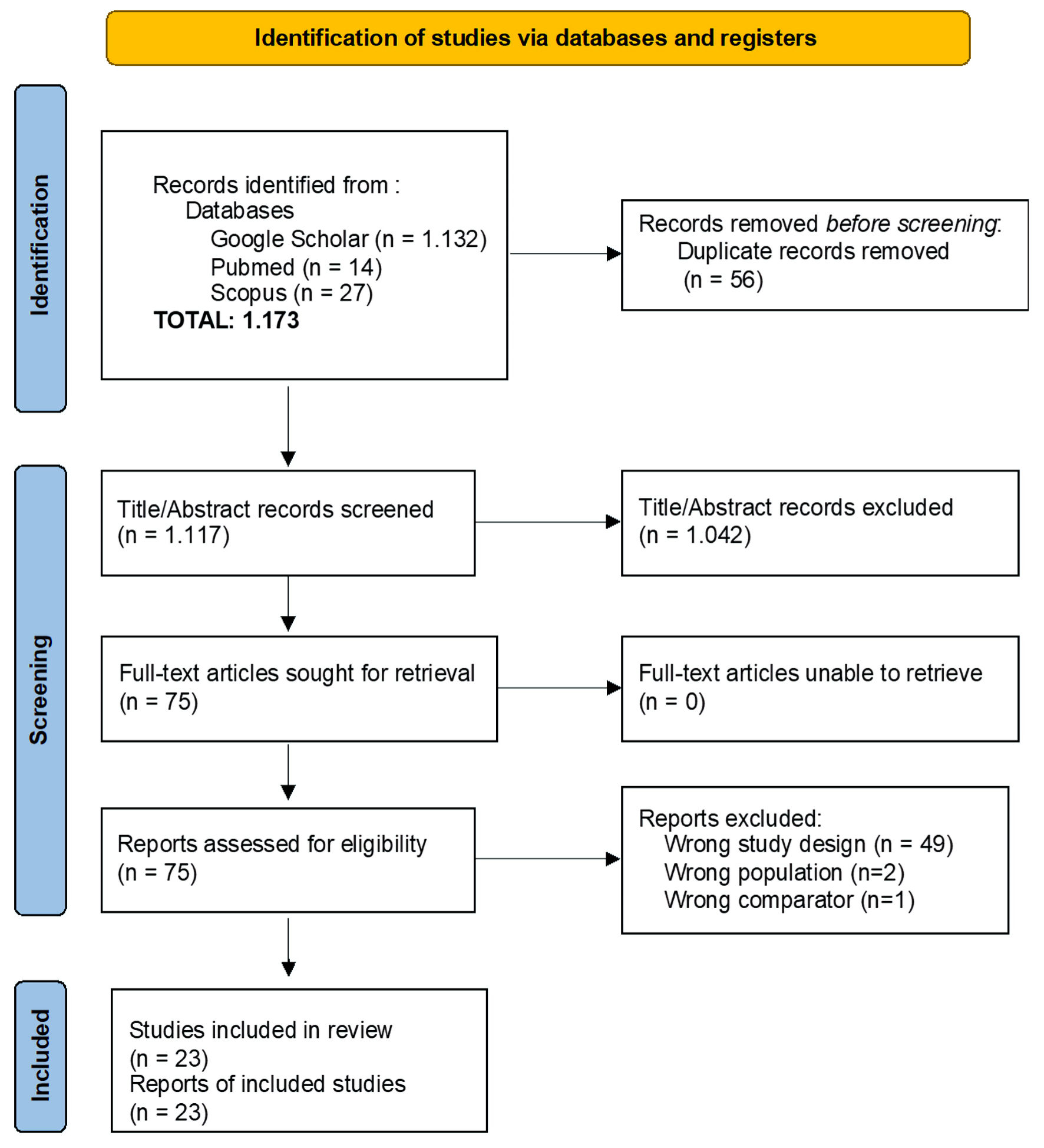
↓ Figure 1. PRISMA flow diagram of study selection process for the systematic review and meta-analysis. PRISMA: Preferred Reporting Items for Systematic Reviews and Meta-Analyses.
