| Journal of Neurology Research, ISSN 1923-2845 print, 1923-2853 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Neurol Res and Elmer Press Inc |
| Journal website https://jnr.elmerpub.com |
Case Report
Volume 15, Number 3, August 2025, pages 123-129
Atypical Presentation of Herpes Simplex Virus Type 1 Meningoencephalitis With Nonclassical Imaging in a Young Adult
Taseal Ahmeda, Amy Heraia, c, Samir Ruxmohanb
aSchool of Medicine, St. George’s University, University Centre, True Blue, St.
George’s, Grenada
bNeurology and Neurocritical Care, Exceed Healthcare,
Dallas, TX 75208, USA
cCorresponding Author: Amy Herai, School of Medicine, St.
George’s University, University Centre, True Blue, St. George’s, Grenada
Manuscript submitted May 7, 2025, accepted August 9, 2025, published online August 28,
2025
Short title: Atypical Case of HSV-1 Meningoencephalitis
doi:
https://doi.org/10.14740/jnr1033
| Abstract | ▴Top |
Herpes simplex virus type 1 (HSV-1) is a leading cause of viral meningoencephalitis. Clinical management requires timely diagnosis and treatment, yet early detection can be hindered by atypical imaging and antiviral resistance with rapid neurological decline. We report a case of a 35-year-old previously healthy man, who developed subacute headache, fever, and altered mental status following recent travel. Initial cerebrospinal fluid (CSF) analysis showed elevated protein and red blood cells without significant pleocytosis. Repeat lumbar puncture (LP) returned positive HSV-1 polymerase chain reaction (PCR) and elevated opening pressures. Despite empiric antiviral therapy and intravenous immunoglobulin, his condition rapidly deteriorated requiring intubation, lumbar drainage, and hypertonic therapy. Magnetic resonance imaging (MRI) showed cortical ribboning on diffusion-weighted imaging (DWI) and fluid-attenuated inversion recovery (FLAIR) sequences without classical frontotemporal involvement. Initial treatment resistance and suspicion for autoimmune encephalitis prompted early initiation of plasma exchange, intravenous immunoglobulin and steroids. Despite aggressive management, the patient progressed to septic shock and multiorgan failure, ultimately transitioning to comfort care. This case highlights the diagnostic and therapeutic challenges in HSV-1 meningoencephalitis with atypical imaging, nonclassical CSF findings, and lack of response to multimodal treatment regimes. It underscores the importance of maintaining a broad differential, initiating early empiric therapy, and integrating serial imaging and repeat diagnostics in cases of rapid neurological decline. Early consideration of autoimmune encephalitis and timely immunotherapy may be warranted when clinical features are ambiguous. HSV-1 encephalitis should remain high on the differential even when classical radiographic and laboratory markers are absent.
Keywords: HSV-1; Meningoencephalitis; Atypical MRI; Cortical ribboning
| Introduction | ▴Top |
One of the leading causes of meningitis and encephalitis where prognosis is dependent on timely diagnosis and treatment initiation is herpes simplex virus (HSV). Approximately 90% of cases are attributed to herpes simplex virus type 1 (HSV-1) [1]. The virus can cause severe manifestations with encephalitis usually caused by infection or autoimmune etiology [2]. The reactivation of the virus from an earlier infection results in apoptosis in the central nervous system (CNS) parenchyma, leading to temporal lobe lesions and presenting symptoms of fever, altered level of consciousness, seizures, and focal neurological deficits [2, 3]. HSV meningoencephalitis is associated with high mortality, morbidity, and financial burden; untreated mortality approaches 70% and is associated with neurological complications such as dysphasia, amnesia, and behavioral disturbances [2, 4]. Magnetic resonance imaging (MRI) imaging is pivotal in early diagnosis of HSV meningoencephalitis, typically revealing signal abnormalities in the mesial temporal lobes, inferior frontal lobes, and insular cortex [5]. While MRI findings are primarily used for diagnosis, they have also been explored as a prognostic tool specifically in HSV meningoencephalitis [5]. Cerebrospinal fluid (CSF) cultures and gram stains are not reliable for diagnosing viral meningoencephalitis [6]. HSV CSF analysis has shown xanthochromia, pleocytosis with increased protein levels and normal or slightly elevated glucose or lactate concentration, though confirmation of an active infection is based on molecular tests [7]. Polymerase chain reaction (PCR) is the diagnostic method of choice for HSV-1 meningoencephalitis, offering high sensitivity and specificity in detecting HSV-1 DNA, even on the first day of infection [6]. We present a unique case highlighting the complexities in diagnosing and treating HSV-1 meningoencephalitis when rapid neurological deterioration occurs and standard diagnostic tests yield atypical results.
