| Journal of Neurology Research, ISSN 1923-2845 print, 1923-2853 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Neurol Res and Elmer Press Inc |
| Journal website https://jnr.elmerpub.com |
Case Report
Volume 16, Number 1, March 2026, pages 40-44
New-Onset Human Immunodeficiency Virus Presenting as Varicella Zoster Reactivation With Central and Peripheral Nervous System Manifestations: Report of Two Cases
Anu Khannaa, b, Catherine Boldiga, Kathleen Murraya
aDepartment of Neurology, University of South Florida, Tampa, FL, USA
bCorresponding Author: Anu Khanna, Department of Neurology, University of South Florida, Tampa, FL, USA
Manuscript submitted October 31, 2025, accepted January 17, 2026, published online March 13, 2026
Short title: New HIV Presenting as Zoster Neuroinvolvement
doi: https://doi.org/10.14740/jnr1068
| Abstract | ▴Top |
Human immunodeficiency virus (HIV) affects approximately 1.2 million individuals in the United States, with immunosuppression from impaired T-cell-mediate immunity predisposing patients to a range of opportunistic infections. While varicella-zoster virus (VZV) is a known opportunistic pathogen in this population, its presentation as isolated neurological syndromes in newly diagnosed HIV patients remains uncommon and underrecognized. This case series highlights two rare and diagnostically challenging presentations of VZV-related neurological complications that led to the diagnosis of HIV. The first case involves a 30-year-old male presenting with progressive lower extremity weakness, constipation, and urinary incontinence over 10 days. Clinical, radiographic, and serological findings were consistent with VZV meningomyelitis. The second case features a 46-year-old male with acute onset back pain and unilateral leg weakness in the setting of active VZV vesicular lesions. Radiographic findings were consistent with VZV radiculitis. These cases underscore the importance of considering HIV testing in patients with atypical or severe presentations of VZV neurological involvement. This report emphasizes the diagnostic and clinical significance of recognizing such manifestations, which may serve as sentinel events for underlying immunodeficiency.
Keywords: Varicella-zoster virus; Human immunodeficiency virus; Meningomyelitis; Radiculitis
| Introduction | ▴Top |
Human immunodeficiency virus (HIV) prevention and diagnosis remain a large public health focus. The Center for Disease Control (CDC) currently estimates that in 2022, there were 31,800 new HIV infections with an estimated 1.2 million people having HIV in the United States [1]. HIV infection has multiple neurological manifestations depending on the duration of infection. On initial infection, HIV RNA is detectable in cerebrospinal fluid (CSF) as early as 8 days after exposure with resulting central nervous system (CNS) inflammation [2]. Initially, patients may develop acute retroviral syndrome (ARS), which is more typically a “flu-like” illness with lymphadenopathy, fever, fatigue, and malaise [3]. However, in about 10% of patients with ARS, there will be other neurological symptoms such as aseptic meningitis, encephalitis, and facial nerve palsy [4]. The most common chronic HIV neurological manifestations are HIV-associated neurocognitive disorders and distal symmetric polyneuropathy [5].
HIV due to its effects on T-cell-mediated immune reactions, increases the risk for opportunistic infections. In regard to invading the CNS, opportunistic infections may include cryptococcus, toxoplasmosis, cytomegalovirus (CMV), varicella-zoster virus (VZV), herpes simplex (HSV), tuberculosis, John Cunningham virus (JC virus), etc. [6]. The severity and type of infection depend on level of immunosuppression (most easily represented by CD4 count) [7]. Patients with HIV can often have VZV positive antibody in the CSF, in a form of subclinical reactivation without symptoms [8]. However, these levels have been found to remain stable; instead, low CD4 count and lack of antiretroviral therapy are more predictive of the development of zoster [9].