| Journal of Neurology Research, ISSN 1923-2845 print, 1923-2853 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Neurol Res and Elmer Press Inc |
| Journal website https://jnr.elmerpub.com |
Original Article
Volume 16, Number 2, June 2026, pages 99-114
Can an Antidepressant Improve Motor Recovery After Stroke? A Meta-Analysis of Adjuvant Fluoxetine Reveals Both New Hope and Safety Concerns
Figures

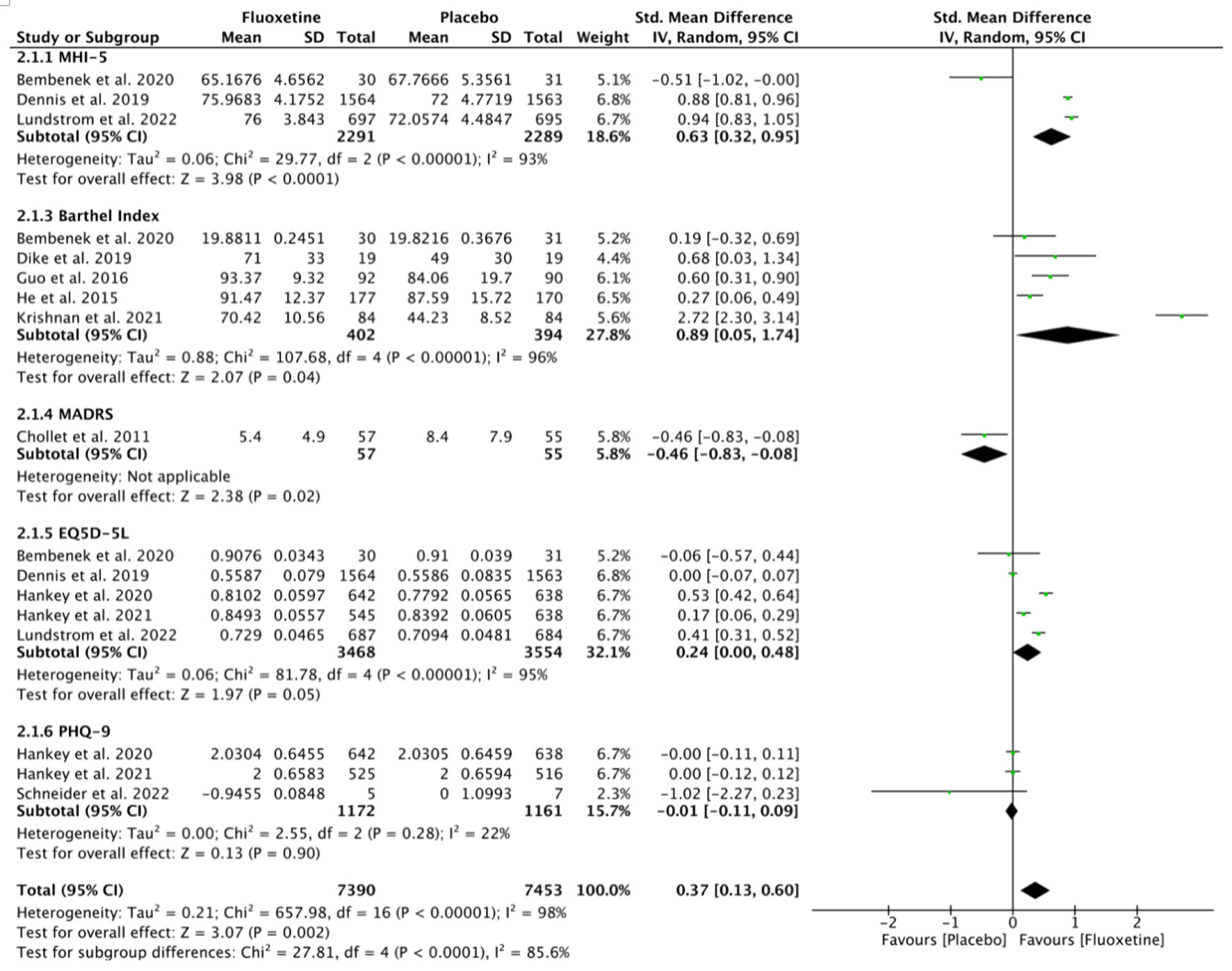
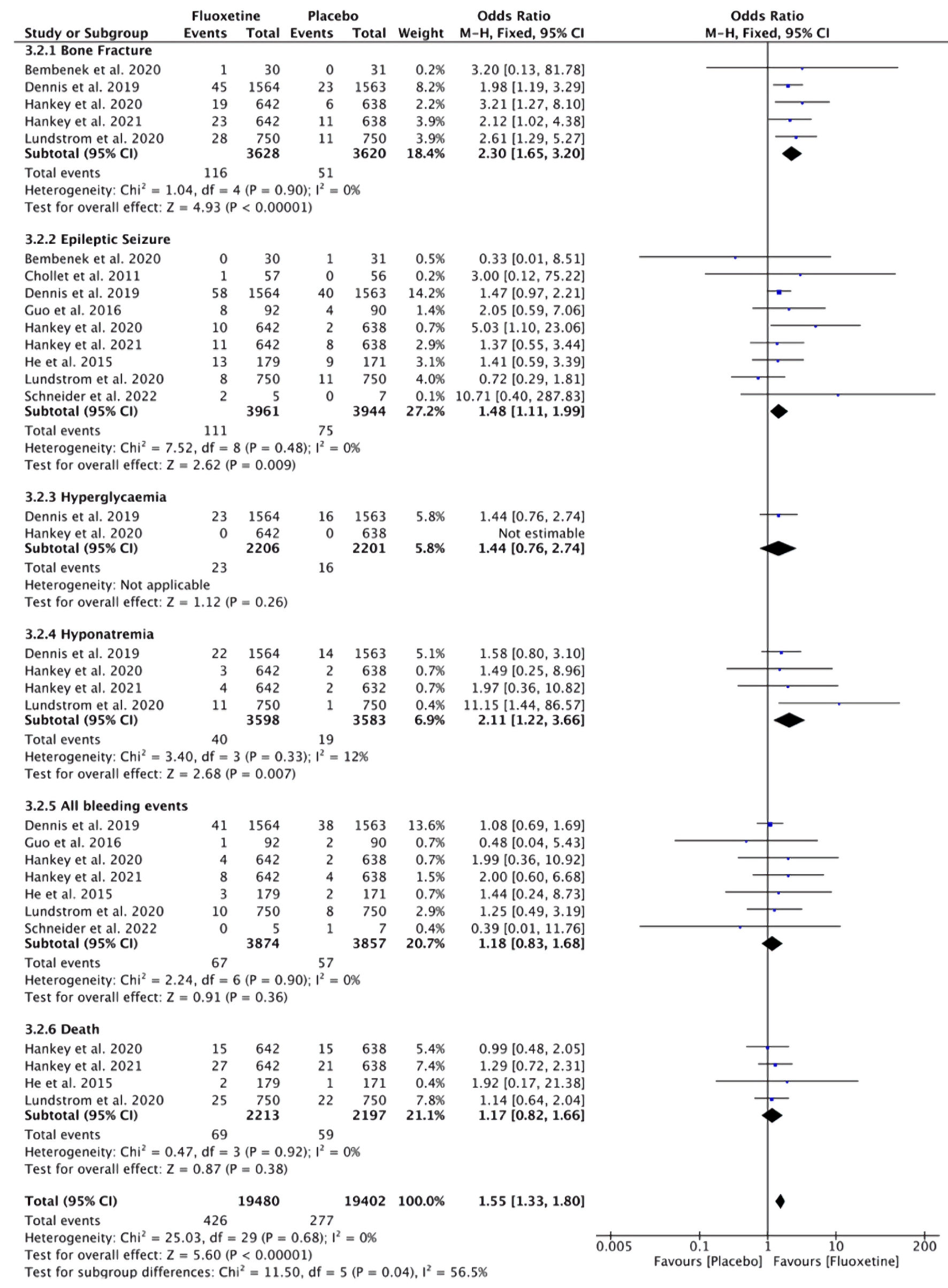
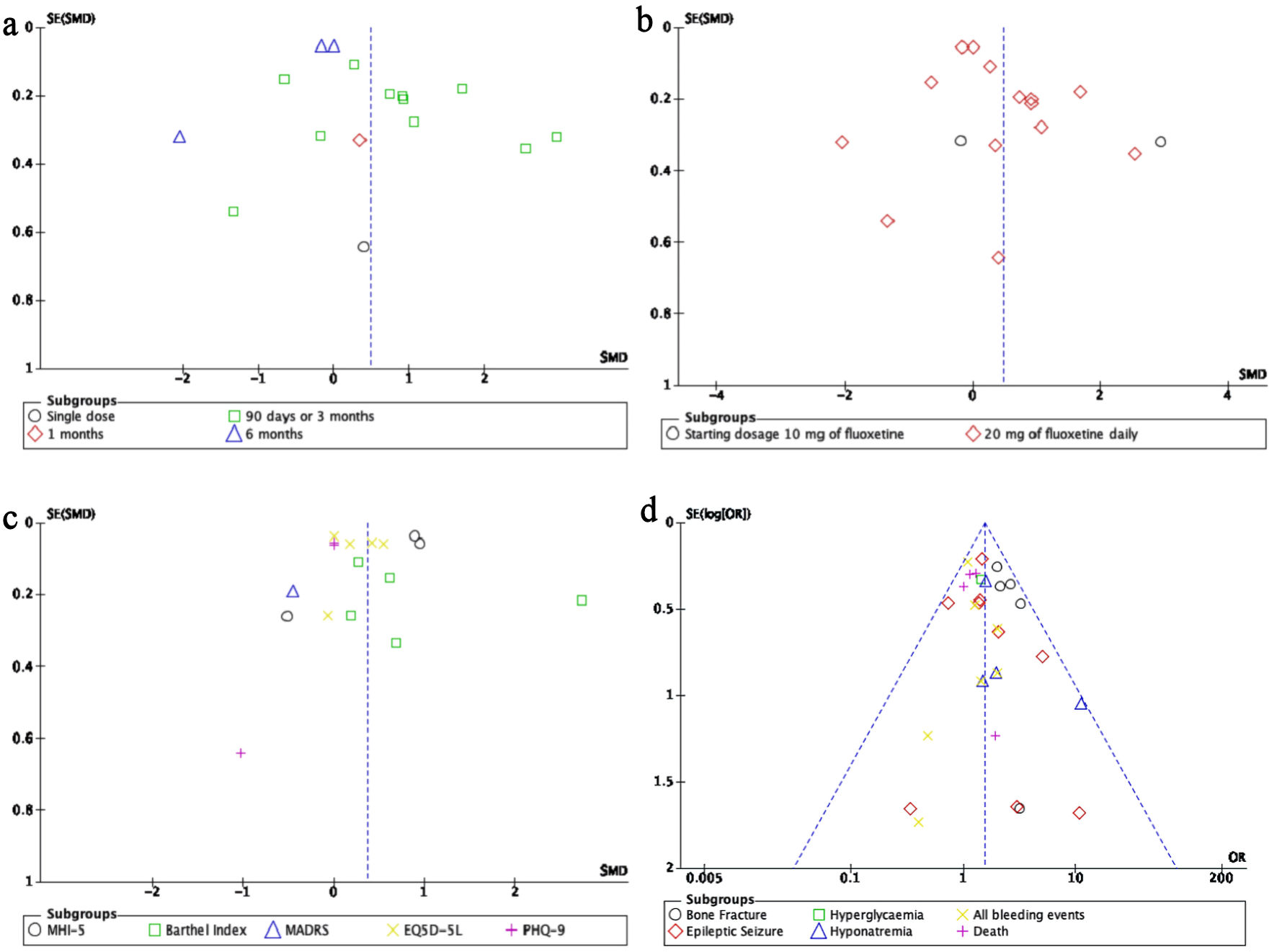
Tables
| Study | Location | Sample size | Diagnosis | Study design | Outcomes and measurement |
|---|---|---|---|---|---|
| BI: Barthel Index; BDI: Beck Depression Inventory; EQ-5D-5L: EuroQol 5 Dimensions 5 Levels; FIM: Functional Independence Measure; FMA: Fugl–Meyer Assessment; FMMS: Fugl–Meyer Motor Scale; JTHFT: Jebsen–Taylor Hand Function Test; MADRS: Montgomery–Asberg Depression Rating Scale; MAS: Modified Ashworth Scale; MHI-5: Mental Health Inventory-5; MMSE: Mini-Mental State Examination; MoCA: Montreal Cognitive Assessment; MRC: Medical Research Council; mRS: modified Rankin Scale; NHPT: Nine-Hole Peg Test; NIHSS: National Institutes of Health Stroke Scale; PHQ-9: Patient Health Questionnaire-9; PMD: Perimetric Mean Deviation; rTMS: Repetitive Transcranial Magnetic Stimulation; SF-36: 36-Item Short Form Health Survey; SGPALS: Saltin–Grimby Physical Activity Level Scale; SIS: Stroke Impact Scale; TICSm: Telephone Interview for Cognitive Status–modified; VAS: Visual Analog Scale; VFQ-25: 25-Item Visual Function Questionnaire. | |||||
| Asadollahi et al, 2018 [24] | Iran | 90 | Acute ischemic stroke | Double-blind randomized controlled trial | FMMS (30, 60, 90 days); adverse events |
| Babul et al, 2017 [15] | Bangladesh | 128 | Acute ischemic stroke | Randomized placebo controlled trial | RMI (30, 90 days); motor recovery domains |
