| Journal of Neurology Research, ISSN 1923-2845 print, 1923-2853 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Neurol Res and Elmer Press Inc |
| Journal website https://jnr.elmerpub.com |
Review
Volume 16, Number 2, June 2026, pages 67-76
Seizures and Epilepsy in Children With SARS-CoV-2 Infection: Clinical Phenotypes, Mechanisms, and Long-Term Outcomes
Department of Pediatric Neurology, “Karamandaneio” Children’s Hospital, Patras, Greecee
Manuscript submitted January 29, 2026, accepted March 19, 2026, published online May 4, 2026
Short title: Seizures in Children With COVID-19
doi: https://doi.org/10.14740/jnr1100
| Abstract | ▴Top |
Seizures have emerged as a common neurological manifestation of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection in children, encompassing a wide clinical spectrum ranging from isolated febrile seizures to severe inflammatory and immune-mediated neurological disorders. This narrative review aims to synthesize current evidence on the pathophysiological mechanisms, clinical phenotypes, diagnostic approach, management strategies, and long-term outcomes of seizures and epilepsy associated with pediatric coronavirus disease 2019 (COVID-19). A structured narrative literature review was conducted using PubMed and Embase, including studies published between 2020 and 2025 that addressed seizures or epilepsy in children aged 0–18 years in the context of acute SARS-CoV-2 infection, multisystem inflammatory syndrome in children (MIS-C), post-infectious autoimmune neurological syndromes, and long-term neurological sequelae. Available evidence indicates that most seizures occurring during pediatric SARS-CoV-2 infection represent acute symptomatic events, commonly related to fever or transient systemic and neuroinflammatory responses, and are generally associated with a favorable short-term outcome. Distinct clinical phenotypes have been described, including febrile seizures, afebrile acute symptomatic seizures, seizures associated with MIS-C, and seizures occurring in the setting of post-infectious autoimmune central nervous system disease. Although the long-term risk of epilepsy appears to be low for the majority of affected children, those with severe systemic inflammation, encephalopathy, autoimmune neurological involvement, abnormal neuroimaging findings, or status epilepticus may be at increased risk for persistent neurocognitive impairment or epileptological sequelae. Emerging data further suggest that seizure incidence and clinical characteristics in children may vary across different phases of the pandemic and circulating viral variants. Overall, seizures associated with pediatric SARS-CoV-2 infection are most often transient and self-limited; however, a clinically important subset of patients requires comprehensive neurodiagnostic evaluation and structured longitudinal follow-up. Prospective, multicenter pediatric studies with standardized neurodiagnostic protocols and long-term outcome assessment are needed to better define epilepsy risk and neurodevelopmental trajectories following COVID-19–related seizures.
Keywords: SARS-CoV-2; Pediatric seizures; Epilepsy; Febrile seizures; MIS-C; Neuroinflammation
| Introduction | ▴Top |
Since the emergence of the coronavirus disease 2019 (COVID-19) pandemic, increasing attention has been directed toward the neurological manifestations associated with infection by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). Although children generally experience milder respiratory illness than adults, neurological involvement has been increasingly recognized in pediatric populations and may involve both the central and peripheral nervous systems [1–3].
Among these manifestations, seizures have been repeatedly reported in association with pediatric SARS-CoV-2 infection across several clinical contexts, including acute infection, multisystem inflammatory syndrome in children (MIS-C), and post-infectious immune-mediated neurological disease [1, 4–7]. Current evidence indicates that most seizures occurring during SARS-CoV-2 infection in children represent acute symptomatic events related to fever, systemic illness, or transient neuroinflammatory responses, rather than direct viral injury to the central nervous system (CNS) [5, 6].
Nevertheless, a subset of pediatric patients may develop more complex neurological presentations, including afebrile seizures, status epilepticus (SE), encephalopathy, and, rarely, new-onset epilepsy [4, 7]. These manifestations have been described particularly in children with severe systemic inflammation, MIS-C, or post-infectious immune-mediated neurological complications [1, 2].
It is also important to recognize that children with preexisting epilepsy may experience transient seizure exacerbation during acute systemic illness, including SARS-CoV-2 infection. In most cases, seizure worsening appears to be related to fever, metabolic stress, sleep disruption, or difficulties with medication adherence rather than direct viral effects on neuronal tissue [8].
| Age-Dependent Mechanisms and Pathophysiology of Seizures in Pediatric SARS-CoV-2 Infection | ▴Top |
Seizures associated with SARS-CoV-2 infection in children arise from a complex interplay of inflammatory, immune-mediated, genetic, metabolic, and developmental factors. Unlike adults, in whom vascular, hypoxic, and degenerative mechanisms often predominate, pediatric seizure susceptibility is strongly modulated by neurodevelopmental stage, immune system maturation, and fever sensitivity. Importantly, current evidence suggests that direct viral neuroinvasion plays a limited role in seizure generation, with indirect mechanisms accounting for the majority of neurological manifestations in children [3, 9].
Systemic and neuroinflammation
Systemic inflammation is considered one of the principal mechanisms contributing to seizure susceptibility during pediatric SARS-CoV-2 infection. Acute COVID-19 and MIS-C are characterized by increased circulating levels of proinflammatory cytokines, including interleukin-6 (IL-6), interleukin-1β (IL-1β), and tumor necrosis factor-α (TNF-α), which are known to influence neuronal excitability and synaptic transmission [10, 11].
