Figures
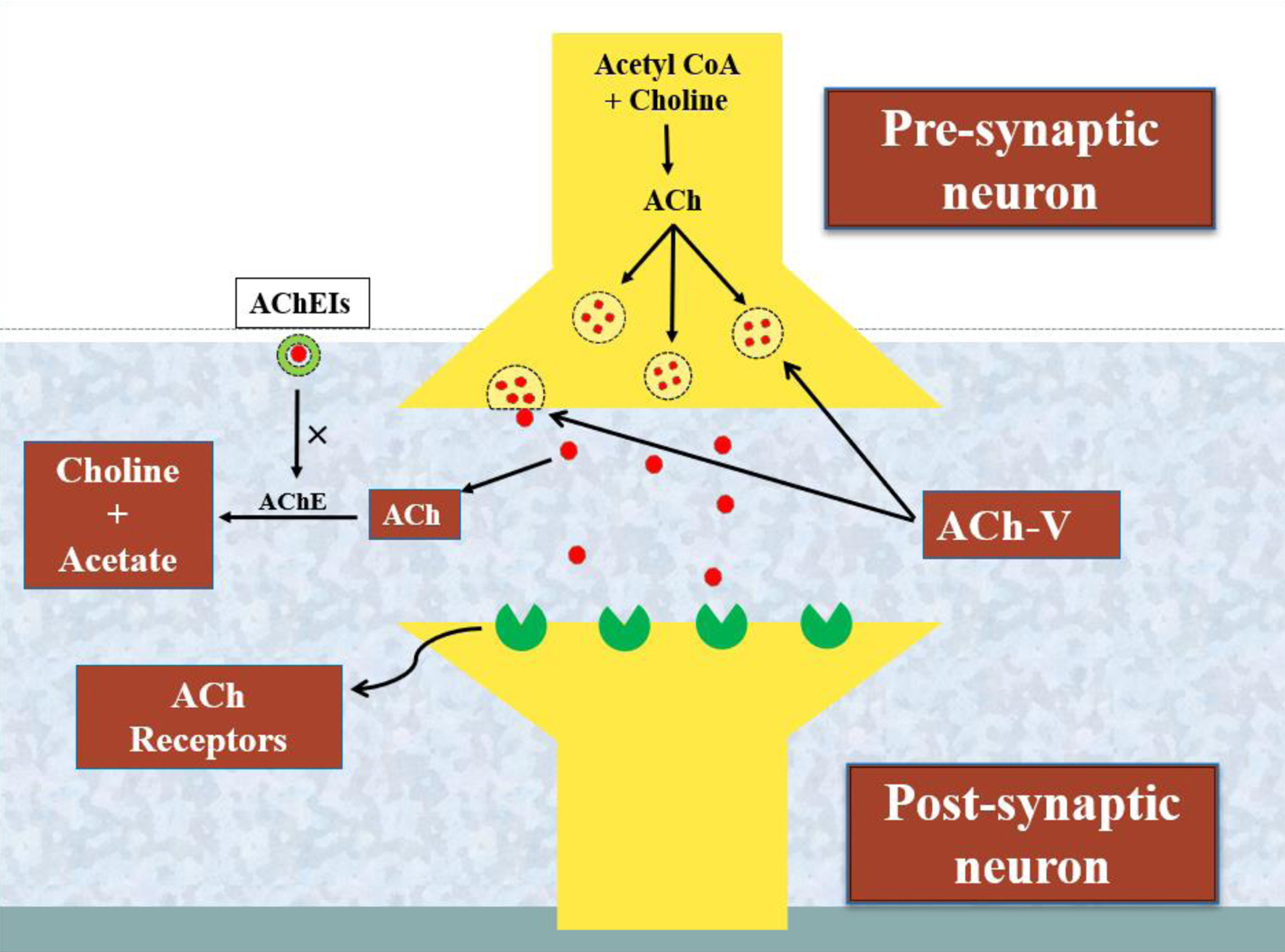
↓ Figure 1. A schematic representation of the
activity of acetylcholinesterase and the processing of the neurotransmitter acetylcholine. The enzyme
choline acetyltransferase converts acetyl-CoA and choline into acetylcholine (ACh). Furthermore, ACh is
released from ACh vesicles (ACh-V) to bind ACh receptors and performs a variety of neurological tasks
that are impaired by the degrading action of another enzyme, acetylcholinesterase (AChE). In this way,
the acetylcholinesterase inhibitors (AChEIs) reverse the brain functioning by blocking the activity of
AChE in Alzheimer’s disease (AD).
