Therapy for Patients With Parkinson’s Disease: A Systematic Review
Lucas Brito Meiraa , Andrey Teixeira
Ferreiraa
, Joao Vitor Andrade Fernandesa,
c
, Andre de Sa
Braga Oliveirab
aCenter for Medical Sciences, Federal University of Paraiba, Joao Pessoa,
Brazil
bDepartment of Morphology, Federal University of Paraiba, Joao Pessoa,
Brazil
cCorresponding Author: Joao Vitor Andrade Fernandes, Medical Sciences
Center, Federal University of Paraiba, Joao Pessoa, Paraiba, Brazil
Manuscript submitted February 3, 2024, accepted January 15, 2025, published online January 20,
2025
Short title: tDCS and Parkinson’s Disease
doi:
https://doi.org/10.14740/jnr874
| Abstract | ▴Top |
Parkinson’s disease (PD) is a neurodegenerative disorder that leads to motor and non-motor impairments, including tremors, bradykinesia, and rigidity. Current treatments, such as pharmacological therapies and deep brain stimulation, have efficacy and side effects limitations. Transcranial direct current stimulation (tDCS) is a non-invasive technique that may offer a complementary approach to managing PD motor symptoms. This review aims to evaluate the efficacy of tDCS in improving motor outcomes in PD patients. This systematic review followed PRISMA guidelines and was registered on the PROSPERO platform (CRD4202457096). We included randomized controlled trials (RCTs) that assessed tDCS in PD patients with a control group. Studies were identified through PubMed, Embase, and Cochrane Library searches up to January 2025. Data on participant demographics, interventions, outcomes, and results were extracted and analyzed. Risk of bias was assessed using the Cochrane RoB 2 tool. Five studies involving 176 participants (mean age 67.13 years) were included. Results indicated that tDCS improved motor outcomes, including gait, posture, and balance. Notably, it demonstrated enhanced dual-task gait speed and improvements in gait cadence. Also, it was reported that there were reductions in freezing of gait severity, while improvements in trunk stability and postural control. Despite promising findings, heterogeneity across studies was observed, particularly regarding stimulation parameters and outcome measures. This review provides evidence that tDCS can improve motor symptoms in PD, particularly gait and posture. However, variability in stimulation protocols and small sample sizes limit generalizability. Larger, standardized trials are needed to confirm tDCS’s efficacy, optimize treatment parameters, and assess long-term effects. tDCS shows potential as an adjunctive therapy for managing motor symptoms in PD patients.
Keywords: Parkinson’s disease; Transcranial direct current stimulation; Motor symptoms; Gait; Systematic review
| Introduction | ▴Top |
Parkinson’s disease (PD) is characterized as a neurodegenerative disorder that progressively starts with the loss of dopamine-producing neurons in the brain. This leads to both motor and non-motor symptoms. These symptoms include tremors, bradykinesia, rigidity, and postural instability, and impair patient quality of life, diminishing their mobility and independence [1-4]. Therapies often focus on physical therapy, pharmacological interventions and, in some cases, surgical procedures such as deep brain stimulation can be applied. However, these approaches have limitations, such as lower efficacy over time, side effects, and invasiveness [5].
Transcranial direct current stimulation (tDCS), on the other hand, is a non-invasive technique with potential benefits for PD patients [6, 7]. This technique involves a low electrical current delivered to the scalp that can modulate neuronal activity. Some studies suggest that it can improve motor function in patients with PD, for instance, in outcomes such as cadence, stride length, the Timed Up and Go (TUG) test, gait capacity, balance, and the Unified Parkinson’s Disease Rating Scale (UPDRS) III [8-10]. Despite that, the evidence is limited and inconsistent, highlighting the need to understand tDCS’s efficacy and safety.
