| Journal of Neurology Research, ISSN 1923-2845 print, 1923-2853 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Neurol Res and Elmer Press Inc |
| Journal website https://jnr.elmerpub.com |
Review
Volume 16, Number 2, June 2026, pages 55-66
The Orexin System in Pediatric Learning Difficulties: Neurobiological Foundations for Educational, Psychoeducational and Behavioral Therapies
Meng Kiat Tana, b , Kok Hwee Chiaa
aMerlion Paediatric Therapy Clinic Pte Ltd., Singapore 299552, Singapore
bCorresponding Author: Meng Kiat Tan, Merlion Paediatric Therapy Clinic Pte Ltd., Singapore 299552, Singapore
Manuscript submitted February 11, 2026, accepted April 9, 2026, published online April 30, 2026
Short title: Orexin in Pediatric Learning Difficulties
doi: https://doi.org/10.14740/jnr1104
| Abstract | ▴Top |
The orexin (also known as hypocretin) system, located in the lateral hypothalamus, plays a crucial role in regulating arousal, attention, motivation, and memory consolidation through its extensive neural projections across the brain. This article examines the neurobiological foundations linking orexin dysfunction to pediatric learning difficulties and explores how educational, psychoeducational and behavioral therapies may leverage orexin-mediated pathways to improve learning outcomes. The orexin system’s integrative functions in coordinating sleep-wake cycles, reward processing, arousal and cognitive performance provide a theoretical framework for understanding specific learning difficulties, attention deficits, and developmental conditions. Educational therapy approaches that optimize circadian timing, enhance intrinsic motivation, and support memory consolidation may work, in part, through supporting orexin system function. Similarly, educational therapy addressing arousal regulation, executive function, alongside behavioral therapy targeting adaptive behavioral responses and habit formation, may benefit from orexin-informed strategies. However, significant limitations remain in current research, including the lack of direct evidence linking orexin modulation to therapeutic outcomes. Future directions should prioritize translational and controlled intervention studies to establish evidence-based practices that bridge neuroscience discoveries with educational applications for children with learning difficulties.
Keywords: Orexin; Learning difficulties; Neurodevelopment disorders; Educational therapy; Behavioral therapy; Early intervention
| Introduction | ▴Top |
Learning difficulties affect approximately 10–20% of school-aged children globally, encompassing a broad spectrum of conditions including specific learning disorders (SLD) and neurodevelopmental disorders, including attention-deficit/hyperactivity disorders (ADHD), autism spectrum disorders (ASD), and intellectual disabilities [1, 2]. These conditions impose significant challenges not only on affected children and their families but also on educational systems worldwide. Traditional approaches to interventions have primarily focused on behavioral and cognitive strategies, often without explicit consideration of underlying neurobiological mechanisms that may influence learning capacity.
The emergence of “neurodivergent and neuroeducation” as a discipline has highlighted the potential for neuroscientific discoveries to inform educational practice. However, a significant gap persists between clinical findings and classroom applications. Many neurobiological insights remain confined to research settings, failing to translate into practical interventions that could benefit children with learning challenges. This disconnect is particularly pronounced in the realm of neuromodulatory systems that regulate fundamental processes including cognitive learning, attention, motivation, and memory consolidation.
As claimed in the review conducted by Jaszberenyi et al [3], the orexin system plays a central role in coordinating multiple brain functions essential for cognition and learning. This unique position makes it particularly relevant for understanding and potentially addressing learning difficulties that often involve disruptions across multiple domains of functioning.
The orexin system: a critical neural network
The orexin system was discovered in 1998 by two independent research groups, the Yanagisawa group [4] and the Sutcliffe group [5], initially recognized for its role in feeding behavior and sleep-wake regulation. However, subsequent research has revealed far more extensive functions, establishing orexin as a critical regulator of arousal, attention, motivation, memory and learning. The orexin system comprises approximately 50,000 to 80,000 neurons clustered exclusively in the lateral hypothalamus and perifornical area (Fig. 1) [6]. These neurons project throughout the central nervous system (CNS), influencing diverse brain regions and functions [3].
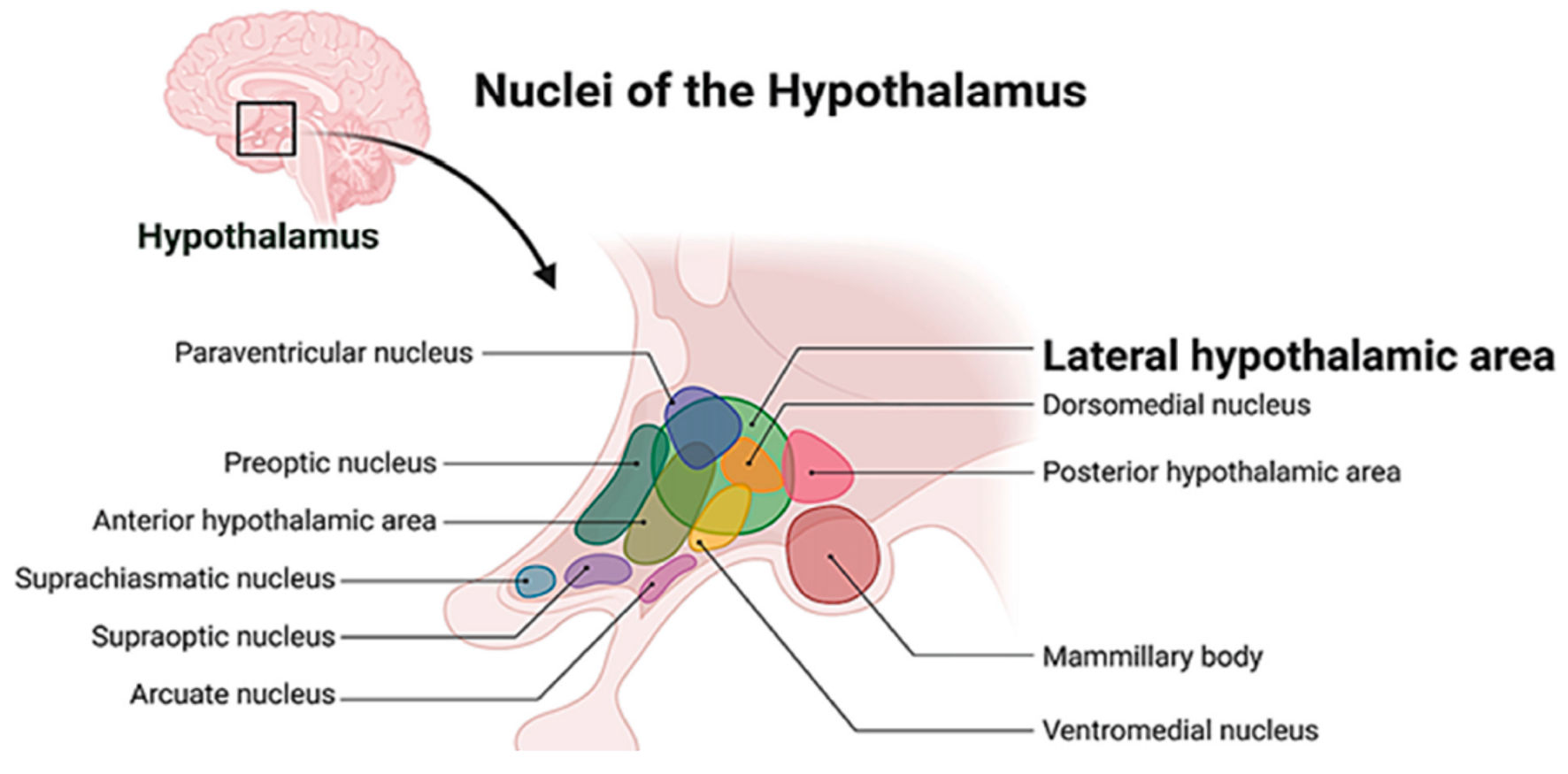 Click for large image | Figure 1. The localization of lateral hypothalamic area. |
The orexin system produces two neuropeptides, orexin-A and orexin-B (Table 1) [3–5]. They act on two G-protein coupled receptors (GPCRs), orexin-1-receptor (OX1R) and orexin-2-receptor (OX2R). These receptors are distributed differentially across the brain, with OX1R showing high expression in areas associated with reward and emotion, while OX2R is more prominent in arousal and sleep-wake regulatory regions. This anatomical organization provides the foundation for orexin’s multifaceted roles in behavior and cognition [7–9].