Experimental and clinical evidence indicates that inflammatory cytokines may lower seizure threshold through several mechanisms, including modulation of γ-aminobutyric acid (GABA)–mediated inhibition, enhancement of glutamatergic neurotransmission, and activation of microglial inflammatory pathways [11]. These neuroimmune interactions have been widely implicated in the pathophysiology of acute symptomatic seizures occurring during systemic infections.
In children with SARS-CoV-2 infection, systemic inflammation may also contribute to transient disruption of the blood–brain barrier (BBB), facilitating the entry of circulating cytokines and inflammatory mediators into the CNS and promoting neuroinflammatory responses even in the absence of direct viral invasion [10, 12]. This mechanism has been proposed as a plausible explanation for seizures occurring during otherwise uncomplicated systemic infection.
Although the neurotropic potential of SARS-CoV-2 has been investigated, current evidence supporting direct viral invasion of neuronal tissue as a major mechanism of seizure generation remains limited. Neurological symptoms associated with SARS-CoV-2 infection are thought to arise predominantly from indirect mechanisms, including systemic inflammation, immune dysregulation, and vascular or metabolic disturbances rather than direct cytopathic effects within the CNS [3, 9].
Taken together, available data suggest that inflammation-mediated neuronal hyperexcitability and transient neuroimmune dysregulation represent key contributors to seizure susceptibility in pediatric COVID-19, while direct viral neuroinvasion appears to play a less prominent role in most cases.
Immune-mediated and post-infectious neurological mechanisms
Post-infectious immune-mediated neurological complications represent an additional pathway through which seizures may occur in children following SARS-CoV-2 infection. A range of autoimmune and inflammatory CNS disorders have been reported in temporal association with COVID-19, including autoimmune encephalitis and demyelinating conditions such as acute disseminated encephalomyelitis (ADEM) and myelin oligodendrocyte glycoprotein antibody–associated disease (MOGAD) [13–15]. These disorders are characterized by immune-mediated inflammation affecting cortical and subcortical brain structures and may clinically manifest with seizures, encephalopathy, and focal neurological deficits.
MIS-C is a hyperinflammatory condition that occurs predominantly in pediatric populations several weeks after SARS-CoV-2 infection and is characterized by systemic inflammation, multiorgan involvement, and elevated inflammatory markers [1, 2]. Neurological manifestations have been reported in a substantial proportion of children with MIS-C and may include headache, encephalopathy, altered mental status, and seizures [2]. Although the precise mechanisms underlying neurological involvement remain incompletely understood, current evidence suggests that immune dysregulation, cytokine-mediated neuroinflammation, and endothelial activation may contribute to transient cerebral dysfunction in affected patients [1, 2].
Importantly, seizures occurring in the context of MIS-C are generally considered acute symptomatic events related to systemic inflammation and immune-mediated CNS dysfunction, rather than evidence of primary epileptogenic injury. Nevertheless, severe inflammatory burden, encephalopathy, or associated structural or inflammatory brain abnormalities may increase the risk of short-term neurological complications and warrant careful clinical monitoring.
Taken together, these observations support the concept that immune-mediated inflammatory processes represent an important contributor to seizure susceptibility in a subset of children with SARS-CoV-2 infection, particularly in those with post-infectious inflammatory syndromes.
Genetic susceptibility and developmental vulnerability
SARS-CoV-2 infection may act as a precipitating factor in children with underlying genetic or developmental predisposition to seizures. Fever and systemic inflammation can unmask latent channelopathies or developmental epileptic encephalopathies, particularly in children harboring pathogenic variants in genes such as sodium channel, neuronal, type 1, alpha subunit (SCN1A), protocadherin 19 (PCDH19), and other epilepsy-associated ion channel genes [16]. This phenomenon may mirror mechanisms observed in fever-sensitive genetic epilepsies, in which inflammatory stressors interact with intrinsic neuronal hyperexcitability.
Age-related differences in neuronal network maturation further modulate seizure susceptibility. During infancy and early childhood, excitatory synaptic activity predominates, inhibitory circuits are immature, and fever-related seizure thresholds are lower. These developmental features provide a biological explanation for the predominance of febrile and acute symptomatic seizures in younger children with COVID-19, as well as for the relative rarity of chronic epilepsy following infection in the absence of structural or immune-mediated injury [5, 6, 16, 17].
Metabolic, hypoxic, and systemic stressors
Systemic illness associated with moderate to severe COVID-19 may precipitate seizures through secondary metabolic and physiological disturbances. Fever, dehydration, electrolyte imbalance, hypoglycemia, and hypoxia are well-recognized seizure precipitants in childhood and may act synergistically with inflammatory and immune-mediated mechanisms [18, 19]. In critically ill children, these factors can contribute to prolonged or refractory seizures, including SE, particularly in the setting of multiorgan dysfunction or intensive care unit admission.
Indirect psychosocial and environmental influences
Beyond biological mechanisms, indirect psychosocial factors have played a significant role in seizure exacerbation during the COVID-19 pandemic. Disruption of sleep patterns, heightened psychological stress, reduced access to healthcare services, and challenges in medication adherence have been associated with seizure worsening in children with preexisting epilepsy, independent of direct viral effects [8, 20]. Although these factors do not represent direct consequences of SARS-CoV-2 neurotropism, they constitute an important contextual mechanism influencing seizure burden in pediatric populations during the pandemic period.