Therefore, while tDCS shows some improvements in motor symptoms in PD patients, the literature remains fragmented, and clinical evidence remains scarce. This systematic review aims to evaluate the efficacy of tDCS as an adjuvant therapy for motor symptoms of patients with PD. By collecting data from various studies, we want to determine the consistency of tDCS effects across different methods, identify gaps in the literature, and provide a better understanding of this therapy and a non-invasive option. Also, it is a goal to guide future research directions, emphasizing both the benefits and limitations of tDCS in motor symptoms of PD.
| Methods | ▴Top |
Protocol and registration
This systematic review was carried out per the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines [11], and the registration of this research project was completed on the PROSPERO platform CRD4202457096.
Eligibility criteria
The inclusion criteria for articles were: 1) eligible randomized controlled trials (RCTs); 2) studies that included any form of tDCS in the PD population; and 3) articles that had a control group. Furthermore, the exclusion criteria were: 1) articles that compared different types of tDCS; 2) articles that compared different intensities of stimulation; and 3) articles lacking a specific analysis of the subgroup of patients with PD. No restrictions were applied regarding age, gender, healthcare professional’s classification, or the date of service use.
Search and selection strategy
A systematic search was conducted from inception to January 2025 across PubMed, Embase, and Cochrane Library databases, to identify key studies evaluating the impact of tDCS on motor therapy in individuals with PD. No filters were applied regarding language, target audience, or publication date. The specific search strategies employed for each database are detailed in Table 1. After the initial search, two authors independently removed the duplicates, screened titles and abstracts, and evaluated the articles in full for eligibility. A consensus-based discussion with a third author resolved discrepancies.
 Click to view |
Table 1. Search Strategy |
Data extraction and statistical analysis
The data from each article were distributed in a Google Spreadsheet table. The following information was included: article title, first author, year of publication, eligibility criteria, number of participants, type of randomization, intervention, control group, outcomes, and results.
Quality assessment and risk of bias
Two independent authors assessed the risk of bias in each RCT using the Cochrane Collaboration Risk of Bias tool version 2 (RoB 2). To evaluate small study effects for the primary outcomes, comparison-adjusted funnel plots were utilized.
| Results | ▴Top |
Search results
As seen in Figure 1, 429 studies were identified. After removing duplicates and applying eligibility criteria, five articles were included in the final analysis [10, 12-15].
 Click for large image |
Figure 1. PRISMA flow diagram for the systematic review and meta-analysis. Diagram illustrates the identification, screening, and inclusion process of studies for this systematic review. |
Description of studies and participants
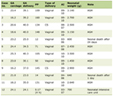
The five articles included were conducted in five countries: United States of America, Israel, Brazil, Italy, and Taiwan (Republic of China). In total, there were 176 participants, 122 men and 54 women. The patients had an average age of 67.13 years old. More information about the studies is provided in Table 2 [10, 12-15].
 Click to view |
Table 2. Characteristics of the Included
Studies |
Outcome result measures
This systematic review highlights several key findings from various studies about the effects of tDCS in improving motor outcomes for individuals with PD. One study reported that applying tDCS to the dorsolateral prefrontal cortex (DLPFC) (P < 0.001), the primary motor cortex (P = 0.048), and the cerebellum (P = 0.001) significantly improved dual-task gait speed performance when compared to baseline [10]. However, it is noteworthy that there were no significant enhancements in single walking or TUG performance compared to control groups.
Another article found a significant difference in gait cadence between the real tDCS and the sham groups (P = 0.014), indicating that tDCS may effectively modify gait patterns in PD patients [12]. There were no significant differences in other outcomes (such as gait speed and duration) between groups in his study. Furthermore, one study presented secondary analyses showing that tDCS, compared with sham, overall reduced self-reported severity of freezing of gait (FOG) (P = 0.05) and significantly increased daily living step counts (P = 0.04) [14]. Among individuals with mild-to-moderate FOG severity, tDCS in comparison with sham notably improved performance on FOG-provoking tests (P = 0.048) and self-reported experiences of freezing (P = 0.05).
