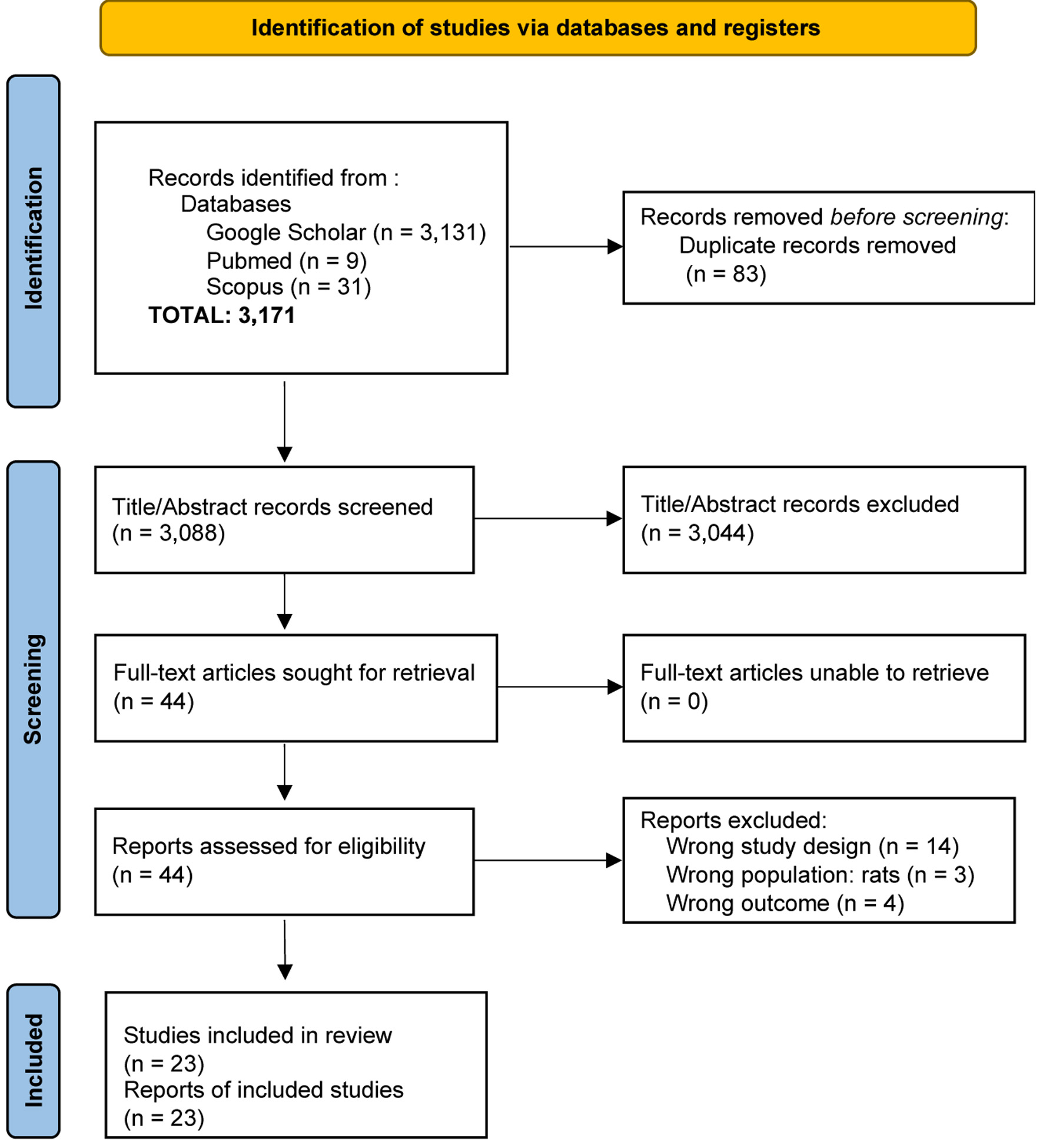
↓ ↓ Figure 1. Flow
diagram of the study selection process for articles included in the systematic review and
meta-analysis.