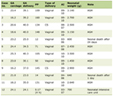 Click to view | Table 1. Differences Between Neurotransmitters and Neuropeptides |
What makes the orexin system particularly relevant to learning difficulties is its function as a state-dependent modulator. Rather than simply turning processes on or off, orexin neurons adjust their activity based on internal states, emotions and moods, conscious thoughts processes, environmental demands, and behavioral context. This dynamic regulation allows the system to optimize cognitive performance for current circumstances, a function that may be disrupted in various learning difficulties [10–13].
Objectives and scope
The primary aim of this article is to examine the role of the orexin system in pediatric learning processes and to explore how educational therapy (EdTx), psychoeducational therapy (PsyEdTx) and behavioral therapy (BeTx) may leverage orexin-mediated pathways to improve outcomes for children with learning difficulties broadly defined, including SLD, ADHD and related neurodevelopmental conditions such as ASD that impact academic achievement and adaptive functioning. While the orexin-informed principles apply broadly across learning difficulties, their evidential base is strongest in ADHD and requires cautious extension to isolated SLD or ASD. This framework should be interpreted with this gradient of evidence in mind. We propose that understanding orexin functions provides a neurobiological framework for conceptualizing learning difficulties and developing more targeted educational interventions. Our secondary aim is to establish a theoretical foundation for orexin-informed educational practices while acknowledging current limitations and identifying future research directions. This article is intended for researchers, clinicians and educators in neuroeducation working with neurodivergent children with learning difficulties, and for those seeking to understand the neurobiological foundations of learning, attention, motivation and memory. The scope encompasses both basic neuroscience research on orexin function and applied research on EdTx, PsyEdTx and BeTx, with particular emphasis on pediatric populations.
| The Orexin System: Neurobiological Development | ▴Top |
The orexin system’s unique anatomical organization underlies its integrative functions in learning and cognition. Orexin neurons project extensively throughout the CNS, creating one of the most widespread neuromodulatory networks in the brain. The developmental trajectory of the orexin system has important implications for understanding pediatric learning difficulties.
Orexin neurons begin to differentiate during the second trimester of human gestation, and orexin peptides can be detected by the third trimester, though the system continues to mature throughout childhood and adolescence, paralleling the development of cognitive functions it helps regulate. Orexin neuron number reaches adult levels by early childhood (around 2 to 3 years in rodent models with similar trajectories inferred in humans), but receptor expression, synaptic connectivity and functional integration with arousal, reward, and prefrontal circuits continue to refine through late childhood and adolescence [10, 12, 13]. This protracted maturation includes enhanced integration with circadian systems during puberty, contributing to delayed sleep phase in many adolescents. During early childhood, orexin system connectivity continues to expand and refine, with the period of greatest synaptic plasticity, roughly corresponding to the preschool and early elementary years, coinciding with continued orexin system development [7, 8, 13]. This overlap suggests that orexin dysfunction during critical periods could have lasting effects on learning capacity. During childhood and adolescence, the orexin system undergoes significant developmental changes that parallel the maturation of learning and attention capacities, with orexin neuron number reaching adult levels by early childhood, but receptor expression, neural connectivity, and functional integration continuing to develop throughout the pediatric period. This protracted development may contribute to age-related differences in learning capacity and vulnerability to various learning difficulties [10, 12–14].
Adolescence represents another important developmental phase for the orexin system, where its role in regulating sleep-wake cycles becomes increasingly important as circadian rhythms shift during puberty, with many adolescents experiencing delayed sleep phase that may reflect changes in orexin system function. These circadian disruptions can significantly impact learning and academic performance [12, 14–16]. Individual differences in orexin system development may contribute to the heterogeneity seen in learning difficulties, with some children having genetic variations affecting orexin production, receptor sensitivity, or neural connectivity, while others experience environmental influences that alter orexin system development, such as chronic stress, sleep disruption, or nutritional factors. The prolonged development of the orexin system also suggests potential for intervention during critical periods, with educational and behavioral intervention approaches that support optimal orexin system function during key developmental phases potentially having lasting benefits for learning and academic achievement [17–19].
| Orexin and Core Learning Processes | ▴Top |
The extensive connectivity of the orexin system enables it to coordinate multiple brain functions relevant to learning. Ascending projections from orexin neurons target brainstem arousal nuclei, including the prefrontal cortex (PFC), nucleus accumbens (NAc), locus coeruleus (LC) for norepinephrine synthesis, raphe nuclei (RN) for serotonin synthesis, ventral tegmental area (VTA) for dopamine synthesis, tuberomammillary nucleus (TMN) for histamine synthesis, and pedunculopontine tegmental nucleus (PTN) for acetylcholine synthesis. Through these connections, orexin helps maintain cortical arousal and attention, fundamental prerequisites for effective learning (Fig. 2) [20, 21].
