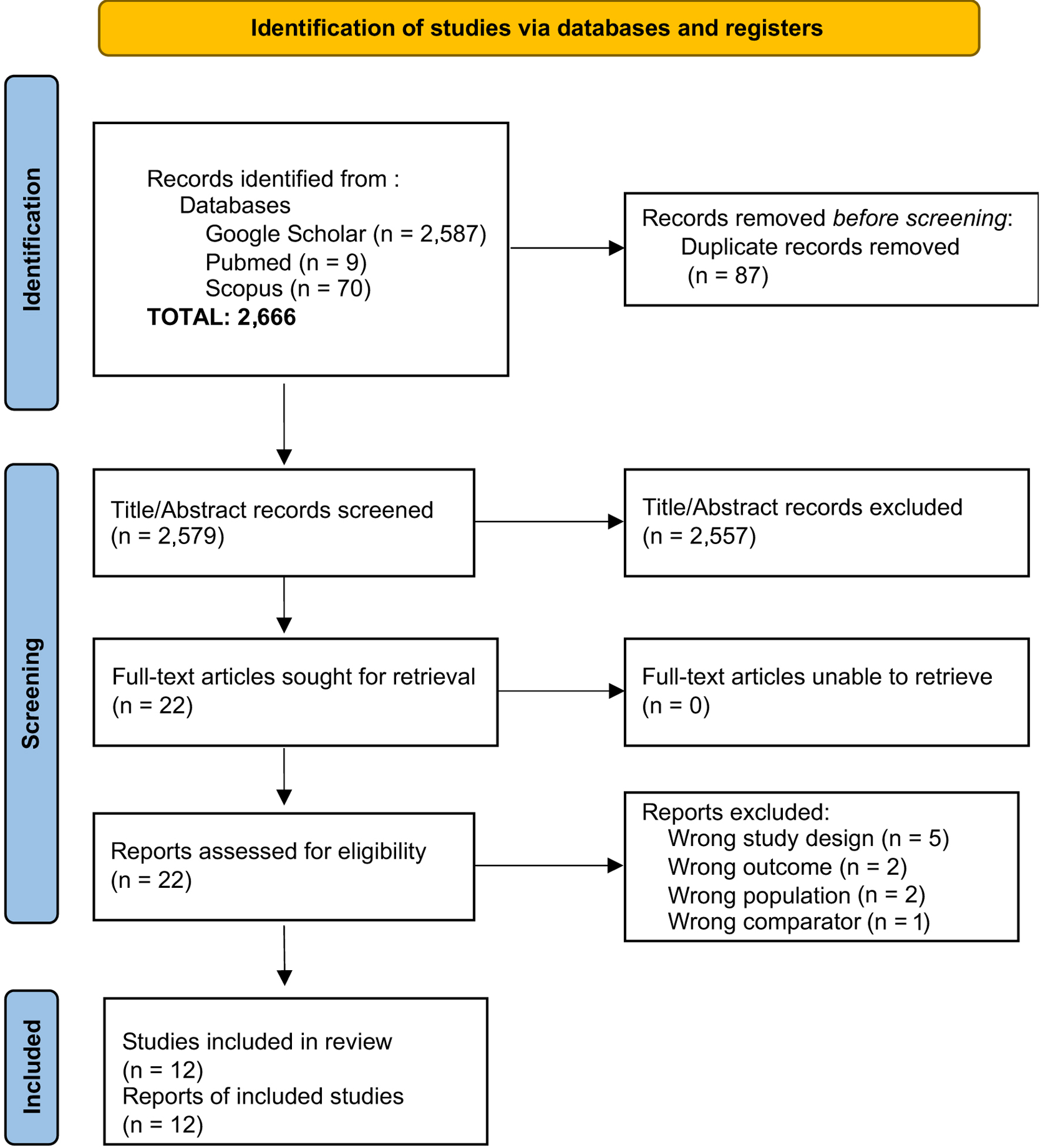
↓ Figure 1. Flow diagram of the study selection process for articles included in the systematic review and meta-analysis.