| Journal of Neurology Research, ISSN 1923-2845 print, 1923-2853 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Neurol Res and Elmer Press Inc |
| Journal website https://jnr.elmerpub.com |
Original Article
Volume 16, Number 1, March 2026, pages 30-39
The Role of Calcitriol (Vitamin D) as Neuroprotection and Prognostic Biomarker in Patients With Ischemic and Hemorrhagic Stroke
Figures

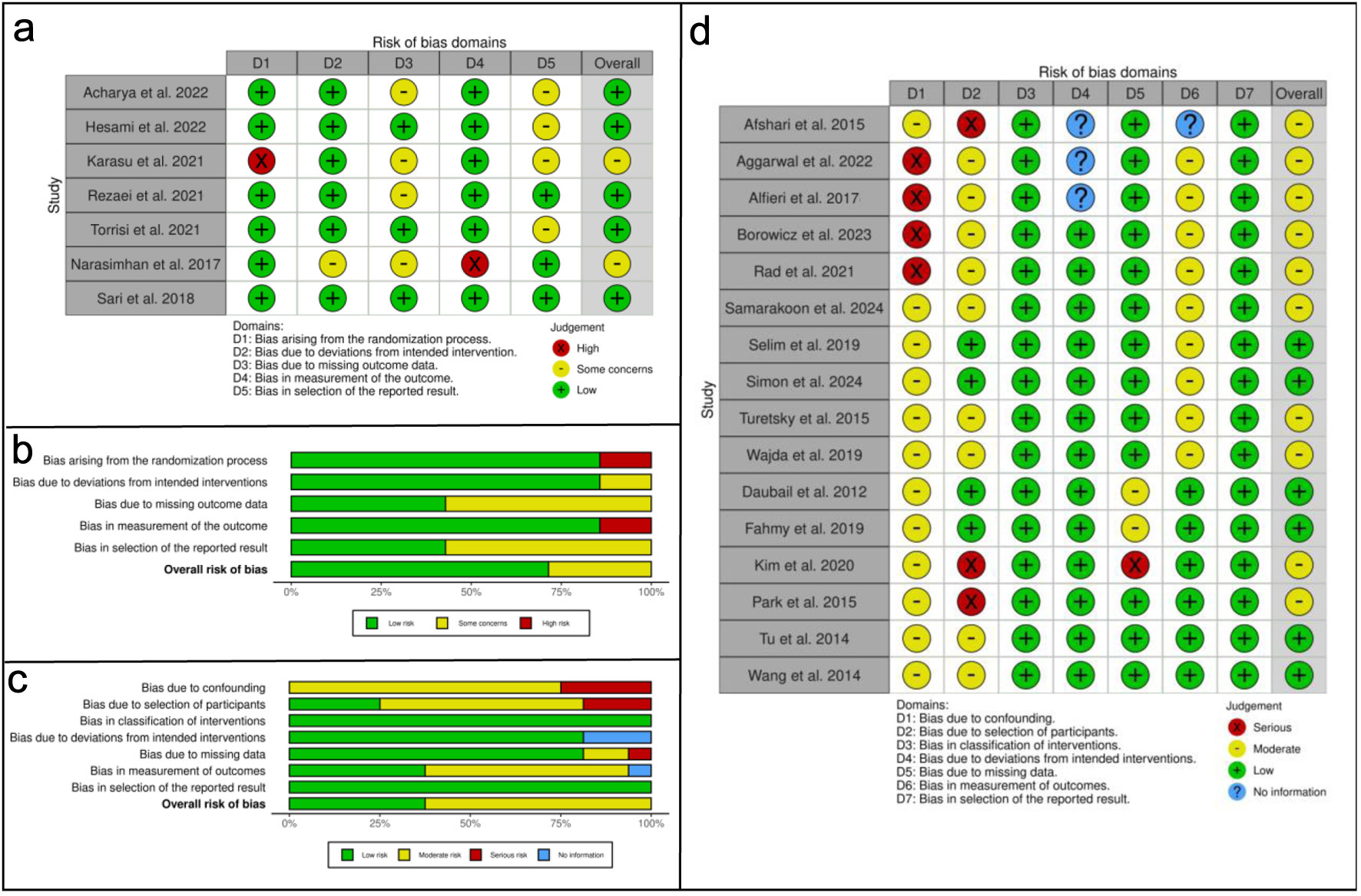
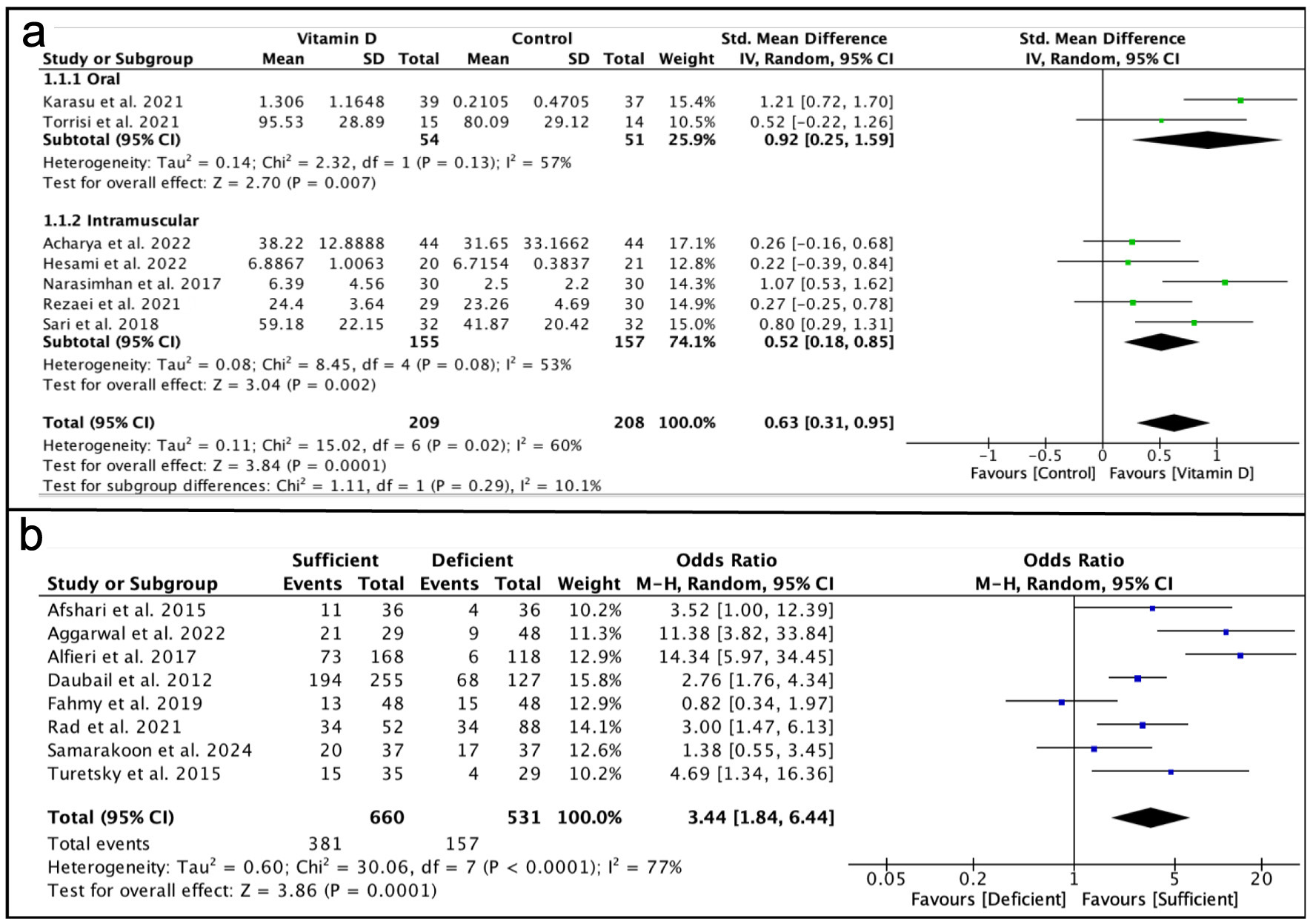
Tables
| No. | Author | Location | Sample size | Study design | Diagnosis | Intervention regimen | Control regimen | Outcome measurement tools |
|---|---|---|---|---|---|---|---|---|
| 1. | Acharya et al, 2022 | India | 325 | Randomized controlled trial | Ischemic stroke and hemorrhagic stroke | Single intramuscular dose of 600,000 International Units (IU) of vitamin D | Vitamin D not administered | Scandinavian Stroke Scale (SSS) score |
| 2. | Hesami et al, 2022 | Iran | 570 | Randomized controlled trial | Ischemic stroke | Single intramuscular dose of 600,000 IU of vitamin D3 | Vitamin D not administered | The Neuron-Specific Enolase (NSE) level, the National Institutes of Health Stroke Scale (NIHSS), and Barthel Index (BI) |
| 3. | Karasu et al, 2021 | Turkey | 76 | Retrospective | Ischemic stroke and hemorrhagic stroke | Weekly oral dose of 50,000 IU for 4–12 weeks, with a total dose ranging from 200,000 to 600,000 IU | Vitamin D not administered | Brunnstrom Recovery Stage and Functional Ambulation Classification (FAC) score |
| 4. | Rezaei et al, 2021 | Iran | 60 | Randomized controlled trial | Ischemic stroke | Single intramuscular dose of 300,000 IU of vitamin D | Vitamin D not administered | NIHSS, modified Rankin Scale (mRS), and the Mini-Mental State Examination (MMSE) |