| Case Report | ▴Top |
A 35-year-old male with no significant past medical history presents to an outside facility with several days of fever (maximum temperature 39.4 °C at presentation), headache, nausea, vomiting, diarrhea and neck stiffness. He reported having a fever not responsive to antipyretics for 1 week following recent travel with his wife to Southeast Asia (Bali, Indonesia). At the outside emergency department, he was started on empiric antibiotics and underwent an unsuccessful lumbar puncture (LP) (Table 1, LP1). He was subsequently admitted to the intensive care unit (ICU). Initial non-contrast head computed tomography (CT) was unremarkable. Following ICU admission, a successful LP yielded CSF with a protein of 123 mg/dL (normal 15 - 45 mg/dL), red blood cell (RBC) count of 9,000 cells/mm3 (normal 0 cells/mm3), white blood cell (WBC) count of 11 cells/mm3 (normal 0 - 5 cells/mm3), and glucose of 81 mg/dL (normal 40 - 70 mg/dL), but opening pressure was not obtained (Table 1, LP2). Meningitis and encephalitis panels that included HSV-1 and HSV-2 testing returned negative. The patient’s neurological condition deteriorated, requiring readmission to the ICU due to worsening mentation and agitation refractory to Precedex drip. A routine electroencephalogram (EEG) performed at the outside facility was unremarkable and showed no significant epileptic waveforms or lateralized periodic discharges (LPDs). Initial diagnostic impressions revolved around infectious meningoencephalitis with an unclear etiology. Although the patient did not meet the threshold for systemic inflammatory response syndrome (SIRS) criteria, the patient was started on empiric antibiotics of ceftriaxone, vancomycin and acyclovir. There was no elevated leukocytosis after RBC count correction on LP CSF analysis (corrected CSF WBC = measured CSF WBC - (CSF RBC/500)), which raised concern for possible autoimmune encephalitis. Due to the possible differential of autoimmune encephalitis, the patient was initiated on intravenous immunoglobulin (IVIG). The outside facility ICU was unable to complete the IVIG course and due to the possible need for plasma exchange (PLEX) therapy, the patient was transferred to our institution for a higher level of care.
 Click to view |
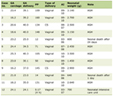
Table 1. Summary of Cerebrospinal Fluid
Findings From Four LPs Performed Over the Clinical Course |
On arrival, the patient was severely more obtunded and required intubation for airway protection later in the evening. A neurological exam on fentanyl was significant for following simple commands, cranial nerves were intact, moving both upper and lower extremities to noxious stimuli, and grimacing to noxious stimuli. Brain MRI was unremarkable and had no evidence of cortical ischemia or mass lesions. PLEX was then initiated with the guidance from the nephrology team due to the suspected differential of autoimmune encephalitis and was tolerated by the patient. The Infectious Disease Unit was consulted for infectious meningoencephalitis as it was higher on the differential, and the consensus was to continue antiviral and antibacterial coverage with further workup. The patient was subsequently restarted on intravenous (IV) acyclovir, ceftriaxone and vancomycin and high-dose steroids for possible HSV meningoencephalitis. CT chest/abdomen/pelvis scan for malignancy was negative. Clinical staff reported right-sided tremor movements, indicating the need for EEG, which demonstrated generalized polymorphic slowing with no epileptiform discharges or seizures and was discontinued as there was no correlation between the EEG and right-sided tremors. MRI brain without contrast showed no enhancement, masses, or strokes but revealed possible diffusion-weighted imaging (DWI) cortical ribboning with corresponding T2 fluid-attenuated inversion recovery (FLAIR) signal abnormalities (Figs. 1, 2). A repeat LP guided by interventional radiology demonstrated an opening pressure of 28 cm H2O (normal 10 - 20 cm H2O) (Table 1, LP3) and yielded a positive HSV-1 PCR, CSF reactive HSV-1 immunoglobulin (Ig)G with negative CSF HSV-1 IgM, and negative HSV-2 PCR and CSF IgG/IgM, while the rest of the meningitis/encephalitis PCR panel remained otherwise negative. This prompted holding of the empiric antibiotics and continued treatment with acyclovir and the systemic steroids. Given the concern for a secondary autoimmune process, an anti-N-methyl-D-aspartate (NMDA) receptor antibody test and a broad autoimmune encephalitis panel (ENS2 panel including LGI1, CASPR2, AMPA-R, and GABA_B-R antibodies, anti-Glycine-R, anti-DPPX, anti-mGluR5) were sent; all antibodies returned negative, effectively excluding the most common antibody-mediated encephalitis. A third LP again showed elevated opening pressure of 30 cm H2O (normal 10 - 20 cm H2O), and the patient exhibited facial grimacing on arousal and pain on neck extension, further indicating intracranial hypertension despite the absence of overt CSF inflammation on previous imaging (Table 1, LP4). The patient underwent lumbar drain placement for increased intracranial pressure (ICP) which drained about 10 mL per hour. Hypertonic saline was also initiated due to elevated lumbar pressures. Repeat MRI brain with contrast had no significant findings compared to the last study. Neurological exam after lumbar drain placement showed improvement with following commands, but the decreased mentation was a barrier to extubating the patient and liberating them from the ventilator.