| Journal of Neurology Research, ISSN 1923-2845 print, 1923-2853 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Neurol Res and Elmer Press Inc |
| Journal website https://jnr.elmerpub.com |
Original Article
Volume 16, Number 2, June 2026, pages 88-98
Genotype-Guided Dual Antiplatelet Therapy in CYP2C19 Loss-of-Function Carriers With Stroke or Transient Ischemic Attack: A Meta-Analysis of Ticagrelor–Aspirin Versus Clopidogrel–Aspirin
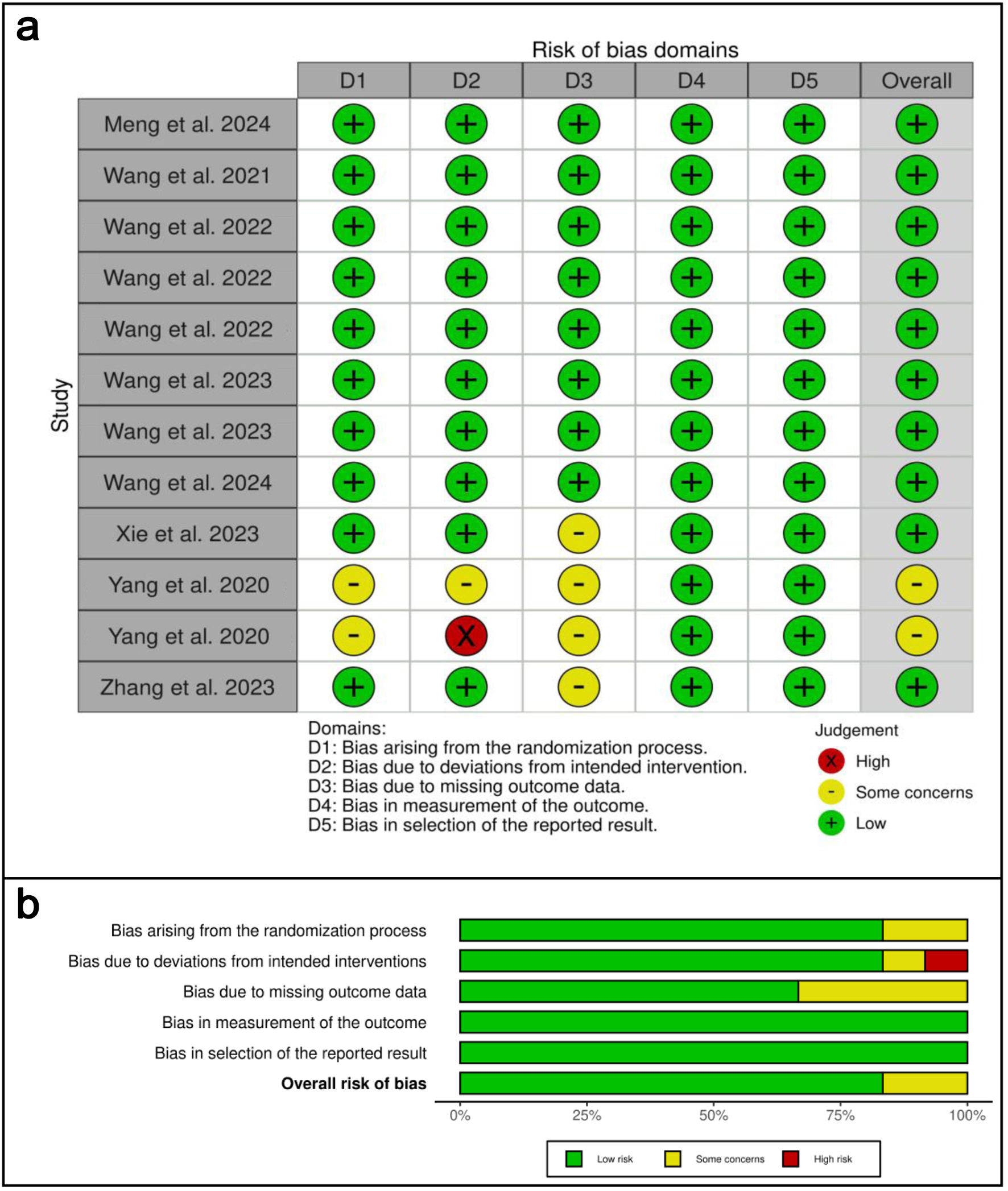
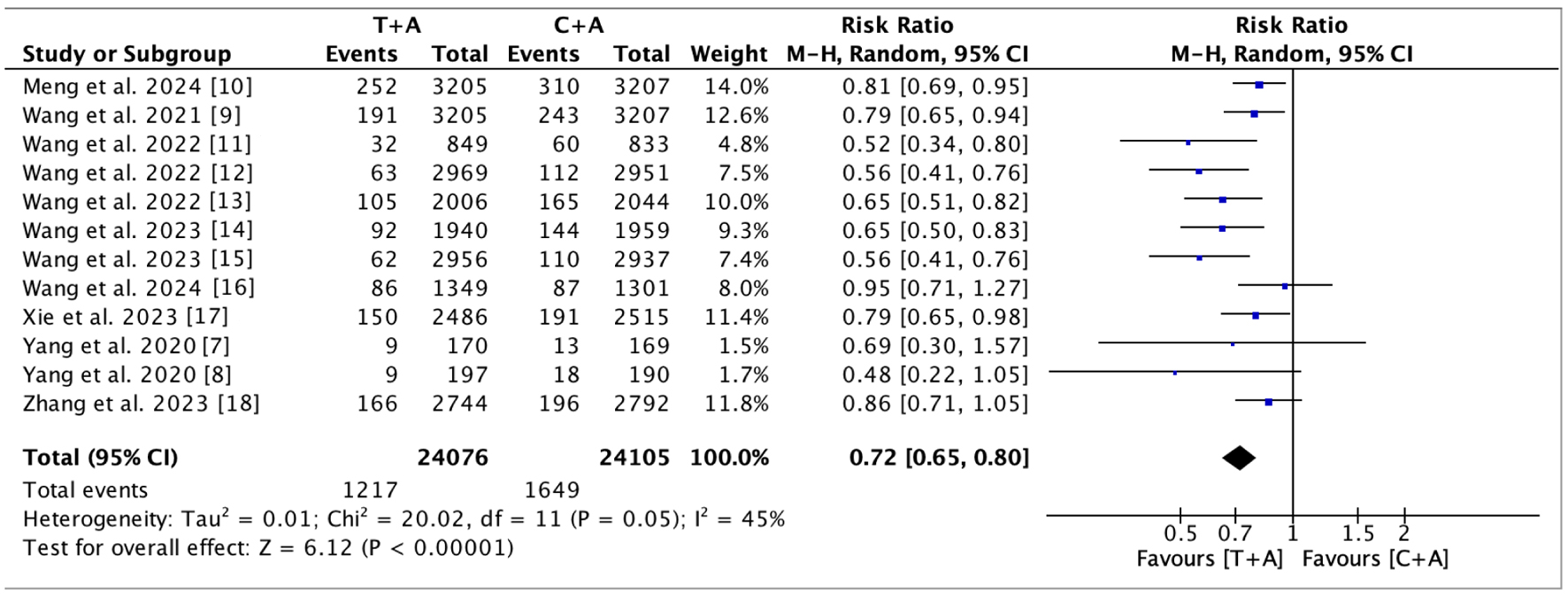
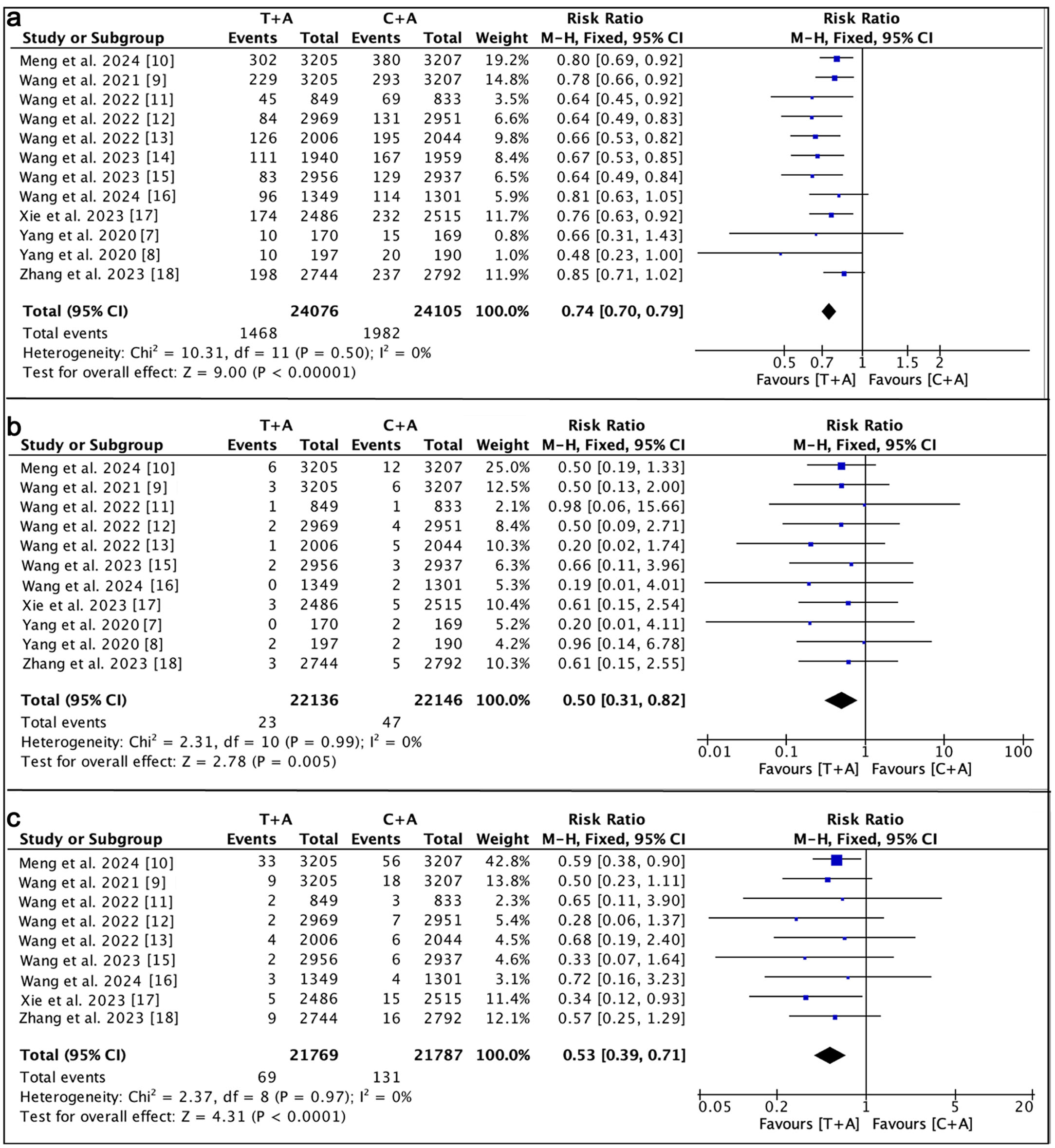
Figures

Table
| No. | Author | Sample size | Study design | Diagnosis | Intervention regimen | Control regimen | ||
|---|---|---|---|---|---|---|---|---|
| Ticagrelor | Aspirin | Clopidogrel | Aspirin | |||||
| 1. | Meng et al, 2024 [10] | 6,412 | The CHANCE-2 trial, a multicenter randomized double-blind trial | Transient ischemic attack who carried CYP2C19 loss-of-function alleles | Loading dose of 180 mg on day 1, followed by 90 mg twice daily for days 2–90 | Loading dose of 75–300 mg on day 1, followed by 75 mg daily for 21 days | Loading dose of 300 mg, followed by 75 mg daily for days 2–90 | Loading dose of 75–300 mg on day 1, followed by 75 mg daily for 21 days |
| 2. | Wang et al, 2021 [9] | 6,412 | Randomized, doubled-blind trial | Transient ischemic attack who carried CYP2C19 loss-of-function alleles | Loading dose of 180 mg on day 1, followed by 90 mg twice daily for days 2–90 | Loading dose of 75–300 mg on day 1, followed by 75 mg daily for 21 days | Loading dose of 300 mg, followed by 75 mg daily for days 2–90 | Loading dose of 75–300 mg on day 1, followed by 75 mg daily for 21 days |