Summary of pathophysiological framework
Taken together, seizures in pediatric SARS-CoV-2 infection predominantly reflect indirect mechanisms related to systemic and neuroinflammation, immune-mediated CNS injury, genetic vulnerability, and developmental neurobiology, rather than direct viral invasion of neuronal tissue. The relative contribution of these mechanisms varies according to age, severity of systemic illness, and presence of preexisting neurological or genetic risk factors. This mechanistic framework provides the foundation for understanding the heterogeneous clinical phenotypes of seizures observed in children with COVID-19 and informs age-appropriate diagnostic and management strategies [1, 3, 4, 10, 13, 16].
The proposed pathophysiological mechanisms underlying seizures in pediatric SARS-CoV-2 infection are multifactorial and involve interactions between systemic inflammation, immune-mediated CNS effects, fever, and host susceptibility. These mechanisms and their potential interactions are summarized schematically in Figure 1.
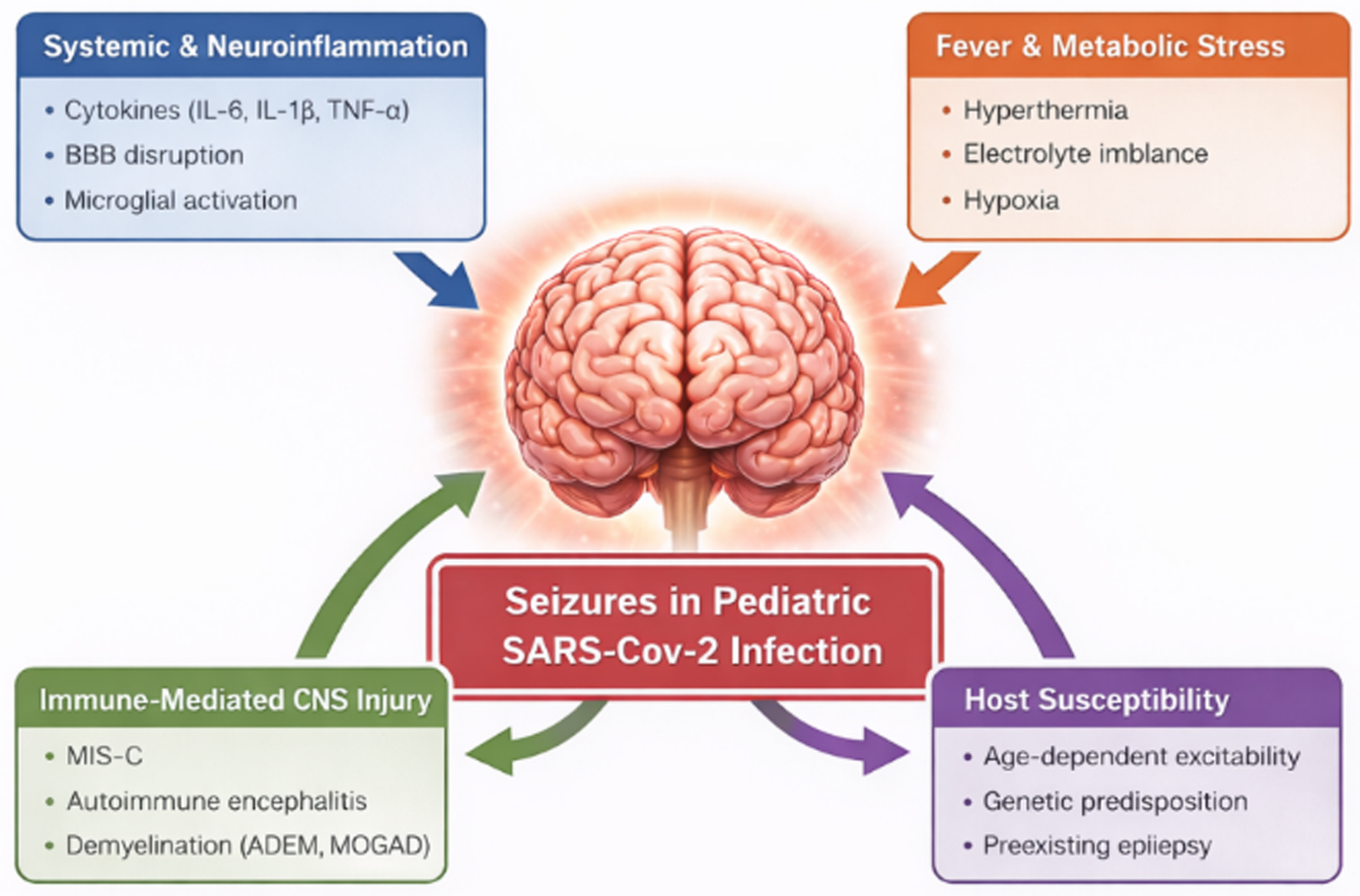 Click for large image | Figure 1. Proposed pathophysiological mechanisms underlying seizures in pediatric SARS-CoV-2 infection. Seizures in children arise from systemic and neuroinflammation, fever and metabolic stress, immune-mediated CNS injury, and host susceptibility. Cytokines, BBB disruption, and developmental or genetic factors modulate seizure risk and clinical severity. BBB: blood–brain barrier; CNS: central nervous system; SARS-CoV-2: severe acute respiratory syndrome coronavirus 2. |
| Clinical Phenotypes of Seizures in Pediatric SARS-CoV-2 Infection | ▴Top |
Seizures associated with SARS-CoV-2 infection in children represent a heterogeneous spectrum of clinical manifestations, reflecting differences in age-related susceptibility, systemic inflammatory responses, and the presence or absence of direct or immune-mediated CNS involvement. Current evidence suggests that seizure occurrence in pediatric COVID-19 is most often transient and related to acute systemic or inflammatory processes rather than permanent epileptogenic injury.