 Click for large image |
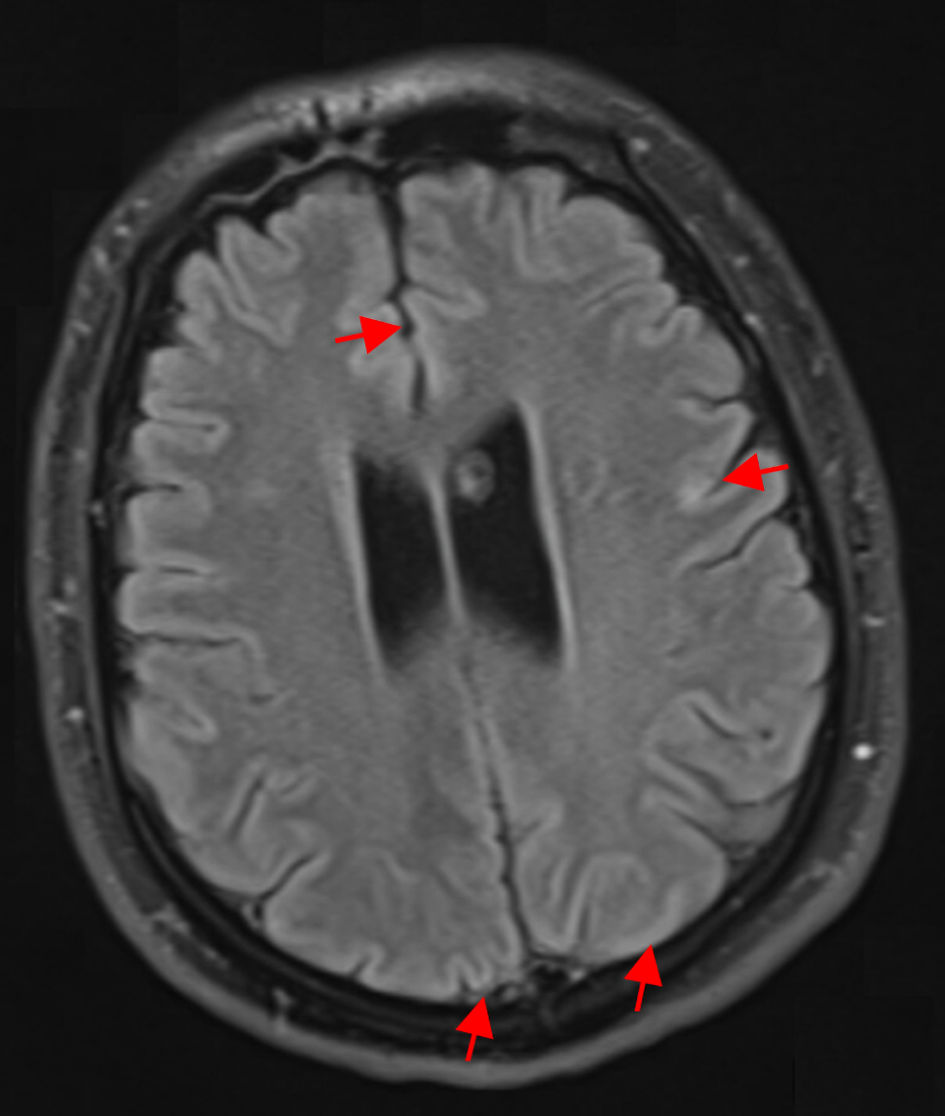
Figure 1. Axial FLAIR MRI without contrast at the level of the temporal lobes, demonstrating absence of the expected mesial temporal hyperintensities typically seen in HSV-1 encephalitis. There is curvilinear diffusion hyperintensity in the frontal and parietal cerebral cortex (red arrows). Both hippocampi, amygdalae, and insular cortices maintain normal signal intensity and grey-white differentiation. This lack of medial temporal FLAIR hyperintensity is atypical for HSV-1 encephalitis, which classically presents with unilateral or bilateral temporal lobe FLAIR hyperintensities and edema. FLAIR: fluid-attenuated inversion recovery; MRI: magnetic resonance imaging; HSV-1: herpes simplex virus type 1. |
 Click for large image |
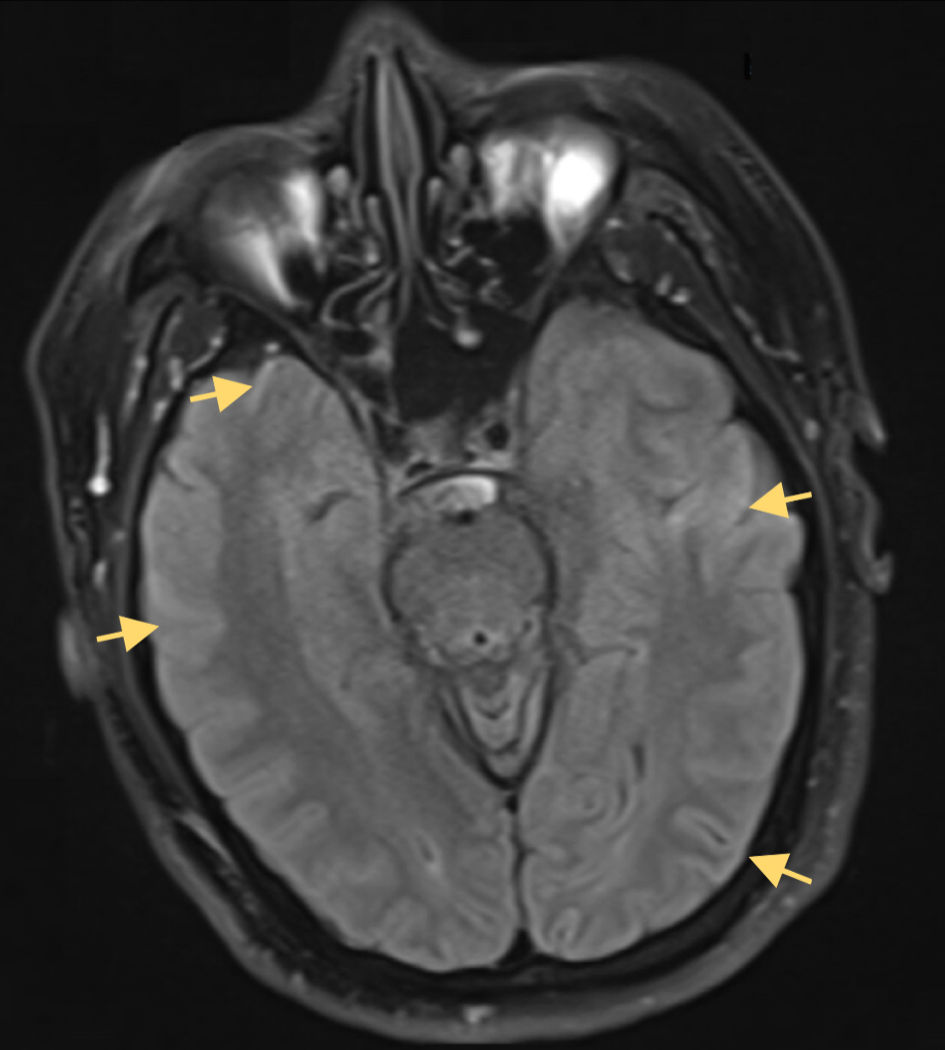
Figure 2. Axial DWI MRI without contrast of the brain demonstrating cortical ribboning curvilinear diffusion hyperintensity along the cerebral cortex in the temporal and parietal lobes (yellow arrows). The corresponding FLAIR sequence (Fig. 1) also showed cortical hyperintensities. No abnormal diffusion or signal is noted in the medial temporal lobes or insula, which is atypical for HSV-1 encephalitis. Classically, HSV-1 MRI lesions involve the mesial temporal and inferior frontal regions; in this case, the cortical ribboning pattern indicates a more diffuse cortical involvement and can be seen in encephalitic processes but is nonspecific. FLAIR: fluid-attenuated inversion recovery; DWI: diffusion-weighted imaging; MRI: magnetic resonance imaging; HSV-1: herpes simplex virus type 1. |
During the subsequent ICU course, the patient developed worsening clinical status complicated by bloodstream infections and ventilator-associated pneumonia, leading to septic shock and multiorgan dysfunction. Despite broad-spectrum antibiotics and aggressive supportive care, his condition continued to deteriorate. Given the patient’s poor neurological prognosis and escalating medical complexity, goals of care discussions were initiated with the family. After multiple interdisciplinary meetings, a decision was made to transition the patient to do-not-resuscitate/do-not-intubate (DNR/DNI) status and pursue comfort-focused measures. Life-sustaining therapies were subsequently withdrawn, and the patient was compassionately transitioned to end-of-life care.
| Discussion | ▴Top |
Diagnostic considerations and therapeutic challenges in HSV-1 meningoencephalitis
HSV-1 meningoencephalitis carries a high risk of morbidity and mortality, especially without anti-viral treatment, despite advances in therapy [6]. Our case highlights the varying degrees of clinical presentation with meningoencephalitis, which may include changes in consciousness, behavioral abnormalities, focal neurologic signs, and seizures [6, 7]. The symptoms include both an encephalopathic component and signs of meningeal inflammation, coining the term “meningoencephalitis” to reflect the coexistence of these conditions [7]. Given the patient’s recent travel to Indonesia, several tropical infections were considered. Southeast Asia is endemic for dengue, Zika, chikungunya, and Japanese encephalitis, all of which can lead to fever and encephalitis [8, 9]. Dengue is hyperendemic in Bali and can rarely cause CNS involvement, though our patient lacked rash or hemorrhagic signs, and arboviral serologies were negative. Rabies was unlikely without animal exposure or hydrophobic spasms [8, 9]. Other regional pathogens (e.g., Burkholderia pseudomallei, Mycobacterium tuberculosis) were also evaluated: PCR panels were negative, and imaging did not suggest these etiologies [8]. Ultimately, extensive infectious workup including a multiplex encephalitis panel was negative, keeping HSV-1 as the principal diagnosis despite atypical features. Given the broad infectious differential diagnoses, early initiation of empiric acyclovir and broad-spectrum antibiotics is crucial, as bacterial meningoencephalitis and septic encephalopathy can mimic HSV encephalopathy [7, 10]. PCR analysis of CSF remains the gold standard for confirming HSV-1 meningoencephalitis, with high sensitivity and specificity; however, false-negative results can occur, particularly when testing is performed early in the disease course or if the