| Bembenek et al, 2020 [8] | Poland | 61 | Acute stroke (ischemic or hemorrhagic stroke) | Randomized, double-blind, placebo-controlled study | mRS (6 months); SIS (6 months); NIHSS (6, 12 months); MRC and Brunnstrom scales (arm/hand deficit); MHI-5 (mood); VAS (recovery); EQ-5D-5L (HRQoL); adverse events |
| Berends et al, 2009 [25] | Netherlands | 10 | Chronic ischemic stroke | A crossover, placebo-controlled (double-blind), randomized trial | Grip strength and motor function (dynamometer) |
| Chollet et al, 2011 [26] | France | 118 | Acute ischemic stroke | Double-blind, placebo-controlled trial | FMMS (baseline–90 days); NIHSS, mRS, MADRS (baseline, 30, 90 days); adverse events |
| Dehghani et al, 2024 [16] | Iran | 90 | Acute ischemic stroke | Double-blind interventional study with placebo | BDI-II (depression); FMMS (90 days) |
| Dennis et al, 2019 [27] | UK | 3,127 | Acute stroke (ischemic or hemorrhagic stroke) | Pragmatic, multicenter, parallel group, double-blind, randomized, placebo-controlled trial | mRS (6 months); survival (6, 12 months); SIS (health status); Likert scale (social function); VAS (recovery); MHI-5 (mood); SF-36 vitality (fatigue); EQ-5D-5L (HRQoL); adverse events |
| Dike et al, 2019 [17] | Nigeria | 60 | Acute supratentorial ischemic stroke | Single-blind randomized controlled trial | FMMS change (entry–exit); BI and mRS (entry–exit); adverse effects; adverse effects of fluoxetine |
| Gong et al, 2020 [28] | China | 254 | Acute ischemic stroke | Double-blind and randomized controlled trial | FMMS and mRS (30, 90 days); adverse events |
| Guo et al, 2016 [18] | China | 267 | Acute ischemic stroke | Single-blind randomized controlled trial | NIHSS (15, 90, 180 days); BI (90, 180 days) |
| Hankey et al, 2020 [22] | Australia | 1,280 | Acute stroke (ischemic or hemorrhagic stroke) | Randomized, parallel group, double-blind, placebo-controlled trial | mRS (6 months); survival; PHQ-9 (depression); TICSm (cognition); SIS (health status); SF-36 vitality (fatigue); EQ-5D-5L (HRQoL); adverse events |