A different study reported pronounced improvements in several motor function variables in the tDCS group compared to the sham group, including enhanced overall posture (P = 0.014), reduced lateral trunk inclination during standing (P = 0.013), increased total range of motion of the trunk (P = 0.012), and improved Functional Independence Measure (FIM) scores (P = 0.048) [13]. Lastly, another trial, also evaluating postural responses to tDCS, showed that the active group experienced a reduction in recovery time following postural perturbations (P < 0.001) and decreased reliance on prefrontal executive-attentional resources (P = 0.017), indicating enhanced movement automaticity. There was also a trend toward shorter medial gastrocnemius (MG) muscle onset latency in the active tDCS group post-intervention (P = 0.040), though this effect did not persist at follow-up (P = 0.056) [15].
Collectively, these studies underscore the potential of tDCS to enhance motor functions, including gait, posture, and overall mobility, in individuals with PD, highlighting its promise as a therapeutic intervention for managing motor symptoms associated with the condition.
Risk of bias
The risk of bias evaluation for all five included articles revealed an overall low risk of bias among all studies. Figure 2 presents further details, including assessing each domain of the analyses for every article.
 Click for large image |
Figure 2. Risk of Bias 2 Tool assessment. Risk of bias assessment for included studies using the Risk of Bias 2 Tool. The figure presents “traffic light” plots with domain-level judgments for each study and bar plots summarize the distribution of risk-of-bias judgments across domains. Green indicates low risk of bias, reflecting high methodological quality. |
Discussion
This systematic review investigated the role of tDCS as an adjunctive therapy for motor symptoms in PD, addressing an important gap regarding its efficacy and safety. Current treatments for PD are often limited by invasiveness, side effects, and reduced effectiveness over time, then tDCS offers a non-invasive alternative that can improve motor outcomes, including gait speed, posture, and balance. This review synthesized data from five RCTs, indicating that tDCS can enhance motor functions such as dual-task gait speed and trunk stability. However, outcome variability underscores the need to clarify further the most effective stimulation parameters and treatment protocols.
The observed motor improvements, such as those reported in dual-task gait speed and cadence, align with existing evidence that tDCS can modulate cortical excitability, which may enhance motor performance in PD patients [6, 16]. The studies included in this review suggest that stimulation of regions like the primary motor cortex and DLPFC might reinforce pathways affected by dopaminergic neuron loss, potentially offering patients improved motor stability [17, 18]. For example, one study demonstrated gains in gait speed with DLPFC stimulation [10]. Meanwhile, another trial reported enhanced cadence with motor cortex targeting, supporting the hypothesis that strategic electrode placement can produce specific motor benefits [12].
Additional findings, such as reductions in FOG and improvements in trunk posture and balance, indicate that tDCS might benefit more complex motor functions, which are typically challenging to treat with standard therapies [19, 20]. For instance, it was shown that tDCS could reduce FOG severity, particularly in patients with mild-to-moderate symptoms, which is significant given the limited efficacy of pharmacological interventions in addressing FOG [14]. Furthermore, two articles highlighted improvements in trunk stability and postural control, reduced recovery time after perturbations and decreased reliance on prefrontal executive-attentional resources, suggesting improved movement automaticity and postural stability in PD, showing that tDCS may support better balance and gait, both critical for maintaining mobility and reducing fall risk in PD [13, 15].
The variability in tDCS efficacy across studies reflects differences in stimulation protocols, highlighting the need to establish standardized parameters. Electrode placement, for example, greatly impacts which cortical areas are targeted [21]. Motor cortex stimulation has shown promise for basic motor functions, while DLPFC targeting affects dual-tasking abilities. Similarly, current intensity and duration may play a role in effectiveness, as some studies applied currents of 1 mA while others used 2 mA, with differing session lengths. Standardizing these variables across trials would likely improve comparability and help identify optimal protocols for PD.