For conceptual clarity and clinical relevance, seizure presentations in children with SARS-CoV-2 infection can be broadly categorized into several principal clinical phenotypes: 1) febrile seizures, 2) afebrile acute symptomatic or encephalopathy-related seizures, 3) seizures associated with MIS-C, and 4) seizures occurring in the context of post-infectious autoimmune neurological disease. Additional clinical considerations include SE occurring across phenotypes, seizure exacerbation in children with preexisting epilepsy, and the need to distinguish infection-related seizures from vaccine-associated events. This framework facilitates clinical interpretation, risk stratification, and follow-up planning in affected patients [1–6, 13, 14].
Febrile seizures
Febrile seizures represent the most frequently reported seizure phenotype in children with SARS-CoV-2 infection. By definition, febrile seizures occur in neurologically healthy children between 6 months and 5 years of age in association with fever and in the absence of intracranial infection, metabolic disturbance, or prior unprovoked seizures. Several observational studies conducted during the COVID-19 pandemic have documented a notable number of febrile seizure presentations among children infected with SARS-CoV-2, particularly during periods dominated by the Omicron variant [4–6].
Compared with febrile seizures triggered by other respiratory viruses, some studies have suggested that COVID-19–associated febrile seizures may demonstrate a slightly higher proportion of complex features, including prolonged duration, focal semiology, or recurrence within 24 h. Nevertheless, the overall clinical course appears similar to that observed with febrile seizures related to other viral infections [5, 6].
Several cohorts have reported seizures associated with fever in children outside the classical age range, including infants younger than 6 months and children older than 5 years [4, 5]. In such cases, the term fever-associated seizures may be more appropriate than classical febrile seizures, as these events fall outside the standard diagnostic definition. The atypical age distribution observed in COVID-19 cohorts may reflect heightened inflammatory responses, variant-specific viral characteristics, or increased medical surveillance during the pandemic.
Despite these variations, current evidence suggests that the short- and long-term prognosis of COVID-19–associated febrile seizures is generally favorable, with most children experiencing complete recovery and a low risk of subsequent epilepsy in the absence of additional neurological risk factors [6, 17].
Afebrile acute symptomatic seizures and encephalopathy-related seizures
Afebrile seizures occurring in temporal association with SARS-CoV-2 infection represent a distinct clinical phenotype and are typically classified as acute symptomatic seizures. These events arise in close temporal relation to systemic or neurological insults and usually reflect transient cerebral dysfunction rather than chronic epileptogenic processes.
Such seizures may occur during the acute phase of infection or shortly thereafter and are frequently accompanied by neurological symptoms including altered mental status, headache, confusion, or behavioral changes [9]. Neuroimaging findings in these cases are often mild and reversible. One of the most frequently reported patterns is mild encephalopathy with reversible splenial lesion (MERS), characterized by transient diffusion restriction in the splenium of the corpus callosum [21].
Electroencephalographic findings in affected children commonly demonstrate generalized background slowing or focal epileptiform discharges, consistent with acute encephalopathic processes rather than established epilepsy [22]. Clinical outcomes are generally favorable, with resolution of neurological symptoms and imaging abnormalities within days to weeks. Available follow-up data suggest that progression to chronic epilepsy is uncommon in this phenotype [9, 21].
Seizures associated with MIS-C
Seizures occurring in the context of MIS-C represent a more severe and clinically distinct phenotype. MIS-C is characterized by profound systemic hyperinflammation, cardiovascular involvement, and multiorgan dysfunction following SARS-CoV-2 infection, with neurological manifestations reported in a significant proportion of affected patients [1, 2].
Neurological involvement in MIS-C may include encephalopathy, headache, focal neurological deficits, and seizures, which may present either as isolated acute symptomatic seizures or, less commonly, as SE. Neuroimaging abnormalities and electroencephalographic changes consistent with diffuse cerebral dysfunction have also been reported in this context.
Compared with febrile or uncomplicated afebrile seizures, seizures associated with MIS-C are more frequently linked to severe systemic illness, prolonged hospitalization, and admission to intensive care units, often requiring aggressive immunomodulatory therapy including intravenous immunoglobulin and corticosteroids [1].
Although many children demonstrate substantial neurological recovery following appropriate treatment, emerging evidence suggests that MIS-C–related neurological involvement may carry an increased risk of persistent neurocognitive or behavioral sequelae, highlighting the importance of structured neurological follow-up in this population [2, 23].
Post-infectious autoimmune and inflammatory epileptogenic syndromes
A smaller subset of pediatric patients may develop seizures as part of post-infectious autoimmune neurological syndromes following SARS-CoV-2 infection. Reported conditions include autoimmune encephalitis, particularly anti-N-methyl-D-aspartate (NMDA) receptor encephalitis, as well as inflammatory demyelinating disorders such as ADEM and MOGAD [13–15].