Varicella infects 90–100% of the population in temperate regions [10]. After initial infection, VZV becomes latent primarily in the neurons of the human ganglia (dorsal root ganglia, trigeminal ganglia, and autonomic ganglia) with only about 10–20% of the population experiencing reactivation later in life [10, 11]. Similar to immunocompetent hosts, most patients with HIV will initially develop pain followed by a vesicular rash from VZV reactivation. However, about 35% of immunocompromised patients develop further complications [10] such as encephalitis, vasculitis, and meningomyelitis [12–14]. VZV can also present as Elsberg syndrome. Elsberg syndrome is a neuroinflammatory disorder preceded by a viral infection such as VZV, HSV, and COVID that results in a cauda equina-like syndrome with saddle anesthesia, bladder, and bowel incontinence due to lumbosacral radiculitis [15]. Generally, the course is self-limited [16]. In this case series, we present two patients with VZV reactivation who were newly diagnosed with HIV.
| Case Reports | ▴Top |
The first patient was a 30-year-old male with no known medical history who presented to the hospital for lower extremity weakness. He reported 12 days prior, he woke up and noticed that his left leg felt numb from the knee to the ankle, which progressed to his right leg 2 days later. The weakness progressed, contributed to falls, and he was unable to move the leg anymore. He also reported severe pain when something touched the bottom of his right foot. He describes severe urinary urgency with voiding attempts every 10 min and intermittent urinary incontinence. He reports no bowel movement for 3 weeks and endorses numbness involving the groin and buttocks. He denied numbness or weakness in his arms, headache, vision changes, or speech changes.
On examination, he was alert and oriented to person, place, time, and situation. His right pupil was 2 mm and left pupil was 5 mm. Cranial nerves were otherwise intact. In terms of motor, he had decreased tone in his lower extremities. The muscle bulk appeared normal. He had full strength in his upper extremities and his left lower extremity. He had 0/5 strength throughout his right lower extremity. Reflexes were 3+ symmetrically at biceps, brachioradialis, triceps, patellar, and Achilles. Hoffman was negative bilaterally. Plantar reflex on the left was flexor (Babinski negative). The patient refused testing of the right plantar reflex due to pain. Coordination: finger-to-nose was intact bilaterally. Sensation: light touch, temperature, vibration, and proprioception were intact in upper extremities. Sensation was absent distal to the T4 dermatome (nipple line). Gait: Unable to test due to significant lower extremity weakness.
In the emergency room, labs were drawn and pertinent labs are listed in Table 1. Magnetic resonance imaging (MRI) of the entire spine demonstrated abnormal T2 signal abnormality extending throughout the thoracic spinal cord and ascending to the C7 level of the cervical spinal cord. Lumbar spine MRI additionally revealed enhancement of the cauda equina nerve roots. Brain MRI was unremarkable (negative). Lumbar puncture was performed and the results are listed in Table 2. VZV PCR was positive in both the CSF and serum. VZV IgG in the serum was positive while IgM was negative. He was started on intravenous (IV) ganciclovir (due to a shortage of IV acyclovir) and combined antiretroviral therapy (cART). He received IV ganciclovir for 10 days and then transitioned to oral valacyclovir prior to discharge to rehab. Since discharge from rehab, he has been inconsistently taking his valacyclovir and cART and has continued to experience pain and weakness in his lower extremities. On repeat MRI brain and total spine 2 months later, no abnormal enhancement was seen.
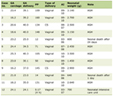 Click to view | Table 1. Pertinent Serum Studies From Cases 1 and 2 |
 Click to view | Table 2. Pertinent CSF Findings From Case 1 With Reference Ranges |
The second patient was a 46-year-old male with no known medical history, presenting to the emergency room with lower back pain. He reported that 2 weeks prior to presentation, he woke up with low back pain radiating down the left posterior leg. The pain was progressive since onset and exacerbated at night and by movement. A couple of days later, he noted left lower extremity weakness and paresthesias/numbness. The weakness predominantly involved the left hip and knee. The numbness was localized to the left leg between the knee and ankle. Additionally, he developed a vesicular rash (Fig. 1) in the L4 dermatome. He also reported intermittent headache which resolved with resting. He denied neck stiffness, vision changes.