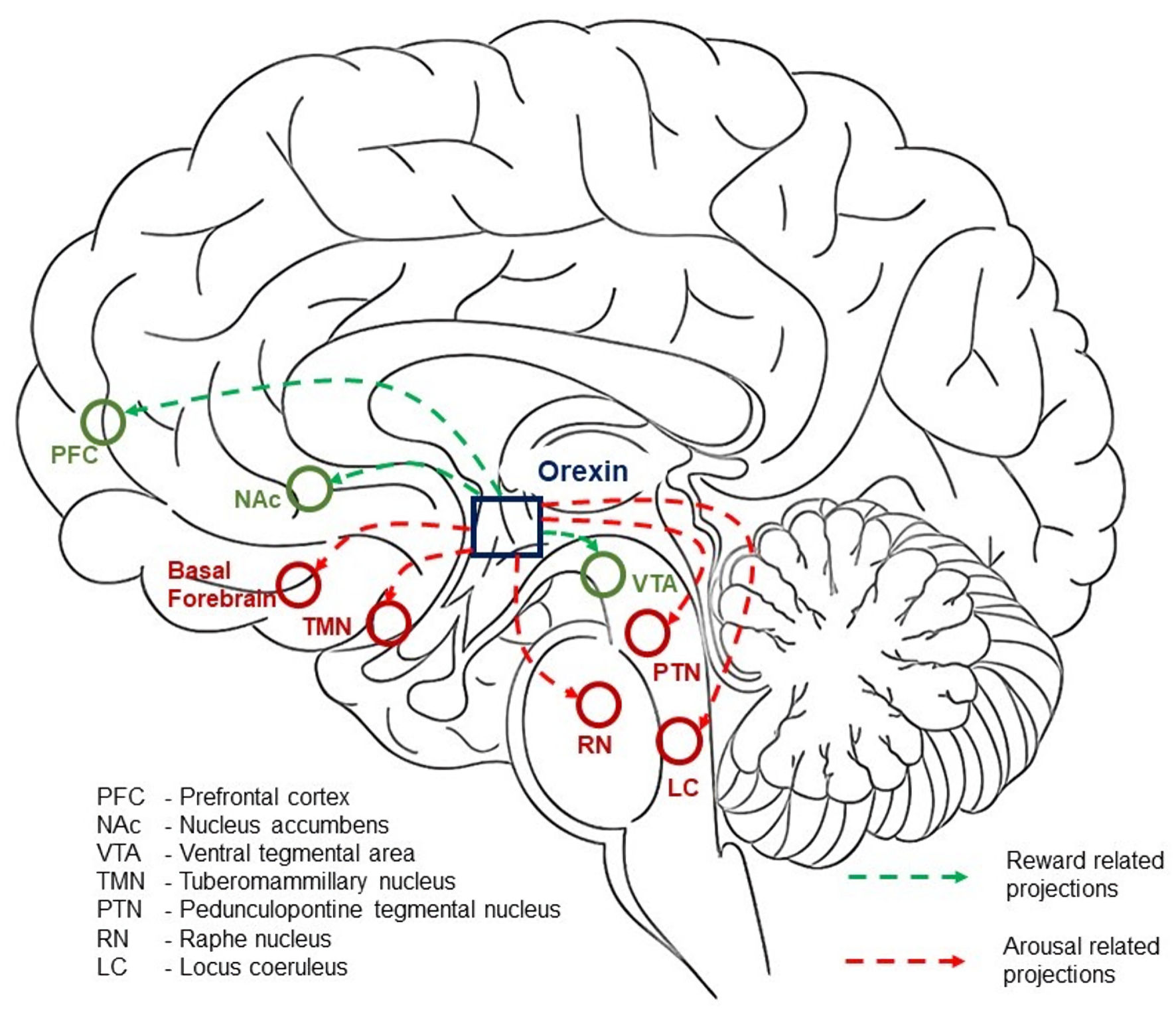 Click for large image | Figure 2. Orexin signaling pathway. |
Arousal, attention, vigilance and sleep-wake functions
Arousal regulation and sleep-wake control
The orexin system maintains arousal and wakefulness by projecting to multiple brain regions. Orexin neurons project to several arousal-promoting brain areas, including the LC, RN, TMN, PTN and basal forebrain, where they enhance the neurotransmission of norepinephrine, serotonin, histamine, and acetylcholine. This arousal-promoting effect works mainly through the OX2R receptor. Studies show that blocking only this receptor is enough to induce sleep, while blocking the OX1R receptor has little effect on sleepiness [16, 20, 21]. The orexin system coordinates information from our internal body clock, sleep pressure (time period of staying awake), and motivational factors to keep our sleep-wake states stable and prevent unwanted switches between sleeping and waking. When this system is disrupted, people experience fragmented sleep-wake patterns and may have dream-sleep (rapid eye movement (REM)) intrude into wakefulness. This stabilizing role makes the orexin system essential for maintaining steady wakefulness and optimal alertness, which are crucial for learning and cognitive performance [22, 23].
Sustained and selective attention mechanism
According to Villano et al [24], attention is the mental process that allows us to focus on important information while ignoring distractions. It includes several types of attention: sustained attention (maintaining focus over time), selective attention (choosing what to focus on), and divided attention (managing multiple tasks). Sustained attention relies heavily on the orexin system, which acts as the brain’s main regulator of arousal and wakefulness. Recent brain studies show that orexin signaling keeps specific neurons in the PFC active, which helps control attention and maintain arousal brain states. Loss of orexin, as occurs in the sleep disorder such as narcolepsy, lead to marked impairment in sustained attention and cognitive efficiency [25, 26].
Selective attention depends on how the orexin system works together with cholinergic system. Orexin neurons project to the basal forebrain and activate cholinergic neuros that synthesis acetylcholine for maintaining attention. This activation happens through the OXR2 receptor and results in increased acetylcholine release in the PFC, a brain area crucial for focused thinking [24, 27]. When acetylcholine levels increase, the brain becomes better at distinguishing important signals from background noise, which improves our ability to detect relevant information and stay focused on our goals. This partnership between orexin and acetylcholine is essential for maintaining both general alertness and selective focus during mentally challenging activities. A practical example of this occurs in classrooms, where children need to continually use selective attention to concentrate on classrooms instructions while filtering out distractions like background conversations or movements from other students [28–30].
Memory formation, consolidation and retrieval
The hippocampal formation receives substantial orexin innervation through both direct projections to hippocampal subfields and indirect influence via cholinergic and norepinephrinergic pathways. This connectivity positions orexin to influence multiple aspects of memory processing, from initial encoding through consolidation and retrieval.
Synaptic plasticity and encoding mechanisms
Activation of orexin receptors in the hippocampus, particularly in the CA1 region and dentate gyrus, enhances long-term potentiation (LTP) by coordinating neurotransmitters such as acetylcholine, glutamate, and norepinephrine, which are essential for learning and memory. When orexin-A binds to OX1R receptors, it increases NMDA receptor–mediated calcium influx, activating key intracellular pathways (including ERK/MAPK and PI3K/Akt) that help maintain LTP (Table 2) [31–34]. This encoding process also stimulates the production of synaptic proteins such as postsynaptic density protein, strengthens the structural connections between neurons, and supports long-term information storage. Importantly, these orexin-driven neuroplastic effects are state-dependent, they are strongest during periods of alertness and focused attention, which may explain why learning efficiency improves when individuals are engaged awake and attentive. During developmental stages, this mechanism also provides neuroprotective benefits, enhancing resilience against stress and environmental factors that might otherwise disrupt learning and memory consolidation [34].
 Click to view | Table 2. Activation of Key Intracellular Pathway for Long-Term Potentiation (LTP) via Binding of Orexin and Its Receptors |
Emotional memory enhancement
Orexin modulates emotional learning through its connections with the amygdala, particularly via the basolateral amygdala pathways that influence hippocampal-dependent memory formation. The amygdala plays a central role in enhanced memory for emotional experiences, demonstrating increased activity during both encoding and retrieval of emotional content [35, 36]. This neural circuit explains why emotionally significant events form stronger, more durable memories through orexin interactions with the amygdala and stress hormone systems. These amygdala-hippocampal circuits facilitate long-term retention of emotionally salient material, supporting recent findings about the enhancing effects of emotionally engaged educational contexts [37].