Seizures in this context are often recurrent and may be focal or generalized, frequently occurring in association with encephalopathy, psychiatric symptoms, movement disorders, or focal neurological deficits. Unlike febrile or uncomplicated acute symptomatic seizures, seizures related to autoimmune CNS disorders may reflect sustained inflammatory injury and increased epileptogenic potential.
Delayed diagnosis or delayed initiation of immunotherapy may increase the risk of persistent seizures or evolution into chronic epilepsy in some cases. Consequently, early recognition and prompt initiation of appropriate immunomodulatory therapy represent key determinants of neurological outcome in this subgroup [13, 14].
SE across phenotypes
SE has been reported as an uncommon but potentially severe manifestation across several pediatric COVID-19–related seizure phenotypes. Cases have been described in the setting of severe acute infection, MIS-C, and autoimmune encephalitis, suggesting that multiple pathogenic mechanisms may contribute to prolonged seizure activity in affected children [24, 25].
Both convulsive and non-convulsive forms of SE have been reported, often in children with significant systemic illness, metabolic disturbances, or inflammatory CNS involvement. When SE occurs in a child with SARS-CoV-2 infection, clinicians should consider a broad differential diagnosis including metabolic derangements, CNS inflammation, encephalopathy, and immune-mediated neurological disease.
Although the overall incidence of SE in pediatric COVID-19 appears low, its occurrence is associated with increased morbidity and greater need for intensive neurological monitoring and management.
Seizure exacerbation in children with preexisting epilepsy
Children with established epilepsy may experience transient seizure exacerbation during SARS-CoV-2 infection. Several factors may contribute to increased seizure frequency in this setting, including fever, systemic illness, sleep disruption, psychosocial stress, and difficulties with medication adherence during acute illness [25, 26].
Current evidence suggests that SARS-CoV-2 infection does not appear to confer a higher long-term risk of seizure worsening or epilepsy progression compared with other systemic infections. In most reported cases, seizure control returns to baseline following recovery from the acute illness [8, 25, 26].
Nevertheless, careful clinical monitoring is recommended during infection, particularly in children with poorly controlled epilepsy or complex neurological comorbidities.
Distinction from vaccine-associated seizures
An important clinical consideration is the distinction between seizures temporally associated with SARS-CoV-2 infection and those occurring after COVID-19 vaccination. Large population-based studies and pharmacovigilance data have demonstrated that COVID-19 vaccines are not associated with an increased risk of new-onset epilepsy or sustained seizure worsening in children, including those with preexisting epilepsy [27].
Rare post-vaccination seizures that have been reported are typically febrile and self-limited, reflecting transient immune responses rather than direct neurotoxic or epileptogenic effects of vaccination. Clear differentiation between infection-related and vaccine-associated events is therefore essential for accurate clinical interpretation and public health communication.
| Mechanisms of Seizures in SARS-CoV-2 Infection | ▴Top |
The mechanisms underlying seizures in children with SARS-CoV-2 infection remain incompletely understood. Current evidence suggests that seizures occurring during COVID-19 are most often related to systemic inflammatory responses, fever, metabolic disturbances, or secondary neurological complications, rather than direct viral invasion of the CNS. Several potential mechanisms have been proposed, and these likely interact in a multifactorial manner depending on the clinical context and the individual patient’s susceptibility [3, 6, 7]. The principal mechanisms that have been suggested in the literature are summarized in Table 1 [1–9, 13–15, 18, 19, 24–26, 28–30].
 Click to view | Table 1. Risk-Stratified Diagnostic Considerations for Children Presenting With Seizures in the Context of SARS-CoV-2 Infection |
Systemic inflammation and cytokine-mediated neuroexcitability
One of the most widely discussed mechanisms involves the systemic inflammatory response associated with SARS-CoV-2 infection. Elevated levels of pro-inflammatory cytokines, including IL-6, TNF-α, and other mediators, have been observed in patients with severe COVID-19 and may contribute to neuronal hyperexcitability [6, 7].
Cytokine-mediated inflammation may influence neuronal function through several pathways, including alterations in neurotransmitter balance, activation of microglial cells, and modulation of ion channel activity, which may lower the seizure threshold. In pediatric patients, this mechanism may be particularly relevant in conditions characterized by exaggerated immune responses, such as MIS-C [1].
However, it is important to note that most of the available data supporting these mechanisms are derived from observational studies and extrapolation from other inflammatory neurological conditions, and direct causal relationships remain incompletely established.
Fever and metabolic triggers
Fever represents another important contributor to seizure occurrence in children with SARS-CoV-2 infection. As with other viral illnesses, elevated body temperature can increase neuronal excitability and may precipitate seizures in susceptible individuals, particularly in young children predisposed to febrile seizures [4–6].
In addition to fever, systemic illness may lead to metabolic disturbances, including electrolyte imbalances, dehydration, and hypoxia, all of which can further reduce seizure threshold. These systemic factors likely explain a substantial proportion of seizures occurring during acute SARS-CoV-2 infection, particularly in otherwise healthy children who experience isolated febrile seizures.
BBB dysfunction and neuroinflammation
Another proposed mechanism involves disruption of the BBB in the context of systemic inflammation. Increased vascular permeability and endothelial activation may facilitate the entry of inflammatory mediators into the CNS, potentially contributing to neuronal dysfunction and seizure generation [3, 7].