| He et al, 2016 [19] | China | 350 | Acute ischemic stroke | Single-blind randomized controlled trial | NIHSS (15, 90, 180 days); BI (90, 180 days) |
| Krishnan et al, 2021 [20] | India | 168 | Acute or subacute stroke (ischemic or hemorrhagic stroke) | Randomized, placebo-controlled, single-blind trial | BI; NHPT; finger tapping (baseline, 45, 90 days) |
| Lehto et al, 2020 [29] | Sweden | 1,373 | Acute stroke (ischemic or hemorrhagic stroke) | Randomized controlled trial | Physical activity (SGPALS, 6 months) |
| Lundstrom et al, 2020 [23] | Sweden | 1,500 | Acute stroke (ischemic or hemorrhagic stroke) | Investigator-led, multicenter, randomized, placebo-controlled, double-blind, parallel group trial | mRS (6 months); SIS v3; NIHSS; MoCA; depression diagnosis; medication adherence |
| Lundstrom et al, 2022 [30] | Sweden | 1,500 | Acute stroke (ischemic or hemorrhagic stroke) | Randomized, double-blind, placebo-controlled clinical trial | mRS; SIS v3; MHI-5 (mood); SF-36 vitality (fatigue); EQ-5D-5L (HRQoL) |
| Marquez et al, 2020 [31] | Mexico | 30 | Acute intracerebral hemorrhage | Double blind, placebo controlled, multicenter randomized trial | FMMS change (0–90 days); NIHSS; BI; mRS |
| Mikami et al, 2011 [32] | Japan | 83 | Chronic stroke (ischemic or hemorrhagic stroke) | Double-blind randomized study | DSM-IV depression diagnosis; HDRS-17; mRS; FIM |
| Pinto et al, 2019 [33] | Brazil | 27 | Chronic ischemic stroke | Double-blinded randomized clinical trial | JTHFT; FMA-UE (motor); MAS; BDI; MMSE; rTMS side effects |
| Saadat et al, 2025 [21] | Iran | 60 | Acute ischemic stroke | Placebo-controlled, single-blind clinical trial | NIHSS (motor impairment) |
| Schneider et al, 2023 [34] | New York | 12 | Ischemic stroke | Randomized, placebo-controlled, double-blind, pilot clinical trial | PMD improvement; visual field recovery; VFQ-25; mRS (3 months) |