Memory consolidation and sleep-wake coordination
Memory consolidation, the process by which newly learned information becomes stable and retrievable, depends on dynamic interactions between the orexin system and key memory circuits. Orexin neurons display distinct patterns of activity during the consolidation, and retrieval phases of memory [32]. Beyond wakeful learning, the orexin system also coordinates sleep-wake transitions that are vital for effective memory consolidation. Although orexin neurons are predominantly active during wakefulness, their firing patterns influence how memories are reorganized and strengthened during sleep through modulation of acetylcholine release and hippocampal activity [32, 38–40]. Consequently, sleep disturbances can disrupt consolidation and further hinder learning, an issue particularly relevant for children with learning disorders who often experience sleep irregularities. Interventions that promote optimal arousal and attention during learning, alongside sleep hygiene strategies, can therefore enhance both orexin-mediated encoding and sleep-dependent consolidation, supporting more efficient and lasting memory formation [12, 41].
Memory retrieval
Memory retrieval involves orexin modulation of attention and arousal systems that facilitate access to stored information. During retrieval, optimal orexin signaling helps maintain the focused attention necessary to search memory stores and evaluate retrieved information [32, 35]. Experimental evidence demonstrates that blockade of orexin receptors in the basolateral amygdala impairs memory consolidation through the amygdala-hippocampal pathway. This confirms the vital role of this system in learning and memory processes. These findings suggest that children with retrieval difficulties may benefit from strategies that optimize their arousal and attention states during testing or performance situations, leveraging the orexin system’s natural role in facilitating memory access [35].
Learning and cognitive flexibility
Executive function, neural plasticity, and prefrontal modulation
Orexin supports executive function and learning by coordinating activity in the PFC, the brain region for planning, attention and cognitive flexibility. It works through two main pathways. The first acts directly, where orexin activates layer VI PFC neurons through OX2R receptors, improving cognitive flexibility and helping regulate anxiety [25]. The second works indirectly through the basal forebrain, which releases acetylcholine, a neurotransmitter vital for attention and memory, into the PFC. The more orexin is released, the more acetylcholine is produced, boosting wakefulness and attention. When orexin-A is delivered intranasally, it further increases acetylcholine and glutamate levels in the PFC, enhancing neuron activity and cognitive performance [42–44].
Orexin also strengthens neural plasticity, the brain’s ability to form and refine connections during learning. It regulates how glutamate affects specialized neurons in layer V of the PFC, improving how these neurons communicate and store information [45]. During learning, orexin helps specific medial PFC networks activate and later reactivate during memory recall, linking what is learned to motivation and goal-directed behavior. Together, these processes allow orexin to align arousal, attention, and motivation, forming a powerful system for efficient learning [46].
Cognitive flexibility and working memory
The orexin system is essential for cognitive flexibility (the ability to switch between different tasks or strategies) and working memory, which allows us to temporarily hold and manipulate information. When orexin signaling is disrupted, these executive functions will be impaired. For example, chronic jet lag impairs cognitive flexibility in mice by reducing orexin neuron activity in the hypothalamus, particularly affecting the medial orbitofrontal cortex, a PFC region critical for flexible thinking [47]. Remarkably, intranasal orexin-A administration reverses these deficits, restoring both cognitive flexibility and working memory, which demonstrates that circadian rhythm disturbances impair executive function by disrupting orexin input to the PFC. Orexin receptor activity in the basal forebrain also facilitates cognitive flexibility tasks such as discrimination learning, with OX1R stimulation improving performance and OX1R blockade impairing it, likely through modulation of acetylcholine release to the cortex [44, 47].
Motivation and goal-directed behavior
Integration of arousal, reward, and cognitive persistence
Orexin neurons project extensively to key reward and cognitive control regions, including the VTA, NAc, PFC and amygdala [48, 49]. This broad network allows orexin to act as a bridge between arousal regulation and motivational drive, linking physiological readiness with cognitive engagement during learning. Activation of OX1R receptors on dopaminergic neurons in the VTA and NAc heightens neuronal excitability and promotes phasic dopamine release, sustaining motivation and reward anticipation. Unlike dopamine, which primarily encodes reward prediction errors, orexin provides the energizing component of motivation, maintaining goal-directed behavior and cognitive effort even when immediate rewards are absent [50–52].
Through this mechanism, orexin supports cognitive persistence, or the capacity to sustain attention and effort during demanding tasks. Continuous orexinergic signaling coordinates arousal regulation, selective attention, and reward valuation, enabling individuals to remain engaged despite frustration or delayed gratification [37]. By maintaining dopaminergic activity within the VTA–PFC pathway, orexin prevents premature disengagement and promotes motivational endurance, a capacity essential for extended learning or problem-solving sequences requiring sustained focus [48].
Intrinsic versus extrinsic motivation and reinforcement dynamics
Orexin-dopamine interactions vary with the type of motivation driving learning. During intrinsic motivation, including learning driven by curiosity, interest, or mastery, there is strong orexin-dopamine coupling and enhanced connectivity between reward centers (i.e., VTA, NAc) and cognitive control regions at the PFC and anterior cingulate cortex (ACC). In contrast, extrinsic motivation, based on external incentives, produces weaker and more transient activation, relying mainly on dopaminergic signaling with minimal orexin involvement. Consequently, orexin appears central to the neural sustainability of intrinsic motivation and self-directed learning [37]. Repeated successful learning experiences reinforce this orexin-dopamine circuitry, strengthening associations between effort and reward and building academic self-efficacy [48]. However, chronic failure or discouragement can blunt orexin and dopamine responsiveness, reducing motivational energy and creating negative feedback loops that contribute to learned helplessness and academic disengagement.
Clinical implications for learning difficulties
In children with neurodevelopmental disorders such as ASD, ADHD, and SLD, disruptions in orexin-dopamine coupling may underlie characteristic difficulties with sustained attention and delayed reward processing. Neuroimaging studies reveal weakened connectivity between hypothalamic orexin centers and the mesocorticolimbic dopamine system, resulting in reduced reinforcement signaling during task success. This diminished feedback weakens activation in the ACC and dorsolateral PFC, impairing error monitoring, metacognitive control, and goal maintenance. Consequently, children with poor orexin-dopamine integration may lose effort prematurely during complex learning, leading to task avoidance and cumulative motivational decline. Understanding this neurobiological mechanism underscores the importance of interventions that enhance alertness, intrinsic motivation, and positive reinforcement cycles to restore persistence and resilience in learning [37, 48].
| Orexin Dysfunction in Pediatric Learning Difficulties | ▴Top |
ADHD
ADHD represents the condition with the strongest evidence for orexin system involvement in pediatric learning difficulties. Children with ADHD characteristically display altered reward sensitivity, and executive function deficits consistent with orexin dysregulation. Clinical studies demonstrate reduced orexin-A/B levels in children with ADHD, correlating with severity of inattention. Additionally, dysregulation of orexin-acetylcholine interactions may contribute to impaired cortical arousal and attentional instability characteristic of ADHD [48, 53].