sample is contaminated with blood [7, 11]. Roberts et al compellingly demonstrated that two consecutive negative HSV-1 PCR results can precede a definitive diagnosis, highlighting the necessity of repeated testing and sustained clinical vigilance in suspected HSV encephalitis [12]. Autopsy-confirmed HSV-1 encephalitis has been reported with two initial negative CSF PCRs, only turning positive on a third sample, and a recent review identified over a dozen cases requiring repeat LP or biopsy for confirmation [13, 14]. Thus, a negative PCR especially early in a case does not exclude HSV-1. Repeating PCR after 3 - 7 days is recommended in cases with high clinical suspicion of HSV; however, data suggest that HSV DNA may be cleared from the CSF with antiviral treatment by that time, and delays in performing LP and molecular analysis can obscure accurate diagnosis [7]. Unbiased metagenomic next-generation sequencing (mNGS) of CSF can detect bacteria, viruses, fungi, and parasites without prior assumptions, and has proven diagnostic value in meningitis and encephalitis. In a prospective multicenter study of 204 patients, mNGS identified additional pathogens in 6% of cases not found by conventional testing but required a median turnaround of 48 - 72 h, incurred high per-sample costs, and was available only at specialized centers [15]. Clinical metagenomics also demands complex bioinformatics pipelines and lacks standardized workflows, limiting its routine use outside research settings [16]. Given our patient’s high pre-test probability for HSV-1, the established sensitivity (94-98%) and rapidity of CSF HSV PCR, and the urgency to start acyclovir, we elected to rely on repeat targeted PCR and empirical therapy rather than mNGS. Moreover, comprehensive mNGS workflows incur substantial per-sample costs when accounting for reagents, library preparation, sequencing, and bioinformatics, making adoption infeasible for many community hospitals without specialized microbiology budgets or bioinformatics expertise [15]. Cytochemical analysis of CSF typically reveals hyperproteinorrachia, normoglycorachia, and lymphocytic pleocytosis, supporting a diagnosis of meningoencephalitis when clinical presentation also aligns [17]. Clinical presentation or CSF cytochemical analysis alone does not meet the threshold to rule out other infectious causes of meningoencephalitis and encephalitis without a positive HSV PCR [7]. Case studies have shown better outcomes when rapid diagnosis and initiation treatment with antiviral therapy occur within 48 h of symptom onset [7]. Acyclovir remains the first-line treatment, with ganciclovir as an alternative in cases of acyclovir-related renal failure [18]. IV acyclovir is started on 10 mg/kg every 8 h for 14 - 21 days, which should be started as soon as HSV encephalitis is suspected, even before test results confirm the diagnosis [7]. With acyclovir, renal function is monitored due to kidney toxicity, and hydration along with supportive care is maintained to prevent acute kidney injury [18]. If acyclovir is not tolerated due to renal failure or resistance, the alternative treatment option is ganciclovir, but its effectiveness in treating HSV encephalitis is not as well defined [18]. Despite prompt intervention, HSV-1 meningoencephalitis carries a substantial risk of mortality and long-term neurological deterioration, raising the concern for heightened clinical suspicion and early therapeutic intervention [6, 7].
Atypical neuroimaging features and diagnostic challenges in HSV-1 meningoencephalitis
MRI plays a crucial role in the early diagnosis of HSV meningoencephalitis, as it frequently reveals signal abnormalities in the mesial temporal lobes, inferior frontal lobes, and insula [5]. FLAIR and DWI lesions in the temporal lobes are hallmark findings in HSV-1 meningoencephalitis, particularly when the etiology of the infection is unclear [5]. Studies have shown that a more severe acute disease course correlates with the presence of cytotoxic lesions with lower apparent diffusion coefficient (ADC) values, which predict worse long-term functional outcomes [5]. Additionally, the extent of brain involvement, such as bilateral DWI lesions or FLAIR abnormalities affecting more than three lobes, has been linked to poorer outcomes in the clinical setting [5]. However, despite the hallmark characteristic imaging patterns involving the frontotemporal lobes, clinical manifestations and MRI findings alone are not always sufficient for a definitive diagnosis of HSV [19]. While some studies, such as that by Shirani et al, have reported a strong correlation between HSV-1 PCR positivity and frontotemporal MRI lesions, other investigations have failed to establish this association [6]. Moreover, repeat MRI testing does not necessarily reflect disease progression or treatment efficacy, limiting its utility in guiding ongoing management [7]. In our case, the patient did not have the classical frontotemporal involvement of HSV-1 but exhibited DWI cortical ribboning with corresponding T2 FLAIR imaging with curvilinear diffusion hyperintensity (Figs. 1, 2). This lack of classical frontotemporal involvement, along with the atypical neuroimaging pointing towards other differentials, made it difficult to correlate radiographic findings with the patient’s clinical deterioration or therapeutic response. Atypical neuroimaging patterns of HSV have been described. Samaan et al detail an HSV encephalitis with diffuse multifocal FLAIR hyperintensities (cortex, brainstem) and negative PCR, ultimately diagnosed by HSV IgG in CSF [20]. Others have reported HSV-1 presenting like ischemic stroke or with extra-limbic lesions [21]. Such cases underscore that HSV-1 can present with normal initial MRI or nonclassical lesions [21]. Therefore, HSV-1 meningoencephalitis must be considered even when CSF PCR is negative and imaging is nonclassical, provided the clinical picture is suggestive. The observed cortical ribboning, while suggestive of a diffuse cortical process, was nonspecific and further confounded the diagnostic clarity, raising uncertainty about whether the underlying pathology was infectious, autoimmune, or metabolic in origin. It is important to recognize that cortical ribboning on DWI/FLAIR is not pathognomonic for HSV-1 encephalitis. Sporadic Creutzfeldt-Jakob disease (sCJD) classically produces gyriform diffusion hyperintensities in both cortex and basal ganglia, often preceding clinical myoclonus and dementia on real-time quaking-induced conversion (RT-QuIC) or 14-3-3 testing [22]. The patient’s clinical presentation did not align with CJD, as there was no evidence of rapidly progressive dementia, no myoclonic jerks on examination, nor were there any characteristic periodic sharp-wave complexes on EEG, obviating the need for further prion biomarker testing. Metabolic encephalopathies, like severe hypoglycemia, can likewise produce reversible cortical restricted diffusion, particularly in parieto-occipital regions, with DWI changes appearing within hours of insult and pseudonormalizing over several days once euglycemia is restored [23]. In severe metabolic encephalopathies, like acute hepatic failure, hyperammonemia induces cytotoxic edema, producing transient cortical diffusion restriction, often in parieto-occipital regions, which typically resolves with correction of liver dysfunction [24, 25]. Certain post-infectious or deficiency states, such as Wernicke’s encephalopathy, have also been reported to exhibit cortical ribboning on MRI when thiamine levels are critically low, underscoring the need to consider nutritional and post-infectious causes when this finding is encountered [26]. Likewise, prolonged seizures or status epilepticus can lead to curvilinear cortical DWI hyperintensities reflecting ictal neuronal injury; these diffusion changes frequently normalize within days after seizure control [27]. In our patient, normal serum glucose, liver function tests, lack of nutritional risk factors, and no EEG evidence of clinical seizures argued strongly against metabolic or seizure-related etiologies, leaving HSV-1 meningoencephalitis as the most likely diagnosis. Similarly, cortical ribboning sign on DWI has been observed following successful reperfusion therapy in acute ischemic stroke, our patient had no history of ischemic events and had no vascular abnormalities detected on imaging [28]. Our case highlights the complexity of diagnosing HSV-1 meningoencephalitis when neuroimaging fails to show lesions in frontotemporal regions and the importance of integrating clinical, laboratory, and imaging data to avoid misdiagnosis.