| 3. | Wang et al, 2022 [11] | 6,412 | The CHANCE-2 trial, a multicenter randomized double-blind trial | Transient ischemic attack who carried CYP2C19 loss-of-function alleles | Loading dose of 180 mg on day 1, followed by 90 mg twice daily for days 2–90 | Loading dose of 75–300 mg on day 1, followed by 75 mg daily for 21 days | Loading dose of 300 mg, followed by 75 mg daily for days 2–90 | Loading dose of 75–300 mg on day 1, followed by 75 mg daily for 21 days |
| 4. | Wang et al, 2022 [12] | 6,412 | The CHANCE-2 trial, a multicenter randomized double-blind trial | Transient ischemic attack who carried CYP2C19 loss-of-function alleles | Loading dose of 180 mg on day 1, followed by 90 mg twice daily for days 2–90 | Loading dose of 75–300 mg on day 1, followed by 75 mg daily for 21 days | Loading dose of 300 mg, followed by 75 mg daily for days 2–90 | Loading dose of 75–300 mg on day 1, followed by 75 mg daily for 21 days |
| 5. | Wang et al, 2022 [13] | 6,378 | Post hoc analysis of a multicenter, randomized, double-blind, placebo-controlled trial | Transient ischemic attack who carried CYP2C19 loss-of-function alleles | Loading dose of 180 mg on day 1, followed by 90 mg twice daily for days 2–90 | Loading dose of 75–300 mg on day 1, followed by 75 mg daily for 21 days | Loading dose of 300 mg, followed by 75 mg daily for days 2–90 | Loading dose of 75–300 mg on day 1, followed by 75 mg daily for 21 days |