The limited sample sizes and short-term follow-ups in many of the reviewed studies constrain the generalizability of the findings, as small cohorts and a lack of long-term data make it difficult to assess whether observed motor improvements are sustained or diminished post-stimulation. It is essential to determine whether repeated tDCS sessions are required for sustained benefits or if continuous application has a cumulative effect. Future studies should focus on longer follow-up periods and larger sample sizes to better understand the durability of tDCS benefits. Another critical limitation is the need for more reporting of adverse effects, making it challenging to evaluate the safety of tDCS for long-term use fully. Although tDCS is widely considered safe, thorough documentation of potential side effects is necessary to assess its feasibility as a regular treatment option.
This review suggests that tDCS complements existing treatment strategies for managing motor symptoms in PD, particularly for patients who do not respond well to traditional therapies. Policymakers and clinicians may consider integrating tDCS into clinical practice as an adjunctive treatment, especially in cases where invasive procedures are not viable. Moving forward, more extensive and standardized studies are needed to explore combination therapies and to assess the impact of tDCS on PD across different disease stages.
| Conclusion | ▴Top |
This systematic review provides preliminary evidence that tDCS may offer modest but meaningful improvements in motor symptoms for patients with PD, particularly in gait and postural control. However, the heterogeneity across protocols and sample limitations underscores the need for further standardized and large-scale trials. With continued investigation, tDCS holds promise as a valuable adjunctive therapy for PD, potentially improving patients’ mobility, stability, and quality of life.
Acknowledgments
None to declare.
Financial Disclosure
None to declare.
Conflict of Interest
None to declare.
Author Contributions
Conceptualization: Lucas B. Meira, Andrey T. Ferreira, Joao V.A. Fernandes and Andre de S.B. Oliveira. Project administration: Joao V.A. Fernandes and Andre de S.B. Oliveira. Formal analysis: Joao V.A. Fernandes. Writing original draft: Lucas B. Meira, Andrey T. Ferreira and Joao V.A. Fernandes. Writing review and editing: Lucas B. Meira, Andrey T. Ferreira and Joao V.A. Fernandes.
Data Availability
The authors declare that data supporting the findings of this study are available within the article.
| References | ▴Top |
- Balestrino R, Schapira AHV. Parkinson disease.
Eur J Neurol. 2020;27(1):27-42.
doi pubmed - Beitz JM. Parkinson’s disease: a review. Front Biosci
(Schol Ed). 2014;6(1):65-74.
doi pubmed - Kalia LV, Lang AE. Parkinson’s disease. Lancet.
2015;386(9996):896-912.
doi pubmed - Radhakrishnan DM, Goyal V. Parkinson’s disease: a
review. Neurol India. 2018;66(Supplement):S26-S35.
doi pubmed - Armstrong MJ, Okun MS. Diagnosis and treatment of Parkinson
disease: a review. JAMA. 2020;323(6):548-560.
doi pubmed - Benninger DH, Lomarev M, Lopez G, Wassermann EM, Li X,
Considine E, Hallett M. Transcranial direct current stimulation for the treatment of
Parkinson’s disease. J Neurol Neurosurg Psychiatry. 2010;81(10):1105-1111.
doi pubmed pmc - Cammisuli DM, Cignoni F, Ceravolo R, Bonuccelli U,
Castelnuovo G. Transcranial direct current stimulation (tDCS) as a useful rehabilitation
strategy to improve cognition in patients with Alzheimer’s disease and Parkinson’s
disease: an updated systematic review of randomized controlled trials. Front Neurol.
2022;12:798191.
doi pubmed pmc - Beretta VS, Conceicao NR, Nobrega-Sousa P, Orcioli-Silva D,
Dantas L, Gobbi LTB, Vitorio R. Transcranial direct current stimulation combined with physical
or cognitive training in people with Parkinson’s disease: a systematic review.
J Neuroeng Rehabil. 2020;17(1):74.
doi pubmed pmc - Liu X, Liu H, Liu Z, Rao J, Wang J, Wang P, Gong X, et al.