| Shah et al, 2016 [35] | India | 84 | Acute hemorrhagic stroke | Double-blind, placebo-controlled, randomized controlled clinical study | FMMS change (0–90 days); mRS; drug side effects |
| Tay et al, 2023 [36] | Sweden | 1,369 | Acute stroke (ischemic or hemorrhagic stroke) | Randomized, double-blind, placebo-controlled clinical trial | NIHSS (stroke severity); MADRS (depression/apathy) |
| Study | Interventions regimens | Control regimen | Duration of treatment | Result |
|---|---|---|---|---|
| FMMS: Fugl–Meyer Motor Scale. | ||||
| Asadollahi et al, 2018 [24] | 20 mg per os (PO) of fluoxetine daily and physiotherapy | Placebo (microcrystalline cellulose) and physiotherapy | 90 days | Fluoxetine significantly improved motor recovery compared with placebo. |
| Babul et al, 2017 [15] | 20 mg PO of fluoxetine daily and physiotherapy | Placebo (details not specified) and physiotherapy | 3 months | Early fluoxetine administration with physiotherapy enhanced motor recovery after 3 months. |
| Bembenek et al, 2020 [8] | 20 mg PO of fluoxetine daily | Placebo (details not specified) | 6 months | Fluoxetine did not improve motor recovery or functional outcomes at 6 and 12 months. |
| Berends et al, 2009 [25] | 20 mg PO of fluoxetine single dose | Placebo (details not specified) | Single dose | Fluoxetine showed potential effects on post-stroke motor recovery. |
| Chollet et al, 2011 [26] | 20 mg PO of fluoxetine daily and physiotherapy | Placebo (details not specified) and physiotherapy | 90 days | Early fluoxetine combined with physiotherapy improved motor recovery at 3 months. |
| Dehghani et al, 2024 [16] | 20 mg PO of fluoxetine daily and physiotherapy | Placebo (microcrystalline cellulose) and physiotherapy | 90 days | Fluoxetine improved motor function and supported sustained recovery after stroke. |
| Dennis et al, 2019 [27] | 20 mg PO of fluoxetine daily | Placebo (details not specified) | 6 months | Fluoxetine did not improve functional outcomes but reduced depression and increased bone fracture risk. |