| 6. | Wang et al, 2023 [14] | 6,412 | The CHANCE-2 trial, a multicenter randomized double-blind trial | Transient ischemic attack who carried CYP2C19 loss-of-function alleles | Loading dose of 180 mg on day 1, followed by 90 mg twice daily for days 2–90 | Loading dose of 75–300 mg on day 1, followed by 75 mg daily for 21 days | Loading dose of 300 mg, followed by 75 mg daily for days 2–90 | Loading dose of 75–300 mg on day 1, followed by 75 mg daily for 21 days |
| 7. | Wang et al, 2023 [15] | 6,412 | The CHANCE-2 trial, a multicenter randomized double-blind trial | Transient ischemic attack who carried CYP2C19 loss-of-function alleles | Loading dose of 180 mg on day 1, followed by 90 mg twice daily for days 2–90 | Loading dose of 75–300 mg on day 1, followed by 75 mg daily for 21 days | Loading dose of 300 mg, followed by 75 mg daily for days 2–90 | Loading dose of 75–300 mg on day 1, followed by 75 mg daily for 21 days |
| 8. | Wang et al, 2024 [16] | 5,440 | The CHANCE-2 trial, a multicenter randomized double-blind trial | Transient ischemic attack who carried CYP2C19 loss-of-function alleles | Loading dose of 180 mg on day 1, followed by 90 mg twice daily for days 2–90 | Loading dose of 75–300 mg on day 1, followed by 75 mg daily for 21 days | Loading dose of 300 mg, followed by 75 mg daily for days 2–90 | Loading dose of 75–300 mg on day 1, followed by 75 mg daily for 21 days |
| 9. | Xie et al, 2023 [17] | 6,412 | The CHANCE-2 trial, a multicenter randomized double-blind trial | Transient ischemic attack who carried CYP2C19 loss-of-function alleles | Loading dose of 180 mg on day 1, followed by 90 mg twice daily for days 2–90 | Loading dose of 75–300 mg on day 1, followed by 75 mg daily for 21 days | Loading dose of 300 mg, followed by 75 mg daily for days 2–90 | Loading dose of 75–300 mg on day 1, followed by 75 mg daily for 21 days |