The core symptoms of ADHD align well with known orexin functions. Problems with arousal regulation in ADHD, where a child can be hyper-reactive to some inputs yet under-responsive to others, are consistent with disrupted orexin control of wakefulness and state-dependent responsiveness [54]. Orexin helps sustain cortical arousal over time via activation of basal-forebrain cholinergic systems and by supporting cortical neurotransmission. This may explain why children with ADHD may do well on short tasks but struggle with prolonged attention [55]. Executive function impairments involving working memory, cognitive flexibility, and planning difficulties implicate PFC regions receiving orexin innervation. Furthermore, the altered reward sensitivity observed in ADHD may reflect disrupted orexin-dopamine interactions in reward circuits [48, 56].
Other neurodevelopmental conditions
While evidence is strongest for ADHD, emerging research suggests orexin dysfunction may contribute to learning difficulties in other neurodevelopmental conditions, though this remains an area requiring further investigation. Children with SLD including dyslexia, dyscalculia, and dysgraphia often show comorbid attention difficulties. Whether these reflect primary orexin dysfunction or secondary effects remains unclear. However, understanding orexin’s role in sustained attention and working memory may inform educational and behavioral interventions that optimize timing and arousal states during instruction for these populations [57]. To date, direct biomarker studies measuring orexin-A/B levels specifically in children with SLD or ASD remain scarce. The present framework therefore extrapolates from ADHD models, and future research should prioritize targeted orexin biomarker studies in these populations.
| EdTx: Orexin-Informed Interventions | ▴Top |
Principles and framework of educational and behavioral therapies
Understanding orexin system dysfunction in pediatric learning difficulties provides a neurobiological foundation for targeted non-pharmacological interventions. EdTx represents evidence-based approaches aligning with orexin-mediated mechanisms underlying attention, memory, learning, and motivation. It targets academic skills, executive functioning, and metacognitive strategies to improve performance in areas like reading, writing, or problem-solving.
EdTx, recognized by the World Health Organization (WHO) under procedural code 93.82 since 1986 in WHO’s International Classification of Diseases (ICD), Ninth Edition, Clinical Modifications, Volume 3 [58, 59], bridges neuroscience and education. While medical professionals use the ICD or Diagnostic and Statistical Manual of Mental Disorders (DSM) published by the American Psychiatric Association (APA) [1], educational therapists consult the Educator’s Diagnostic Manual of Disabilities and Disorders (EDM) [60], classified according to the 13 disability categories in the Individuals with Disabilities Education Act (IDEA, 2004).
EdTx focuses on academic skill-building and cognitive strategies, such as phonics instruction or metacognitive techniques, to address SLD. In relation to orexin, EdTx optimizes arousal and attention through timed tasks and reduced cognitive load, potentially stabilizing orexin-mediated cortical activation. Duman et al [61] have demonstrated in their studies EdTx’s effectiveness in reducing SLD in addition to increasing self-esteem and decreasing behavioral problems in children. Educational therapists collaborate with schools and families to implement individualized educational plans (IEPs), assistive technologies, and flexible instructional strategies tailored to each child’s evolving neurocognitive profile [19].
PsyEdTx, a subset of EdTx, combines psychotherapeutic techniques, including cognitive behavior therapy (CBT), mindfulness based cognitive therapy (MBCT), and emotion regulation behavior management (ERBM) training, with educational interventions to address the complex interplay between orexin system dysfunction, learning difficulties, and emotional-behavioral challenges in children with neurodevelopmental conditions [62–64]. This is also relevant to orexin via modulation of amygdala-hippocampal circuits and stress responses that influence motivational and memory consolidation pathways [35, 36].
In contrast, BeTx is a clinical intervention within the broader umbrella of psychotherapy, rooted in learning theories, such as operant conditioning and cognitive-behavioral principles, aimed at modifying maladaptive behaviors and promoting adaptive ones. Its relevance to orexin lies in establishing sleep-wake routines and reinforcement schedules that support stable orexin neuron activity patterns and habit formation via dopamine interactions [8, 14, 48]. Some overlapping with PsyEdTx, BeTx employs structured techniques like reinforcement, modeling, and parent training to target behaviors impacting learning, attention, or sleep, such as impulsivity or poor sleep onset. BeTx is widely used to address symptoms of neurodevelopmental disorders, including ADHD and sleep-wake disorders [65, 66], as defined in the DSM [1] and ICD-11 [67].
Given the protracted maturation of the orexin system, intervention strategies should be adapted to the child’s developmental stage. Younger children (preschool/early elementary) may benefit from highly structured, play-based EdTx and BeTx routines to support emerging connectivity and plasticity [7, 8, 13]. Adolescents could incorporate later school start times or circadian-aligned scheduling to accommodate pubertal shifts in orexin-circadian coupling [14, 15]. Clinicians and therapists should therefore consider developmental age when designing orexin-informed intervention programs.
Optimizing arousal and attention through orexin-aligned routines
The orexin system functions as the master regulator of arousal and wakefulness [3]. Interventions that align with natural orexin neuron activity patterns enhance learning capacity and memory. EdTx could schedule cognitively demanding tasks during mid-morning when orexin signaling and cortisol levels optimally support alertness. For children with ADHD showing delayed sleep phase patterns, EdTx and BeTx could accommodate later classes start times or schedule critical learning during peak orexin-mediated arousal periods [32, 48]. Predictable, low-distraction environments with consistent arrangements and visual schedules could support stable orexin-mediated arousal states [8, 17]. This may reduce anticipatory anxiety and environmental unpredictability, thereby promoting more stable arousal state conducive to sustained attention. When combined with brief movement breaks (as in structured transitions), these routines could further enhance orexin signaling by activating orexin neurons through locomotor and motivational pathways, similar to how physical activity augments norepinephrinergic and cholinergic transmission [22]. Such state-dependent modulation may improve receptor sensitivity over repeated sessions by aligning behavioral context with optimal internal arousal. BeTx could incorporate brief physical activity intervals (3–5 min) between tasks, as movement activates orexin neurons and enhances norepinephrinergic and cholinergic signaling [22].
Enhancing memory consolidation through coordinated sleep-wake cycles
The orexin system coordinates sleep-wake transitions, making sleep hygiene critical for memory consolidation. BeTx could establish consistent sleep-wake schedules that support healthy orexin neuron activity patterns synchronized with circadian rhythms [8, 14]. It could implement calming pre-sleep routines that facilitate natural decline in orexin neuron activity, including dimming lights and avoiding screens [23, 68]. BeTx could create optimal bedroom environments that support orexin system quieting during sleep, allowing memory consolidation through orexin-modulated hippocampal activity patterns [32, 69].
Supporting executive function through prefrontal pathway optimization
Orexin’s dual pathways to the PFC, direct OX2R-mediated excitation and indirect modulation via basal forebrain cholinergic neurons, inform interventions targeting executive functions. EdTx could break complex tasks into manageable steps to reduce working memory demands and prevent cognitive overwhelm that dysregulates orexin-mediated arousal [18, 43]. It could implement external aids including visual schedules and graphic organizers that reduce cognitive load on prefrontal systems depending on orexin-stimulated acetylcholine release [42]. PsyEdTx could teach metacognitive strategies to monitor arousal and emotional states together with cognitive performance, enabling children to recognize their orexin system’s natural fluctuations [70].