Autoimmune encephalitis consideration in antiviral refractory HSV-1 encephalitis
Autoimmune encephalitis should be considered in cases where there is no clinical improvement despite appropriate antiviral and antibacterial therapy, as seen in our patient [7]. HSV-1 meningoencephalitis can trigger a secondary autoimmune response, most commonly anti-NMDA receptor encephalitis, further complicating the clinical picture [7]. Alternative immune-mediated processes should be considered when there is persistence of neurological deterioration despite acyclovir, coupled with a negative repeat HSV PCR [5]. Given the possibility of autoimmune encephalitis, PLEX and IVIG can be initiated as key therapeutic interventions, aimed at modulating the immune response and removing circulating autoantibodies [5, 7]. High-dose steroids can also be used in combination with PLEX and IVIG, given the overlapping presentation of HSV-1 meningoencephalitis and autoimmune encephalitis, reinforcing the need for early initiation of immunotherapy in antiviral treatment resistant cases [7]. The patient’s worsening neurological status, elevated LP pressures, and lack of frontotemporal lesions on imaging supported the decision to pursue immunotherapy early, despite initial anti-viral treatment. Evidence suggests that PLEX yields a clinical response in approximately 64-75% of patients with autoimmune encephalitis, particularly in refractory cases [29, 30]. IVIG is frequently combined with corticosteroids and is regarded as an effective first-line therapy [29]. Although direct comparative studies are limited, both treatments have demonstrated meaningful clinical benefits in managing autoimmune encephalitis [29, 30]. Recognizing the distinction between HSV-1 meningoencephalitis and autoimmune encephalitis is crucial, as autoimmune cases often present with cortical or basal ganglia involvement rather than temporal lobe changes, and CSF findings may reveal oligoclonal bands with mild pleocytosis but normal glucose levels [5]. Early identification and timely immunotherapy are essential in such cases to optimize patient outcomes and prevent long-term neurological complications [5].
Management of HSV meningoencephalitis complications
HSV meningoencephalitis is known to cause cytotoxic edema during early viral replication, followed by vasogenic edema from the host inflammatory response, both of which contribute to elevated opening pressures and ICP [31]. Both of these clinical signs in HSV meningoencephalitis are poor prognostic indicators that necessitate timely intervention [31]. In our patient, rising LP opening pressures, facial grimacing on arousal, and decreased mentation prompted placement of a lumbar drain, an uncommon but sometimes life-saving step. While not routine, CSF diversion via lumbar or ventricular drains has been reported in severe encephalitis to relieve pressure, with lumboperitoneal shunts showing immediate symptom improvement in about 89% of viral meningitis cases and ventriculoperitoneal shunts in about 80% [32]. Safain et al described combining medical therapy (hyperosmolar agents, steroids) with surgical interventions (ventriculostomy, shunting) in refractory HSV meningoencephalitis [31]. Our patient improved clinically after CSF drainage, supporting this aggressive approach. Although corticosteroids have shown outcome benefits in HSV-1 meningoencephalitis by reducing late-phase vasogenic edema, our patient received steroids empirically with no change in the clinical status. Given that acute viral encephalitis can cause elevated ICP and is a known poor prognostic indicator, direct ICP monitoring could have influenced management strategies and guided timely therapeutic interventions. Furthermore, a common complication of viral encephalitis due to elevated ICP are seizures causing secondary brain injury [33]. EEG findings can be pivotal in HSV diagnosis, often demonstrating LPDs, generalized or focal slowing, and periodic sharp waves in frontotemporal regions [20, 33]. In our case, continuous EEG showed generalized slowing without epileptiform discharges and were not specific to any cortical area that HSV-1 usually targets. Evidence has shown that continuous EEG can be used as a marker for progression of disease and thus may prompt prophylactic or therapeutic administration of antiepileptic drugs to prevent any further injury [20, 33]. The absence of hallmark features typically associated with HSV-1 meningoencephalitis can significantly complicate clinical decision-making, resulting in lack of initiation or escalation of treatments in patients who lack definitive diagnostic indicators despite a high index of suspicion
Conclusions
This case demonstrates the diagnostic complexity of HSV-1 meningoencephalitis with rapid neurological deterioration in the absence of classical imaging findings and definitive early diagnostic markers. Despite high clinical suspicion with nonclassical MRI brain features and atypical CSF analysis with positive HSV-1 PCR, the patient’s condition was resistant to standard anti-viral therapy and subsequently developed rapid neurological decline and elevated lumbar pressure. This prompted discussion on possible autoimmune etiology, necessitating empiric immunotherapy and aggressive supportive care. Ultimately, this case highlights the importance of maintaining a broad differential diagnosis and initiating early empiric therapy in deteriorating patients, even in the absence of confirmatory data. Atypical clinical presentations, nonspecific imaging, and false-negative test results can obscure the underlying etiology and delay time-sensitive interventions. Integration of evolving clinical findings, serial imaging, and repeat diagnostic testing is essential in tailoring management for complex neuroinfectious diseases. Prompt recognition and intervention remain critical to improving outcomes in HSV-1 meningoencephalitis, particularly when standard diagnostic paradigms fail to yield definitive answers.