| 10. | Yang et al, 2020 [7] | 339 | Pre-specified subgroup analysis of PRINCE trial | Transient ischemic attack who carried CYP2C19 loss-of-function alleles | Loading dose of 180 mg on day 1, followed by 90 mg twice daily for days 2–90 | Loading dose of 300 mg on day 1, followed by 100 mg daily for 21 days | Loading dose of 300 mg, followed by 75 mg daily for days 2–90 | Loading dose of 300 mg on day 1, followed by 100 mg daily for 21 days |
| 11. | Yang et al, 2020 [8] | 675 | Pre-specified subgroup analysis of PRINCE trial | Transient ischemic attack who carried CYP2C19 loss-of-function alleles | Loading dose of 180 mg on day 1, followed by 90 mg twice daily for days 2–90 | Loading dose of 300 mg on day 1, followed by 100 mg daily for 21 days | Loading dose of 300 mg, followed by 75 mg daily for days 2–90 | Loading dose of 300 mg on day 1, followed by 100 mg daily for 21 days |
| 12. | Zhang et al, 2023 [18] | 6,412 | The CHANCE-2 trial, a multicenter randomized double-blind trial | Transient ischemic attack who carried CYP2C19 loss-of-function alleles | Loading dose of 180 mg on day 1, followed by 90 mg twice daily for days 2–90 | Loading dose of 75–300 mg on day 1, followed by 75 mg daily for 21 days | Loading dose of 300 mg, followed by 75 mg daily for days 2–90 | Loading dose of 75–300 mg on day 1, followed by 75 mg daily for 21 days |