Fostering intrinsic motivation through dopamine circuit engagement
Orexin-dopamine coupling in reward circuits provides specific targets for motivation-enhancing interventions. EdTx could design learning experiences that connect to children’s interests to activate robust orexin-dopamine integration, where orexin projections to VTA sustain dopamine activity and promote connectivity between reward and cognitive control circuits [37]. BeTx could structure appropriately challenging tasks, as repeated successful experiences enhance orexin-VTA dopaminergic activity and strengthen academic self-efficacy [48, 61]. EdTx could provide frequent feedback that sustains orexin-mediated motivational arousal and offer meaningful choices that support autonomous learning [71, 72].
Psychoeducation for families and educators
Comprehensive psychoeducation empowers families and teachers within an orexin-informed framework [64]. It could provide accessible explanations of how the orexin system influences learning, helping caregivers recognize that difficulties reflect orexin dysfunction rather than willful behavior and training them to recognize arousal dysregulation signs, enabling timely supportive strategies [8]. BeTx could engage stakeholders in setting realistic, orexin-informed goals that acknowledge natural variations in orexin-dependent functions.12
Integration of mindfulness-based learning
The orexin system’s connections with basolateral amygdala circuits inform interventions that address emotional learning aspects [35, 36]. PsyEdTx could create psychologically safe environments that enhance memory consolidation through orexin’s amygdala connections, with BeTx demonstrating significant improvements in behavioral problems [37, 61]. PsyEdTx could teach stress management strategies such as mindfulness-based stress reduction (MBSR) that helps children to regulate the orexin system and hypothalamic-pituitary adrenal (HPA) axis stress response system to maintain optimal arousal states [3]. EdTx could explicitly teach that learning difficulties result from neurobiological differences, reducing shame and enhancing self-compassion [73].
| Limitations and Future Research Considerations | ▴Top |
Current research limitations
Application of orexin research to pediatric learning difficulties faces significant limitations, primarily due to the scarcity of direct evidence linking orexin function to educational interventions. While theoretical connections between orexin, arousal regulation, and learning are compelling, the lack of pediatric-specific investigations is a major gap, as the developing orexin system in children may differ functionally from that of adults [19, 23]. To overcome these limitations, educational and clinical approaches should integrate comprehensive psychoeducational diagnostic assessment, evaluation, and profiling (PDAEP) using established framework such as the Cattell–Horn–Carroll (CHC) theory of human cognitive abilities [74]. These provide structured means to understand cognitive strengths and weaknesses while allowing targeted intervention design that considers orexin-related factors, including arousal regulation, sleep-wake rhythms, attention span, memory and motivation [19]. Future research should prioritize large-scale longitudinal and randomized controlled trials to establish the efficacy of orexin-informed interventions across developmental stages. Biomarker validation studies are needed to develop reliable, noninvasive measures of orexin activity for pediatric use [75, 76].
| Conclusions and Clinical Implications | ▴Top |
The orexin system plays a central role in regulating arousal, attention, motivation, memory, and learning, making it a key neurobiological foundation for understanding pediatric learning difficulties. Through its extensive neural connections, orexin links motivation with attention, and supports memory consolidation essential for academic success. Indirect indicators such as sleep quality, attention variability, and arousal regulation can provide useful insights. Intervention planning should integrate orexin-informed principles within evidence-based frameworks. While a single therapy like EdTx can be effective for specific, isolated learning difficulties, it is generally less efficacious than a multimodal approach by integrating EdTx, PsyEdTx and BeTx for complex or comorbid pediatric learning difficulties. They represent promising non-pharmacological applications of these principles, offering practical pathways to enhance learning outcomes for children facing cognitive and motivational challenges.
Acknowledgments
None to declare.
Financial Disclosure
None to declare.
Conflict of Interest
None to declare.
Informed Consent
Not applicable.
Author Contributions
Meng Kiat Tan: conceptualization, formal analysis, investigation, writing—original draft (all sections except “EdTx: Orexin-Informed Interventions”). Kok Hwee Chia: conceptualization, validation, writing—section of “EdTx: Orexin-Informed Interventions,” reviewing and editing.
Data Availability
The authors declare that data supporting the findings of this study are available within the article.
| References | ▴Top |
- APA, American Psychiatric Association. Diagnostic and statistical manual of mental disorders (5th ed., text rev.). American Psychiatric Publishing. 2022.
- Khodeir MS, El-Sady SR, Mohammed HAER. The prevalence of psychiatric comorbid disorders among children with specific learning disorders: a systematic review. Egypt J Otolaryngol. 2020;36:57.
doi - Jaszberenyi M, Thurzo B, Bagosi Z, Vecsei L, Tanaka M. The orexin/hypocretin system, the peptidergic regulator of vigilance, orchestrates adaptation to stress. Biomedicines. 2024;12(2):448.
doi pubmed - Sakurai T, Amemiya A, Ishii M, Matsuzaki I, Chemelli RM, Tanaka H, Williams SC, et al. Orexins and orexin receptors: a family of hypothalamic neuropeptides and G protein-coupled receptors that regulate feeding behavior. Cell. 1998;92(4):573-585.
doi pubmed - de Lecea L, Kilduff TS, Peyron C, Gao X, Foye PE, Danielson PE, Fukuhara C, et al. The hypocretins: hypothalamus-specific peptides with neuroexcitatory activity. Proc Natl Acad Sci U S A. 1998;95(1):322-327.
doi pubmed - Deep S, Sagar SC, Chaube R, Tapadia MG. The hypothalamus. In: Shackelford, T. (eds). Encyclopedia of religious psychology and behavior. Springer, Cham. 2024.
doi - Xia L, Liu HY, Wang BY, Lin HN, Wang MC, Ren JX. A review of physiological functions of orexin: From instinctive responses to subjective cognition. Medicine (Baltimore). 2023;102(26):e34206.
doi pubmed - Hung C, Yamanaka A. The role of orexin neuron activity in sleep/wakefulness regulation. Peptides. 2023;165:171007.
doi pubmed - Jacobson LH, Hoyer D, de Lecea L. Hypocretins (orexins): The ultimate translational neuropeptides. J Intern Med. 2022;291(5):533-556.
doi pubmed - Ten-Blanco M, Flores A, Cristino L, Pereda-Perez I, Berrendero F. Targeting the orexin/hypocretin system for the treatment of neuropsychiatric and neurodegenerative diseases: From animal to clinical studies. Front Neuroendocrinol. 2023;69:101066.
doi pubmed - Vaseghi S, Zarrabian S, Haghparast A. Reviewing the role of the orexinergic system and stressors in modulating mood and reward-related behaviors. Neurosci Biobehav Rev. 2022;133:104516.
doi pubmed - Toor B, Ray LB, Pozzobon A, Fogel SM. Sleep, orexin and cognition. Front Neurol Neurosci. 2021;45:38-51.