Acknowledgments
None to declare.
Financial Disclosure
The authors received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The authors declare no conflict of interest.
Informed Consent
Written informed consent was obtained from the patient’s legal representative for publication of this case report and accompanying images.
Author Contributions
All authors contributed to the work reported in this manuscript. Taseal Ahmed, Amy Herai, and Samir Ruxmohan contributed to concept development, study design, definition of intellectual content, literature search, clinical and experimental studies, data acquisition, data analysis, statistical analysis, manuscript preparation, editing, review, and served as the guarantor of the work.
Data Availability
The authors declare that data supporting the findings of this study are available within the article.
| References | ▴Top |
- Kennedy PG, Chaudhuri A. Herpes simplex encephalitis. J Neurol
Neurosurg Psychiatry. 2002;73(3):237-238.
doi pubmed - Ellul M, Solomon T. Acute encephalitis - diagnosis and management.
Clin Med (Lond). 2018;18(2):155-159.
doi pubmed - Zhu S, Viejo-Borbolla A. Pathogenesis and virulence of herpes simplex
virus. Virulence. 2021;12(1):2670-2702.
doi pubmed - Silwal S, Hassan E, Jain S, Rauf I, Obulareddy SJ, Suleman S, Yousuf
FA, et al. A case of herpes simplex virus meningitis in an immunocompromised individual:
avoiding common diagnostic pitfalls. Cureus. 2023;15(7):e42242.
doi pubmed - Abbuehl LS, Branca M, Ungureanu A, Federspiel A, Leib SL, Bassetti
CLA, Hakim A, et al. Magnetic resonance imaging in acute meningoencephalitis of viral and
unknown origin: frequent findings and prognostic potential. Front Neurol.
2024;15:1359437.
doi pubmed - Shirani K, Khorvash F, Naeini AE, Valiyan Boroujeni M, Yazdani M.
Prevalence of herpes simplex virus-1 in hospitalized adult patients with clinical diagnosis of
meningoencephalitis using real-time polymerase chain reaction: a single-center, cross-sectional
study. J Res Med Sci. 2019;24:7.
doi pubmed - Cag Y, Erdem H, Leib S, Defres S, Kaya S, Larsen L, Poljak M, et al.
Managing atypical and typical herpetic central nervous system infections: results of a
multinational study. Clin Microbiol Infect. 2016;22(6):568.e569-568.e517.
doi pubmed - de Jong W, Rusli M, Bhoelan S, Rohde S, Rantam FA, Noeryoto PA, Hadi
U, et al. Endemic and emerging acute virus infections in Indonesia: an overview of the past
decade and implications for the future. Crit Rev Microbiol. 2018;44(4):487-503.
doi pubmed - Masyeni S, Yohan B, Somia IKA, Myint KSA, Sasmono RT. Dengue
infection in international travellers visiting Bali, Indonesia. J Travel Med.
2018;25(1):tay061.
doi pubmed - Riancho J, Delgado-Alvarado M, Sedano MJ, Polo JM, Berciano J. Herpes
simplex encephalitis: clinical presentation, neurological sequelae and new prognostic factors.
Ten years of experience. Neurol Sci. 2013;34(10):1879-1881.
doi pubmed - Duarte LF, Farias MA, Alvarez DM, Bueno SM, Riedel CA, Gonzalez PA.
Herpes simplex virus type 1 infection of the central nervous system: insights into proposed
interrelationships with neurodegenerative disorders. Front Cell Neurosci. 2019;13:46.
doi pubmed - Roberts JI, Jewett GAE, Tellier R, Couillard P, Peters S. Twice
negative PCR in a patient with herpes simplex virus type 1 (HSV-1) encephalitis.
Neurohospitalist. 2021;11(1):66-70.
doi pubmed - Rajmohan R, Khoury D, Perez-Rosendahl M, Mnatsakanyan L, Groysman L.
Polymerase chain reaction (PCR)-negative herpes simplex virus (HSV) encephalitis in a
62-year-old woman with p-ANCA vasculitis. Cureus. 2022;14(1):e21480.
doi pubmed - Niksefat M, Guillen D, Moshayedi P, Rinaldo CR, Ojha A. Third time's
a charm: diagnosis of herpes simplex encephalitis after two negative polymerase chain reaction
results. Heliyon. 2020;6(6):e04247.
doi pubmed - Wilson MR, Sample HA, Zorn KC, Arevalo S, Yu G, Neuhaus J, Federman
S, et al. Clinical Metagenomic Sequencing for Diagnosis of Meningitis and Encephalitis.