In some cases, neuroimaging findings and cerebrospinal fluid analyses suggest the presence of secondary neuroinflammatory processes, even in the absence of detectable viral RNA in the CNS. These observations support the hypothesis that immune-mediated mechanisms may play a role in certain neurological manifestations associated with COVID-19.
Direct or indirect CNS involvement
Direct neuroinvasion by SARS-CoV-2 has been proposed as a potential mechanism of neurological complications; however, current evidence suggests that direct viral invasion of the CNS appears to be relatively uncommon, particularly in pediatric patients [6, 7].
Neurological symptoms such as anosmia and ageusia have been attributed to viral involvement of olfactory pathways, but these findings do not necessarily imply widespread CNS infection. In most reported pediatric cases, seizures appear to occur in association with systemic illness, inflammatory responses, or secondary neurological conditions, rather than as a direct consequence of viral replication within neural tissue.
Taken together, these observations suggest that seizures associated with SARS-CoV-2 infection in children likely arise from a combination of systemic, inflammatory, and occasionally immune-mediated mechanisms, rather than a single pathogenic pathway.
| Management Considerations | ▴Top |
The management of seizures occurring in children with SARS-CoV-2 infection generally follows standard pediatric seizure management principles, with additional considerations related to the underlying infection and associated systemic or neurological complications. In most cases, treatment strategies do not differ substantially from those used for seizures associated with other viral illnesses.
Acute seizure management
Acute seizures in children with COVID-19 should be managed according to established pediatric seizure treatment protocols. Initial management includes ensuring airway protection, adequate oxygenation, and hemodynamic stability. When pharmacological treatment is required, benzodiazepines remain the first-line therapy for acute seizure termination, followed by second-line antiseizure medications when seizures persist [8].
In cases of SE, rapid escalation of treatment is essential and may require additional antiseizure medications and intensive care monitoring. The management approach remains consistent with established SE treatment algorithms in pediatric patients.
Management of underlying triggers
Because many seizures occurring during SARS-CoV-2 infection are acute symptomatic events, identification and treatment of underlying triggers is an important component of management. These may include fever, metabolic disturbances, dehydration, hypoxia, or systemic inflammation.
Antipyretic therapy, fluid and electrolyte correction, and treatment of systemic infection may contribute significantly to seizure control in affected children. In patients with neurological complications such as encephalitis or MIS-C, management may also include immunomodulatory therapies such as intravenous immunoglobulin or corticosteroids as part of the broader treatment strategy for the underlying condition [1].
Children with preexisting epilepsy
Children with established epilepsy who develop SARS-CoV-2 infection should generally continue their baseline antiseizure medications without interruption. Breakthrough seizures may occur during acute illness due to fever, sleep disruption, or metabolic stress; however, in most cases seizure control returns to baseline following recovery from the infection [25, 26].
Careful monitoring is recommended during acute illness, particularly in children with drug-resistant epilepsy or complex neurological comorbidities.
Follow-up and neurological evaluation
Most children who experience seizures during SARS-CoV-2 infection recover without persistent neurological complications. Nevertheless, clinical follow-up may be warranted in certain situations, particularly in cases involving prolonged seizures, SE, encephalopathy, or suspected autoimmune neurological disorders.
Follow-up evaluation may include neurological examination, electroencephalography, or neuroimaging depending on the clinical presentation and the persistence of neurological symptoms.
| Prognosis and Long-Term Outcomes | ▴Top |
The overall prognosis of seizures occurring in children with SARS-CoV-2 infection appears to be generally favorable. In the majority of reported cases, seizures are transient events occurring during the acute phase of illness and resolve without persistent neurological complications.
Most children presenting with febrile seizures or acute symptomatic seizures in the context of COVID-19 experience complete clinical recovery. Available studies suggest that these seizures typically reflect temporary systemic or inflammatory effects of infection, rather than permanent epileptogenic injury to the CNS [4–6].
Children with neurological complications such as encephalitis, MIS-C, or autoimmune inflammatory disorders may experience a more complex clinical course. In these cases, seizures may occur as part of broader neurological involvement and may require prolonged monitoring and follow-up. Nevertheless, many patients show substantial neurological improvement following appropriate treatment of the underlying condition [1, 2, 13].
Current evidence does not support a clear association between SARS-CoV-2 infection and a significant increase in the risk of new-onset chronic epilepsy in the pediatric population. Most available data are derived from observational studies and case series with relatively limited follow-up, and therefore long-term outcomes remain an area of ongoing investigation [8, 17].
In children with preexisting epilepsy, SARS-CoV-2 infection may occasionally be associated with temporary increases in seizure frequency during acute illness. However, seizure control typically returns to baseline following recovery, and there is currently no evidence that COVID-19 leads to sustained worsening of epilepsy in most pediatric patients [25, 26].
Overall, while seizures represent an important neurological manifestation of SARS-CoV-2 infection in children, current evidence suggests that long-term neurological outcomes are favorable in the majority of cases, particularly when seizures occur as isolated events during acute infection.
| Limitations of Current Evidence | ▴Top |
Despite the growing number of publications describing seizures in children with SARS-CoV-2 infection, the current body of evidence remains subject to several important limitations.
First, a large proportion of the available data is derived from case reports, small case series, and retrospective observational studies, which limits the ability to establish definitive causal relationships between SARS-CoV-2 infection and seizure occurrence. Many studies also include heterogeneous patient populations with varying clinical presentations, ranging from mild febrile illness to severe systemic inflammatory syndromes, making direct comparisons between studies challenging.