doi pubmed - Soya S, Sakurai T. Evolution of orexin neuropeptide system: structure and function. Front Neurosci. 2020;14:691.
doi pubmed - Pizza F, Barateau L, Dauvilliers Y, Plazzi G. The orexin story, sleep and sleep disturbances. J Sleep Res. 2022;31(4):e13665.
doi pubmed - Crowley SJ, Wolfson AR, Tarokh L, Carskadon MA. An update on adolescent sleep: New evidence informing the perfect storm model. J Adolesc. 2018;67:55-65.
doi pubmed - Berridge CW, Schmeichel BE, Espana RA. Noradrenergic modulation of wakefulness/arousal. Sleep Med Rev. 2012;16(2):187-197.
doi pubmed - Weyandt LL, Clarkin CM, Holding EZ, et al. Neuroplasticity in children and adolescents in response to treatment intervention: a systematic review of the literature. Clinical and Translational Neuroscience. 2020;4(2).
doi - Diamond A, Ling DS. Review of the evidence on, and fundamental questions about, efforts to improve executive functions, including working memory. Cognitive and working memory training: Perspectives from psychology, neuroscience, and human development. Oxford University Press. 2020. p. 143-431.
- Fletcher JM, Lyon GR, Fuchs LS, Barnes MA. Learning disabilities: From identification to intervention. Guilford Publications. 2018.
- Shankar A, Williams CT. The darkness and the light: diurnal rodent models for seasonal affective disorder. Dis Model Mech. 2021;14(1):dmm047217.
doi pubmed - Alexandre C, Andermann ML, Scammell TE. Control of arousal by the orexin neurons. Curr Opin Neurobiol. 2013;23(5):752-759.
doi pubmed - Jones BE. Arousal and sleep circuits. Neuropsychopharmacology. 2020;45(1):6-20.
doi pubmed - Mahoney CE, Cogswell A, Koralnik IJ, Scammell TE. The neurobiological basis of narcolepsy. Nat Rev Neurosci. 2019;20(2):83-93.
doi pubmed - Villano I, Messina A, Valenzano A, Moscatelli F, Esposito T, Monda V, Esposito M, et al. Basal forebrain cholinergic system and orexin neurons: effects on attention. Front Behav Neurosci. 2017;11:10.
doi pubmed - Messore F, Narayanan Therpurakal R, Dufour JP, Hoerder-Suabedissen A, Guidi L, Korrell K, Mueller M, et al. An orexin-sensitive subpopulation of layer 6 neurons regulates cortical excitability and anxiety behaviour. Transl Psychiatry. 2025;15(1):147.
doi pubmed - Moscatelli F, Monda A, Messina A, Monda M, Monda V, Villano I, De Maria A, et al. Evaluation of orexin-A salivary levels and its correlation with attention after non-invasive brain stimulation in female volleyball players. Sports Med Open. 2024;10(1):32.
doi pubmed - Ebrahimi MS, Ahmadpour M, Modaberi S, Fayazmilani R. Orexin-A and BDNF in the hippocampus of middle-aged rats: beneficial effects of integrating voluntary physical activity with intermittent fasting. Physiol Behav. 2025;299:115009.
doi pubmed - Wang Y, Fu S, Mao J, Cui K, Jiang H. Hypocretin: a promising target for the regulation of homeostasis. Front Neurosci. 2025;19:1638270.
doi pubmed - Runyon K, Bui T, Mazanek S, Hartle A, Marschalko K, Howe WM. Distinct cholinergic circuits underlie discrete effects of reward on attention. Front Mol Neurosci. 2024;17:1429316.
doi pubmed - Zajo KN, Fadel JR, Burk JA. Orexin A-induced enhancement of attentional processing in rats: role of basal forebrain neurons. Psychopharmacology (Berl). 2016;233(4):639-647.
doi pubmed - Qin H, Fu L, Jian T, Jin W, Liang M, Li J, Chen Q, et al. REM sleep-active hypothalamic neurons may contribute to hippocampal social-memory consolidation. Neuron. 2022;110(23):4000-4014.e4006.
doi pubmed - Li P, Zhu J, Zhang H, Ma G, Li X, Ding Y, Hou X, et al. The role of orexin and MCH neurons in the hypothalamus in sleep-wake regulation and learning-forgetting balance. Front Neurosci. 2025;19:1590556.
doi pubmed - Fan Y, Jiang E, Gao H, Bigalke J, Chen B, Yu C, Chen Q, et al. Activation of orexin system stimulates CaMKII expression. Front Physiol. 2021;12:698185.
doi pubmed - Feng Z, Li Q, He Z, Yu B, Mi T, Niu J, He Y, et al. Hypothalamic orexin projections to the hippocampal CA1 region alleviate cognitive and synaptic plasticity impairments induced by blue light exposure. CNS Neurosci Ther. 2025;31(8):e70551.
doi pubmed - Abounoori M, Maddah MM, Ardeshiri MR. Orexin neuropeptides modulate the hippocampal-dependent memory through basolateral amygdala interconnections. Cereb Circ Cogn Behav. 2022;3:100035.
doi pubmed - Roesler R, Parent MB, LaLumiere RT, McIntyre CK. Amygdala-hippocampal interactions in synaptic plasticity and memory formation. Neurobiol Learn Mem. 2021;184:107490.
doi pubmed - Krauth N. Tug-of-War: orexin and dynorphin effects on reward processing circuits. J Neurosci. 2025;45(4):e1979242024.
doi pubmed - Kumar D, Koyanagi I, Carrier-Ruiz A, Vergara P, Srinivasan S, Sugaya Y, Kasuya M, et al. Sparse activity of hippocampal adult-born neurons during REM sleep is necessary for memory consolidation. Neuron. 2020;107(3):552-565.e510.
doi pubmed - Delorme J, Wang L, Kuhn FR, Kodoth V, Ma J, Martinez JD, Raven F, et al. Sleep loss drives acetylcholine- and somatostatin interneuron-mediated gating of hippocampal activity to inhibit memory consolidation. Proc Natl Acad Sci U S A. 2021;118(32):e2019318118.
doi pubmed - Feng H, Wen SY, Qiao QC, Pang YJ, Wang SY, Li HY, Cai J, et al. Orexin signaling modulates synchronized excitation in the sublaterodorsal tegmental nucleus to stabilize REM sleep. Nat Commun. 2020;11(1):3661.
doi pubmed - Lendner JD, Niethard N, Mander BA, van Schalkwijk FJ, Schuh-Hofer S, Schmidt H, Knight RT, et al. Human REM sleep recalibrates neural activity in support of memory formation. Sci Adv. 2023;9(34):eadj1895.
doi pubmed - Erichsen JM, Calva CB, Reagan LP, Fadel JR. Intranasal insulin and orexins to treat age-related cognitive decline. Physiol Behav. 2021;234:113370.
doi pubmed - Calva CB, Fayyaz H, Fadel JR. Increased acetylcholine and glutamate efflux in the prefrontal cortex following intranasal orexin-A (hypocretin-1). J Neurochem. 2018;145(3):232-244.
doi pubmed - Piantadosi PT, Holmes A, Roberts BM, Bailey AM. Orexin receptor activity in the basal forebrain alters performance on an olfactory discrimination task. Brain Res. 2015;1594:215-222.