| 5. | Torrisi et al, 2021 | Italy | 40 | Randomized controlled trial | Ischemic stroke and hemorrhagic stroke | Daily oral dose of 2,000 IU/day of vitamin D3 for 12 weeks | Vitamin D not administered | Montgomery-Aasberg Depression Rating Scale (MADRS) and Functional Independent Measures (FIM) |
| 6. | Narasimhan et al, 2017 | India | 60 | Randomized controlled trial | Ischemic stroke | Single intramuscular dose of 600,000 IU of vitamin D3 | Vitamin D not administered | SSS score |
| 7. | Sari et al, 2018 | Turkey | 64 | Randomized controlled trial | Ischemic stroke | Single intramuscular dose of 300,000 IU of vitamin D | Vitamin D not administered | Brunnstrom Recovery Staging (BRS), Functional Ambulation Scale (FAS), Modified Barthel Index (MBI) scores, and Berg Balance Scale (BBS) |
| No. | Author | Location | Sample size | Study design | Diagnosis | Assessment of stroke severity | Assessment of serum vitamin D levels |
|---|---|---|---|---|---|---|---|
| 8. | Afshari et al, 2015 | Iran | 72 | Case-control | Ischemic stroke | Brain computed tomography | Enzyme-linked immunosorbent assay (ELISA) |
| 9. | Aggarwal et al, 2022 | India | 200 | One-year prospective observational | Ischemic stroke | The National Institutes of Health Stroke scale (NIHSS) and modified Rankin scale (mRS) | Electrochemiluminescence (ECL) method |
| 10. | Alfieri et al, 2017 | Brazil | 286 | Case-control | Ischemic stroke | mRS | Chemiluminescent microparticle immunoassay (CMIA) |
| 11. | Borowicz et al, 2023 | Poland | 80 | Randomized controlled trial | Ischemic stroke | NIHSS, mRS, and Barthel index (BI) | CMIA |