Second, differences in study design, diagnostic criteria, and reporting practices across institutions may contribute to variability in the reported incidence and characteristics of seizures in pediatric COVID-19 cohorts. In some cases, seizures may represent coincidental events or manifestations of underlying neurological conditions, rather than direct consequences of SARS-CoV-2 infection.
Another important limitation is the relatively short duration of follow-up in most published studies. As a result, the long-term neurological outcomes of children who experience seizures during SARS-CoV-2 infection, including the potential risk of developing chronic epilepsy, remain incompletely understood.
Finally, the evolving nature of the pandemic, including the emergence of new viral variants and widespread vaccination, may influence the epidemiology and clinical presentation of neurological manifestations in children. Continued surveillance and prospective studies will therefore be important to better clarify the relationship between SARS-CoV-2 infection and seizure disorders in the pediatric population.
| Conclusions | ▴Top |
Seizures represent an important neurological manifestation that may occur in children with SARS-CoV-2 infection. The spectrum of seizure presentations is heterogeneous and includes febrile seizures, acute symptomatic seizures, seizures associated with MIS-C, and seizures occurring in the context of post-infectious autoimmune neurological disorders.
Current evidence suggests that most seizures observed during pediatric COVID-19 are transient events related to systemic illness, fever, or inflammatory processes, rather than direct viral injury to the CNS. In the majority of cases, affected children experience favorable clinical outcomes, with resolution of seizures following recovery from the acute infection.
Nevertheless, certain clinical scenarios—including MIS-C, encephalitis, or autoimmune neurological syndromes—may be associated with more complex neurological involvement and require careful clinical evaluation and follow-up.
Given that much of the available evidence derives from observational studies and case reports, further prospective research with longer follow-up is needed to better clarify the mechanisms, long-term neurological outcomes, and potential risk of epilepsy following SARS-CoV-2 infection in the pediatric population.
Overall, continued investigation and systematic data collection will be essential for improving our understanding of the relationship between COVID-19 and seizure disorders in children, and for optimizing clinical management and long-term care.
Acknowledgments
The author has no acknowledgments to declare.
Financial Disclosure
The author declares no financial support.
Conflict of Interest
The author declares no conflict of interest.
Informed Consent
This study did not involve human participants or identifiable patient data. Therefore, informed consent was not required.
Author Contributions
The author conceptualized and designed the study, performed the literature search, data extraction, and critical appraisal, interpreted and analyzed the data, and drafted and revised the manuscript. The author approved the final version of the manuscript and takes full responsibility for the integrity and accuracy of the work.
Data Availability
No new data were generated or analyzed in this study. All data supporting the findings of this review are derived from published literature and are available within the article.
Abbreviations
ADEM: acute disseminated encephalomyelitis; BBB: blood–brain barrier; CNS: central nervous system; COVID-19: coronavirus disease 2019; CSF: cerebrospinal fluid; EEG: electroencephalography; FIRDA: frontal intermittent rhythmic delta activity; GABA: γ-aminobutyric acid; IL-6: interleukin-6; IL-1β: interleukin-1β; MERS: mild encephalitis/encephalopathy with reversible splenial lesion; MIS-C: multisystem inflammatory syndrome in children; MOGAD: myelin oligodendrocyte glycoprotein–associated disease; MOG: myelin oligodendrocyte glycoprotein; MRI: magnetic resonance imaging; NMDA: N-methyl-D-aspartate; PCDH19: protocadherin 19; PCR: polymerase chain reaction; SARS-CoV-2: severe acute respiratory syndrome coronavirus 2; SCN1A: sodium channel, neuronal, type 1, alpha subunit; SE: status epilepticus; TNF-α: tumor necrosis factor-α
| References | ▴Top |
- Feldstein LR, Rose EB, Horwitz SM, Collins JP, Newhams MM, Son MBF, Newburger JW, et al. Multisystem inflammatory syndrome in U.S. children and adolescents. N Engl J Med. 2020;383(4):334-346.
doi pubmed - LaRovere KL, Riggs BJ, Poussaint TY, Young CC, Newhams MM, Maamari M, Walker TC, et al. Neurologic involvement in children and adolescents hospitalized in the United States for COVID-19 or multisystem inflammatory syndrome. JAMA Neurol. 2021;78(5):536-547.
doi pubmed - Antoon JW, Hall M, Howard LM, Herndon A, Freundlich KL, Grijalva CG, Williams DJ. COVID-19 and acute neurologic complications in children. Pediatrics. 2022;150(5):e2022058167.
doi pubmed - Pascarella A, Maglione M, Lenta S, Sciorio E, Mancusi R, Tucci C, Angrisani F, et al. Seizures in children with SARS-CoV-2 infection: epidemiological, clinical and neurophysiological characterization. Children (Basel). 2022;9(12):1923.
doi pubmed - Han MJ, Heo JH, Hwang JS, Jang YT, Lee M, Kim SJ. Incidence of febrile seizures in children with COVID-19. J Clin Med. 2023;12(3):1076.
doi pubmed - Jiang J, Shen Z, Cao J. Seizures in febrile children with SARS-CoV-2 infection: clinical features, short-term follow-up. BMC Pediatr. 2024;24(1):257.
doi pubmed - Kong J, Lyu S, Nam SO, Kim YO, Ahn JY, Yeom JS, Lee SH, et al. Influence of SARS-CoV-2 omicron variant infection on seizure occurrence among children: The large-scale multicenter study. Brain Dev. 2025;47(3):104341.