N Engl J Med. 2019;380(24):2327-2340.
doi pubmed - Chiu CY, Miller SA. Clinical metagenomics. Nat Rev Genet.
2019;20(6):341-355.
doi pubmed - Hassan KM yile, Badi H, Lahrichi H, Fadel OE, Marih L, Sodqi M, et al. Herpes simplex virus-1 herpetic meningoencephalitis in adults: three cases in Casablanca, Morocco. Ethiopian Medical Journal. 2023;62(2):127-132.
- Hjalmarsson A, Blomqvist P, Skoldenberg B. Herpes simplex
encephalitis in Sweden, 1990-2001: incidence, morbidity, and mortality. Clin Infect Dis.
2007;45(7):875-880.
doi pubmed - Domingues RB, Fink MC, Tsanaclis AM, de Castro CC, Cerri GG, Mayo MS,
Lakeman FD. Diagnosis of herpes simplex encephalitis by magnetic resonance imaging and
polymerase chain reaction assay of cerebrospinal fluid. J Neurol Sci.
1998;157(2):148-153.
doi pubmed - Zainah H. A unique case of HSV encephalitis with atypical mri findings and negative HSV Pcr. Journal of Hospital Medicine. [Internet] [cited July 21, 2025]; Available from: https://shmabstracts.mystagingwebsite.com/abstract/a-unique-case-of-hsv-encephalitis-with-atypical-mri-findings-and-negative-hsv-pcr/.
- Renard D, Nerrant E, Lechiche C. DWI and FLAIR imaging in herpes
simplex encephalitis: a comparative and topographical analysis. J Neurol.
2015;262(9):2101-2105.
doi pubmed - Fragoso DC, Goncalves Filho AL, Pacheco FT, Barros BR, Aguiar Littig
I, Nunes RH, Maia Junior AC, et al. Imaging of Creutzfeldt-Jakob Disease: Imaging Patterns and
Their Differential Diagnosis. Radiographics. 2017;37(1):234-257.
doi pubmed - Pai V, Sitoh YY, Purohit B. Gyriform restricted diffusion in adults:
looking beyond thrombo-occlusions. Insights Imaging. 2020;11(1):20.
doi pubmed - de Oliveira AM, Paulino MV, Vieira APF, McKinney AM, da Rocha AJ, Dos
Santos GT, Leite CDC, et al. Imaging Patterns of Toxic and Metabolic Brain Disorders.
Radiographics. 2019;39(6):1672-1695.
doi pubmed - Bakharji A, Alarfaj H, Omar S, Alzahrani S, Alzahrani N, Alahmari F,
Al Mulhim S, et al. Brain magnetic resonance imaging findings in hyperammonemic hepatic
encephalopathy: A case series and literature review. Biomed Rep. 2025;22(6):98.
doi pubmed - Taguchi S, Nakura T, Doyu M, Saiki H. Cortical Ribboning as a Key MRI
Finding in Wernicke's Encephalopathy With Altered Mental Status. Cureus.
2025;17(2):e79279.
doi pubmed - Huang TH, Lai MC, Chen YS, Huang CW. Brain imaging in epilepsy-focus
on diffusion-weighted imaging. Diagnostics (Basel). 2022;12(11):2602.
doi pubmed - Miyamoto T, Sogabe S, Korai M, Yamaguchi I, Ishihara M, Shimada K, et al. The significance and characteristics of the patients with Ribbon-like signal hyperintensity after acute ischemic stroke [Internet]. 2023 [cited Apr 21, 2025];2023.08.21.23294396. Available from: https://www.medrxiv.org/content/10.1101/2023.08.21.23294396v1.
- DeSena AD, Noland DK, Matevosyan K, King K, Phillips L, Qureshi SS,
Greenberg BM, et al. Intravenous methylprednisolone versus therapeutic plasma exchange for
treatment of anti-N-methyl-D-aspartate receptor antibody encephalitis: A retrospective review.
J Clin Apher. 2015;30(4):212-216.
doi pubmed - Moser T, Harutyunyan G, Karamyan A, Otto F, Bacher C, Chroust V,
Leitinger M, et al. Therapeutic Plasma Exchange in Multiple Sclerosis and Autoimmune
Encephalitis: a Comparative Study of Indication, Efficacy and Safety. Brain Sci.
2019;9(10):267.
doi pubmed - Safain MG, Roguski M, Kryzanski JT, Weller SJ. A review of the
combined medical and surgical management in patients with herpes simplex encephalitis. Clin
Neurol Neurosurg. 2015;128:10-16.
doi pubmed - Kiefer L, Adam D, Mudugal D, Burnett MS. Viral meningitis mimicking benign intracranial hypertension: a report of two cases. Interdisciplinary Neurosurgery. 2020;20:100646.
- Baten A, Melo-Bicchi M, Gutierrez C. T69. Continuous Electroencephalogram (CEEG) as a biomarker for progression of Herpes Simplex Virus Encephalitis (HSE). Clinical Neurophysiology. 2018;129:e28.
This
article is distributed under the terms of the Creative Commons Attribution Non-Commercial 4.0
International License, which permits unrestricted non-commercial use, distribution, and
reproduction in any medium, provided the original work is properly cited.
Journal
of Neurology Research is published by Elmer Press Inc.