| Dike et al, 2019 [17] | 20 mg PO of fluoxetine daily and standard therapy | Placebo (details not specified) and standard therapy | 1 months | Fluoxetine if started in the acute phase of ischemic stroke may improve functional status. It also shows a positive trend towards enhancing motor recovery |
| Gong et al, 2020 [28] | 20 mg PO of fluoxetine daily | Placebo (no anti-depressive medication) | 90 days | Fluoxetine improved motor ability at 90 days based on FMMS scores. |
| Guo et al, 2016 [18] | 20 mg PO of fluoxetine daily | Placebo (details not specified) | 90 days | Early fluoxetine administration improved neurological functional prognosis after ischemic stroke. |
| Hankey et al, 2020 [22] | 20 mg PO of fluoxetine daily | Placebo (details not specified) | 6 months | Fluoxetine did not improve functional outcomes and increased risks of falls, fractures, and seizures. |
| He et al, 2016 [19] | 20 mg PO of fluoxetine daily and standard therapy (20 mg/day atorvastatin) | Placebo (details not specified) and standard therapy (20 mg/day atorvastatin) | 90 days | Fluoxetine treatment improved long-term neurological functional outcomes after ischemic stroke |
| Krishnan et al, 2021 [20] | 20 mg PO of fluoxetine daily | Placebo (details not specified) | 90 days | Early fluoxetine treatment was safe and may enhance motor function in patients with severe motor impairment. |
| Lehto et al, 2020 [29] | 20 mg PO of fluoxetine daily | Placebo (details not specified) | 6 months | Fluoxetine was not associated with increased physical activity levels after stroke. |
| Lundstrom et al, 2020 [23] | 20 mg PO of fluoxetine daily | Placebo (details not specified) | 6 months | Fluoxetine did not improve functional outcomes but reduced depression and increased fracture and hyponatremia risk. |