Transcranial direct current stimulation for Parkinson’s disease: a systematic review and
meta-analysis. Front Aging Neurosci. 2021;13:746797.
doi pubmed pmc - Wong PL, Yang YR, Huang SF, Fuh JL, Chiang HL, Wang RY.
Transcranial direct current stimulation on different targets to modulate cortical activity and
dual-task walking in individuals with Parkinson’s disease: a double blinded randomized
controlled trial. Front Aging Neurosci. 2022;14:807151.
doi pubmed pmc - Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC,
Mulrow CD, Shamseer L, et al. The PRISMA 2020 statement: an updated guideline for reporting
systematic reviews. Syst Rev. 2021;10(1):89.
doi pubmed pmc - Da Silva DCL, Lemos T, De Sa Ferreira A, Horsczaruk CHR, Pedron CA, De Carvalho Rodrigues E, De Oliveira LAS. Effects of acute transcranial direct current stimulation on GAIT kinematics of individuals with Parkinson disease. Top Geriatr Rehabil. 2018;34(4):262-268.
- De Icco R, Putorti A, Allena M, Avenali M, Dagna C,
Martinelli D, Cristina S, et al. Non-invasive neuromodulation in the rehabilitation of Pisa
syndrome in Parkinson’s disease: a randomized controlled trial. Front Neurol.
2022;13:849820.
doi pubmed pmc - Manor B, Dagan M, Herman T, Gouskova NA, Vanderhorst VG,
Giladi N, Travison TG, et al. Multitarget transcranial electrical stimulation for freezing of
gait: a randomized controlled trial. Mov Disord. 2021;36(11):2693-2698.
doi pubmed - Beretta VS, Orcioli-Silva D, Zampier VC, Moraca GAG, Pereira
MP, Gobbi LTB, Vitorio R. Eight sessions of transcranial electrical stimulation for postural
response in people with Parkinson’s disease: a randomized trial. Gait Posture.
2024;114:1-7.
doi pubmed - Pol F, Salehinejad MA, Baharlouei H, Nitsche MA. The effects
of transcranial direct current stimulation on gait in patients with Parkinson’s disease:
a systematic review. Transl Neurodegener. 2021;10(1):22.
doi pubmed pmc - Gbadeyan O, McMahon K, Steinhauser M, Meinzer M. Stimulation
of dorsolateral prefrontal cortex enhances adaptive cognitive control: a high-definition
transcranial direct current stimulation study. J Neurosci.
2016;36(50):12530-12536.
doi pubmed pmc - Magrinelli F, Picelli A, Tocco P, Federico A, Roncari L,
Smania N, Zanette G, et al. Pathophysiology of motor dysfunction in Parkinson’s disease
as the rationale for drug treatment and rehabilitation. Parkinsons Dis.
2016;2016:9832839.
doi pubmed pmc - Elsner B, Kugler J, Pohl M, Mehrholz J. Transcranial direct
current stimulation (tDCS) for idiopathic Parkinson’s disease. Cochrane Database Syst
Rev. 2016;7(7):CD010916.
doi pubmed pmc - Gomez Palacio Schjetnan A, Faraji J, Metz GA, Tatsuno M,
Luczak A. Transcranial direct current stimulation in stroke rehabilitation: a review of recent
advancements. Stroke Res Treat. 2013;2013:170256.
doi pubmed pmc - Opitz A, Yeagle E, Thielscher A, Schroeder C, Mehta AD,
Milham MP. On the importance of precise electrode placement for targeted transcranial electric
stimulation. Neuroimage. 2018;181:560-567.
doi pubmed pmc
This
article is distributed under the terms of the Creative Commons Attribution Non-Commercial 4.0
International License, which permits unrestricted non-commercial use, distribution, and
reproduction in any medium, provided the original work is properly cited.
Journal
of Neurology Research is published by Elmer Press Inc.