| 12. | Rad et al, 2021 | Iran | 140 | Bi-center cross-sectional | Ischemic stroke and hemorrhagic Stroke | NIHSS and mRS | ECL |
| 13. | Samarakoon et al, 2024 | Sri Lanka | 60 | Prospective case-control | Ischemic stroke | NIHSS and mRS | ELISA |
| 14. | Selim et al, 2019 | Egypt | 138 | Case-control | Ischemic stroke and hemorrhagic stroke | NIHSS and mRS | ELISA |
| 15. | Simon et al, 2024 | India | 86 | Prospective observational | Ischemic stroke | NIHSS | Chemiluminescence immunoassay (CLIA) |
| 16. | Turetsky et al, 2015 | United States | 96 | Retrospectively analyze prospective | Ischemic stroke | NIHSS and mRS | CLIA |
| 17. | Wajda et al, 2019 | Poland | 240 | Retrospective cohort study | Ischemic stroke | NIHSS and mRS | ECL |
| 18. | Daubail et al, 2012 | France | 386 | Observational cohort study | Ischemic stroke and hemorrhagic stroke | NIHSS and mRS | High pressure liquid chromatography coupled with UV detection |
| 19. | Fahmy et al, 2019 | Egypt | 96 | Case-control | Ischemic stroke | NIHSS and mRS | ELISA |
| 20. | Kim et al, 2020 | South Korea | 328 | Single-center retrospective study | Ischemic stroke | NIHSS | Radioimmunoassay kit |
| 21. | Park et al, 2015 | South Korea | 818 | Observational cohort retrospective | Ischemic stroke | NIHSS and mRS | Liquid chromatography tandem mass spectrometry |
| 22. | Tu et al, 2014 | China | 364 | Prospective cohort | Ischemic stroke | NIHSS and mRS | Competitive chemiluminescent immunoassay on a calibrated Elecsys 2010 |
| 23. | Wang et al, 2014 | New York | 326 | Prospective cohort | Ischemic stroke | NIHSS and mRS | The E601 modular (Roche Diagnostics, Mannheim, Germany) with a calibration range from 3 to 70 ng/mL |