doi pubmed - Aracri P, Banfi D, Pasini ME, Amadeo A, Becchetti A. Hypocretin (orexin) regulates glutamate input to fast-spiking interneurons in layer V of the Fr2 region of the murine prefrontal cortex. Cereb Cortex. 2015;25(5):1330-1347.
doi pubmed - Cole S, Keefer SE, Anderson LC, Petrovich GD. Medial prefrontal cortex neural plasticity, orexin receptor 1 signaling, and connectivity with the lateral hypothalamus are necessary in cue-potentiated feeding. J Neurosci. 2020;40(8):1744-1755.
doi pubmed - Duske J, D'Souza N, Mayer D, Dieterich DC, Fendt M. Orexinergic modulation of chronic jet lag-induced deficits in mouse cognitive flexibility. Neuropsychopharmacology. 2025;50(5):762-771.
doi pubmed - Xiao X, Yeghiazaryan G, Eggersmann F, Cremer AL, Backes H, Kloppenburg P, Hausen AC. Deficiency of orexin receptor type 1 in dopaminergic neurons increases novelty-induced locomotion and exploration. Elife. 2025;12.
doi pubmed - Gong WK, Li X, Wang L, Yang Q, Tiran-Cappello A, Liang Z, Samsom J, et al. Prefrontal FGF1 signaling is required for accumbal deep brain stimulation treatment of addiction. Adv Sci (Weinh). 2025;12(16):e2413370.
doi pubmed - Tan MK, Chia KH, Liu AL, Singh H. From synapse to support: How neuroscience can guide educational therapists & counselors in providing better mental wellness & behaviour therapy. ISRG Journal of Clinical Medicine and Medical Research. 2025;2(2):39-59.
doi - Pavlov IP. Conditioned reflexes: An investigation of the physiological activity of the cerebral cortex (Transl. & edited by Anrep GV). Oxford, UK: Oxford University Press. 1927.
- Pavlov PI. Conditioned reflexes: An investigation of the physiological activity of the cerebral cortex. Ann Neurosci. 2010;17(3):136-141.
doi pubmed - Alsoqih NS, Elmorsy EA, Abu-Khudir R, Saber S, Keshk N, Elnaghy F, Hamad RS, et al. A Systems Biology Perspective on Childhood ADHD: Neurochemical Dysregulation, Brain-Behavior Interactions, and Emerging Therapeutics. FASEB J. 2025;39(17):e70981.
doi pubmed - Katzman MA, Katzman MP. Neurobiology of the orexin system and its potential role in the regulation of hedonic tone. Brain Sci. 2022;12(2):150.
doi pubmed - Berry AS, Harrison TM. New perspectives on the basal forebrain cholinergic system in Alzheimer's disease. Neurosci Biobehav Rev. 2023;150:105192.
doi pubmed - Leman TY, Barden S, Swisher VS, Joyce DS, Kaplan KA, Zeitzer JM, Loo SK, et al. Sleep insufficiency and bedtime irregularity in children with ADHD: A population-based analysis. Sleep Med. 2024;121:117-126.
doi pubmed - Peters SU, Shelton AR, Malow BA, Neul JL. A clinical-translational review of sleep problems in neurodevelopmental disabilities. J Neurodev Disord. 2024;16(1):41.
doi pubmed - Chua ACK, Chia NKH. A brief review of educational therapy & its current role: Part 1. Unlimited Human. 2023;4-5.
doi - World Health Organization. International classification of diseases, clinical modifications-ninth revision, volume 3 (ICD-9-CM Ver.3). Geneva, Switzerland: The Author. 1986.
- Pierangelo R, Giuliani G. The Educator’s Diagnostic Manual (EDM) of disabilities and disorders. San Francisco, CA: Jossey-Bass. 2007. Available from: https://www.amazon.sg/dp/0787978124?ref_=mr_referred_us_sg_sg.
- Saday Duman NS, Oner O, Aysev AS. The effect of educational therapy on self-esteem and problem behaviors in children with specific learning disability. Alpha Psychiatry. 2017;18(1):85-92.
doi - Chia KH. A Short Review of Psychoeducational Therapy (PsyEdTx). The Asian Educational Therapist. 2024;2(1):3–11.
doi - Tan MK, Chia KH. Brain network dysfunction and rumination in psychiatric disorders: Integrative insights into educational therapy as a targeted intervention. Asian Journal of Research & Reports in Neurology. 2025;8(1):203-222.
doi - Colizzi M, Lasalvia A, Ruggeri M. Prevention and early intervention in youth mental health: is it time for a multidisciplinary and trans-diagnostic model for care? Int J Ment Health Syst. 2020;14:23.
doi pubmed - Evans SW, Owens JS, Bunford N. Evidence-based psychosocial treatments for children and adolescents with attention-deficit/hyperactivity disorder. J Clin Child Adolesc Psychol. 2014;43(4):527-551.
doi pubmed - Petti T, Gupta M, Fradkin Y, Gupta N. Management of sleep disorders in autism spectrum disorder with co-occurring attention-deficit hyperactivity disorder: update for clinicians. BJPsych Open. 2023;10(1):e11.
doi pubmed - WHO, World Health Organization. International classification of diseases (11th ed.). 2021. https://icd.who.int/en/.
- Hale L, Guan S. Screen time and sleep among school-aged children and adolescents: a systematic literature review. Sleep Med Rev. 2015;21:50-58.
doi pubmed - Rasch B, Born J. About sleep's role in memory. Physiol Rev. 2013;93(2):681-766.
doi pubmed - Roebers CM. Executive function and metacognition: Towards a unifying framework of cognitive self-regulation. Developmental review. 2017;45:31-51.
doi - Ryan RM, Deci EL. Intrinsic and extrinsic motivation from a self-determination theory perspective: Definitions, theory, practices, and future directions. Contemporary educational psychology. 2020;61:101860.
doi - Deci EL, Olafsen AH, Ryan RM. Self-determination theory in work organizations: The state of a science. Annual review of organizational psychology and organizational behavior. 2017;4:19-43.
doi - Dweck CS. From needs to goals and representations: Foundations for a unified theory of motivation, personality, and development. Psychol Rev. 2017;124(6):689-719.
doi pubmed - Caemmerer JM, Keith TZ, Reynolds MR. Beyond individual intelligence tests: application of Cattell-Horn-Carroll theory. Intelligence. 2020;79:101433.
doi - Durlak JA, Weissberg RP, Dymnicki AB, Taylor RD, Schellinger KB. The impact of enhancing students' social and emotional learning: a meta-analysis of school-based universal interventions. Child Dev. 2011;82(1):405-432.
doi pubmed - What Works Clearinghouse. What Works Clearinghouse standards handbook, version 4.1. U.S. Department of Education, Institute of Education Sciences, National Center for Education Evaluation and Regional Assistance. 2020. https://ies.ed.gov/ncee/wwc/Handbooks.
This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (CC BY 4.0), which permits unrestricted use, distribution, and reproduction in any medium, including commercial use, provided the original work is properly cited.
Journal of Neurology Research is published by Elmer Press Inc.