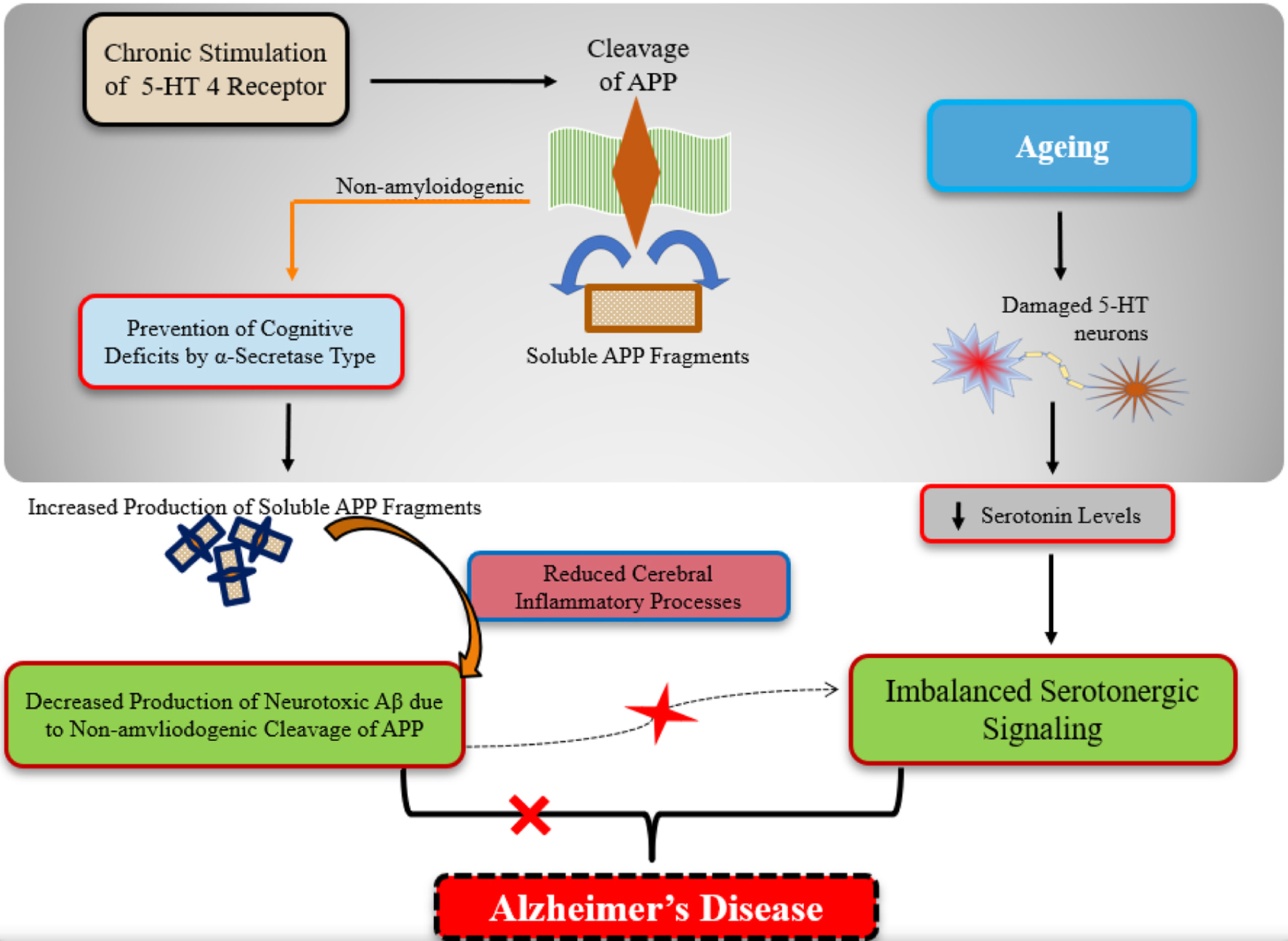
↓ Figure 2. A diagrammatic presentation of the
role of the serotonergic system signaling in aging and Alzheimer’s disease (AD). As we grow
older, the serotonergic neurons tend to get damaged which leads to impair the normal serotonergic
signaling in elderly. In case of AD, the chronic stimulation of the serotonin type-4 receptor leads to
the non-amyloidogenic α-secretase cleavage of amyloid precursor proteins (APPs). This cleavage
type results in the increased production of non-neurotoxic beta-amyloid (Aβ) fragments (i.e.,
soluble forms) and the depleted production of neurotoxic Aβ (i.e., non-soluble forms).