| Lundstrom et al, 2022 [30] | 20 mg PO of fluoxetine daily | Placebo (details not specified) | 6 months | Fluoxetine had no effect on functional outcomes and was associated with poorer memory and communication scores. |
| Marquez et al, 2020 [31] | 20 mg PO of fluoxetine daily and standard therapy | Placebo (details not specified) and standard therapy | 90 days | Fluoxetine combined with standard therapy improved motor recovery after intracerebral hemorrhage. |
| Mikami et al, 2011 [32] | 10 mg/day for the first 3 weeks, 20 mg/day for weeks 4–6, 30 mg/day for weeks 7–9, and 40 mg/day for the final 3 weeks of fluoxetine | Placebo (details not specified) | 3 months | Fluoxetine improved long-term recovery from post-stroke disability. |
| Pinto et al, 2019 [33] | 20 mg PO of fluoxetine daily | Placebo (details not specified) | 90 days | Fluoxetine resulted in smaller motor improvements than placebo and may hinder beneficial plasticity. |
| Saadat et al, 2025 [21] | Starting dosage of 10 mg fluoxetine orally daily, subsequently raised to a maximum dose of 20 mg daily + standard treatment (daily clopidogrel 75 mg and aspirin 80 mg) | Placebo (details not specified) + Standard treatment (daily clopidogrel 75 mg and aspirin 80 mg) | 3 months | Fluoxetine improved motor status and overall health outcomes in stroke patients. |
| Schneider et al, 2023 [34] | 20 mg PO of fluoxetine daily | Placebo (details not specified) | 90 days | Fluoxetine showed a potential trend toward improved visual recovery after stroke. |
| Shah et al, 2016 [35] | Starting dosage of 10 mg fluoxetine orally daily and later increased to 20 mg after a period of 1 week | Placebo (details not specified) | 3 months | Early fluoxetine combined with physiotherapy improved motor recovery after hemorrhagic stroke. |
| Tay et al, 2023 [36] | 20 mg PO of fluoxetine daily | Placebo (details not specified) | 6 months | Fluoxetine was ineffective in preventing post-stroke apathy despite effects on depressive symptoms. |