doi pubmed - Cross JH, Kwon CS, Asadi-Pooya AA, Balagura G, Gomez-Iglesias P, Guekht A, Hall J, et al. Epilepsy care during the COVID-19 pandemic. Epilepsia. 2021;62(10):2322-2332.
doi pubmed - Boronat S. Neurologic care of COVID-19 in children. Front Neurol. 2020;11:613832.
doi pubmed - Almutairi MM, Sivandzade F, Albekairi TH, Alqahtani F, Cucullo L. Neuroinflammation and Its Impact on the Pathogenesis of COVID-19. Front Med (Lausanne). 2021;8:745789.
doi pubmed - Li G, Bauer S, Nowak M, Norwood B, Tackenberg B, Rosenow F, Knake S, et al. Cytokines and epilepsy. Seizure. 2011;20(3):249-256.
doi pubmed - Lee MH, Perl DP, Nair G, Li W, Maric D, Murray H, Dodd SJ, et al. Microvascular Injury in the Brains of Patients with COVID-19. N Engl J Med. 2021;384(5):481-483.
doi pubmed - Hilado M, Banh M, Homans J, Partikian A. Pediatric autoimmune encephalitis following COVID-19 infection. J Child Neurol. 2022;37(4):268-272.
doi pubmed - Peters J, Alhasan S, Vogels CBF, Grubaugh ND, Farhadian S, Longbrake EE. MOG-associated encephalitis following SARS-COV-2 infection. Mult Scler Relat Disord. 2021;50:102857.
doi pubmed - Tenembaum S, Chitnis T, Ness J, Hahn JS, International Pediatric MSSG. Acute disseminated encephalomyelitis. Neurology. 2007;68(16 Suppl 2):S23-36.
doi pubmed - Guerrini R, Genton P, Bureau M, et al. Fever, COVID-19, and genetic epilepsies. Seizure. 2023;109:34-40.
doi - Hesdorffer DC, Benn EK, Cascino GD, Hauser WA. Is a first acute symptomatic seizure epilepsy? Mortality and risk for recurrent seizure. Epilepsia. 2009;50(5):1102-1108.
doi pubmed - Trinka E, Hofler J, Zerbs A. Causes of status epilepticus. Epilepsia. 2012;53(Suppl 4):127-138.
doi pubmed - Abend NS, Wusthoff CJ, Goldberg EM, Dlugos DJ. Electrographic seizures and status epilepticus in critically ill children and neonates with encephalopathy. Lancet Neurol. 2013;12(12):1170-1179.
doi pubmed - Bruni O, Giallonardo M, Sacco R, Ferri R, Melegari MG. The impact of lockdown on sleep patterns of children and adolescents with ADHD. J Clin Sleep Med. 2021;17(9):1759-1765.
doi pubmed - Lee YY, Kim YO. Afebrile benign convulsions with or without a reversible splenial lesion in two pediatric patients with COVID-19. BMC Pediatr. 2023;23(1):198.
doi pubmed - Galanopoulou AS, Ferastraoaru V, Correa DJ, Cherian K, Duberstein S, Gursky J, Hanumanthu R, et al. EEG findings in acutely ill patients investigated for SARS-CoV-2/COVID-19: A small case series preliminary report. Epilepsia Open. 2020;5(2):314-324.
doi pubmed - Avittan H, Kustovs D. Cognition and mental health in pediatric patients following COVID-19. Int J Environ Res Public Health. 2023;20(6):5061.
doi pubmed - Dono F, Nucera B, Lanzone J, Evangelista G, Rinaldi F, Speranza R, Troisi S, et al. Status epilepticus and COVID-19: a systematic review. Epilepsy Behav. 2021;118:107887.
doi pubmed - Kuroda N. Epilepsy and COVID-19: Updated evidence and narrative review. Epilepsy Behav. 2021;116:107785.
doi pubmed - Lim J, Moon JU. Impact of the COVID-19 pandemic on seizure control in pediatric epilepsy: risk factors and clinical outcomes. Healthcare (Basel). 2025;13(2):172.
doi pubmed - Klein NP, Lewis E, Goddard K, et al. Seizure risk after COVID-19 vaccination in children. Pediatrics. 2023;151(2):e2022059274.
doi - Francoeur C, Alcamo AM, Robertson CL, Wainwright MS, Roa JD, Lovett ME, Stulce C, et al. Severe pediatric neurological manifestations with SARS-CoV-2 or MIS-C hospitalization and new morbidity. JAMA Netw Open. 2024;7(6):e2414122.
doi pubmed - Graus F, Titulaer MJ, Balu R, Benseler S, Bien CG, Cellucci T, Cortese I, et al. A clinical approach to diagnosis of autoimmune encephalitis. Lancet Neurol. 2016;15(4):391-404.
doi pubmed - Trinka E, Cock H, Hesdorffer D, Rossetti AO, Scheffer IE, Shinnar S, Shorvon S, et al. A definition and classification of status epilepticus—Report of the ILAE Task Force on Classification of Status Epilepticus. Epilepsia. 2015;56(10):1515-1523.
doi pubmed
This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (CC BY 4.0), which permits unrestricted use, distribution, and reproduction in any medium, including commercial use, provided the original work is properly cited.
Journal of Neurology Research is published by Elmer Press Inc.