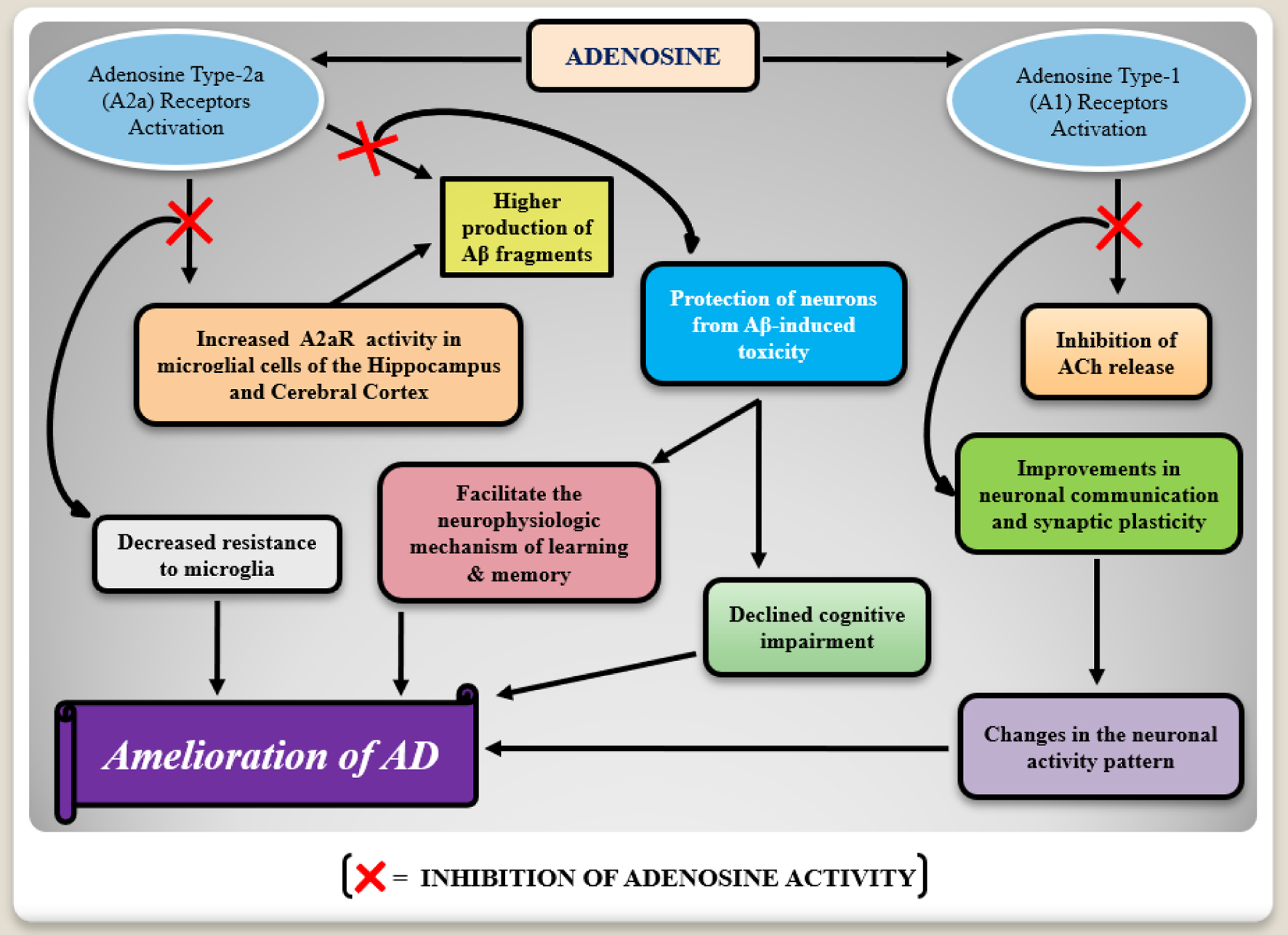
↓ Figure 3. A flow chart mechanism of the
adenosine type-1 (A1) and type-2 (A2) receptors involvement in Alzheimer’s
disease (AD) pathophysiology. Adenosine receptors activation seems to induce degenerative consequences.
Increased expressions of type-2 adenosine receptors (A2aR) in microglia may favor the production of
neurotoxic beta-amyloid (Aβ) proteins, whereas type-1 adenosine receptors are responsible for
acetylcholine (ACh) release inhibition which leads to the degenerative changes in the neuronal activity
pattern in AD. In this way, the inhibition of these receptors activity presents the cognition
improvement.
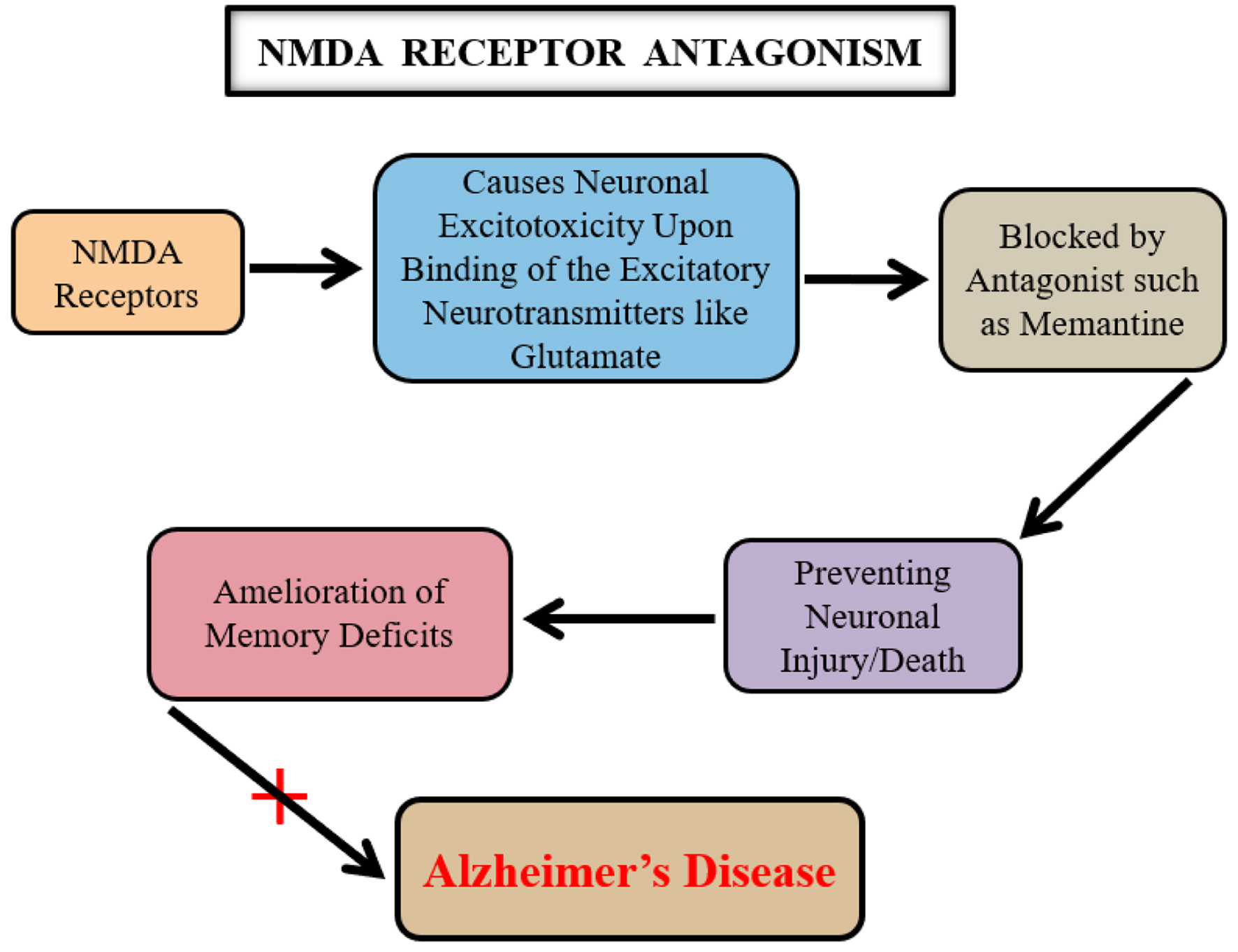
↓ Figure 4. A key relationship among the N-methyl
D-aspartate (NMDA) receptor signaling, glutamate, and Alzheimer’s disease (AD). The
neurotransmitter glutamate causes neuronal excito-toxicity upon its binding to the NMDA receptors. A
prolonged activation of the NMDA glutamate receptors leads to the neuronal dysfunctioning which is
potentially inhibited by using an NMDA receptor antagonist such as memantine. This mechanistic effect of
memantine is therefore counted as beneficial in AD.
Tables
↓ Table 1. Important Signaling Cascades and Their Primary Targets in the
Pathophysiology of AD [9-14]
|
Novel signaling pathways |
Major signaling components |
Effectors |
Effects |
| AD: Alzheimer’s disease; GABA: gamma-amino butyric acid; NMDA: N-methyl
D-aspartate. |
| Cholinergic signaling pathway |
Acetylcholinesterase, an enzyme |
Donepezil |
Inhibitory |
| NMDA-glutamate signaling pathway |
Glutamate, a neurotransmitter |
Memantine |
Antagonism |
| Serotonin signaling pathway |
Serotonin, a monoamine |
Clozapine |
Modulatory |
| GABAergic signaling pathway |
GABA, a neurotransmitter |
Homo-taurine |
Upregulatory |
| Adenosine signaling pathway |
Adenosine, a nucleoside |
Caffeine |
Modulatory/antagonism |
| Histamine signaling pathway |
Histamine, an immunogenic |
Dimebolin |
Antihistamine |
↓ Table 2. Drug Entities Under Phase 3 Clinical Trials Against Possible
Molecular Cascades for the Potential Futuristic Therapeutics in AD Treatment Scenario
|
Molecular signaling cascades |
Agents in phase 3 clinical trials |
References |
| AD: Alzheimer’s disease; GABA: gamma-amino butyric acid; NMDA: N-methyl
D-aspartate. |
| Cholinergic system signaling |
Blarcamesine (ANAVEX2-73) |
18-20 |
|
KarXT |
21, 22 |
| NMDA receptor signaling |
AXS-05 |
20, 22, 23 |
| Serotonin receptor signaling |
Masupirdine |
21, 22 |
|
OPC-34712 |
19-23 |
|
Piromelatine |
21, 22 |
| GABAergic system signaling |
None |
- |
| Adenosine receptor signaling |
Caffeine |
19-22 |
| Histaminergic system signaling |
None |
- |