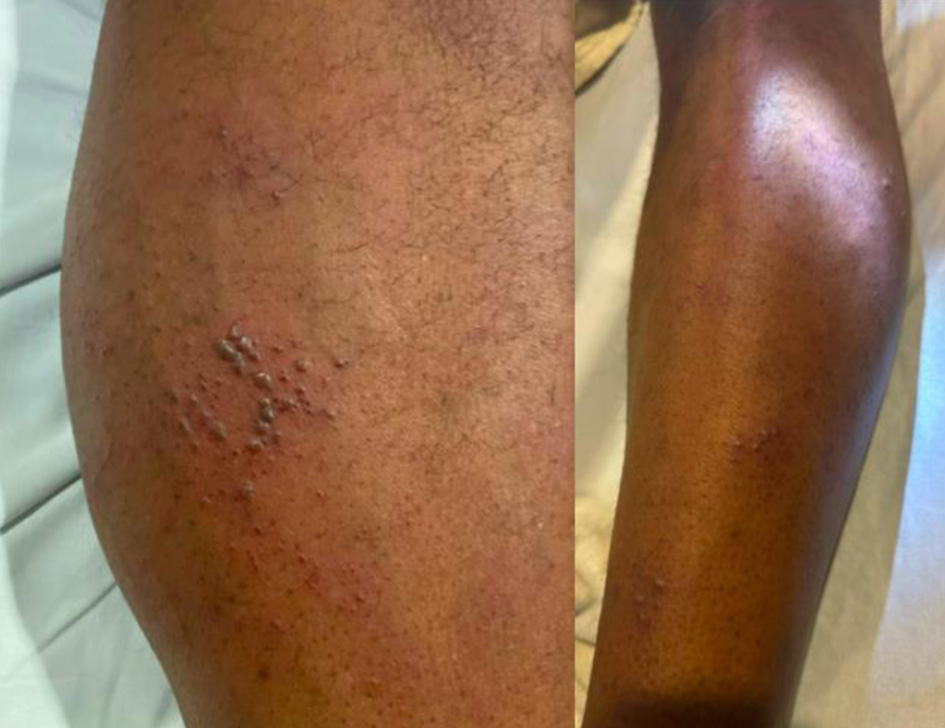 Click for large image | Figure 1. Vesicular rash of case 2 extending from above the knee downwards in an L4 dermatome distribution. |
On exam, he was alert and oriented to person, place, time, and situation. Cranial nerves were intact. Tone and bulk were normal in the upper and lower extremities. He had full strength in the upper extremities and right leg and 4−/5 with hip flexion, 4+/5 with knee flexion, 4/5 with knee extension, 4/5 with foot dorsiflexion, and 5/5 with foot plantarflexion in the left leg. His reflexes were 2+ at the biceps, brachioradialis, triceps, and right patellar and Achilles. His reflexes in the left leg were 0 at the patella and 2+ at the Achilles. Plantar reflexes were flexor (Babinski signs absent), bilaterally. Finger-to-nose was intact bilaterally. Sensation was intact to light touch, temperature, and proprioception in the upper extremities and right lower extremity. He had decreased sensation to light touch and temperature in the left lower extremity, distal to the knee and proximal to the ankle. His gait was normal.
Labs are listed in Table 1. Serum VZV was not tested but vesicle scraping from his lesions was positive for VZV. He underwent MRI of his cervical, thoracic, and lumbar spine which showed enhancing nerve roots in the cauda equina (Fig. 2). Lumbar puncture was recommended to patient; however, he refused. IV acyclovir was administered for 14 days, after which the regimen was converted to oral valacyclovir. He was also started on cART. He has not been seen for follow-up.
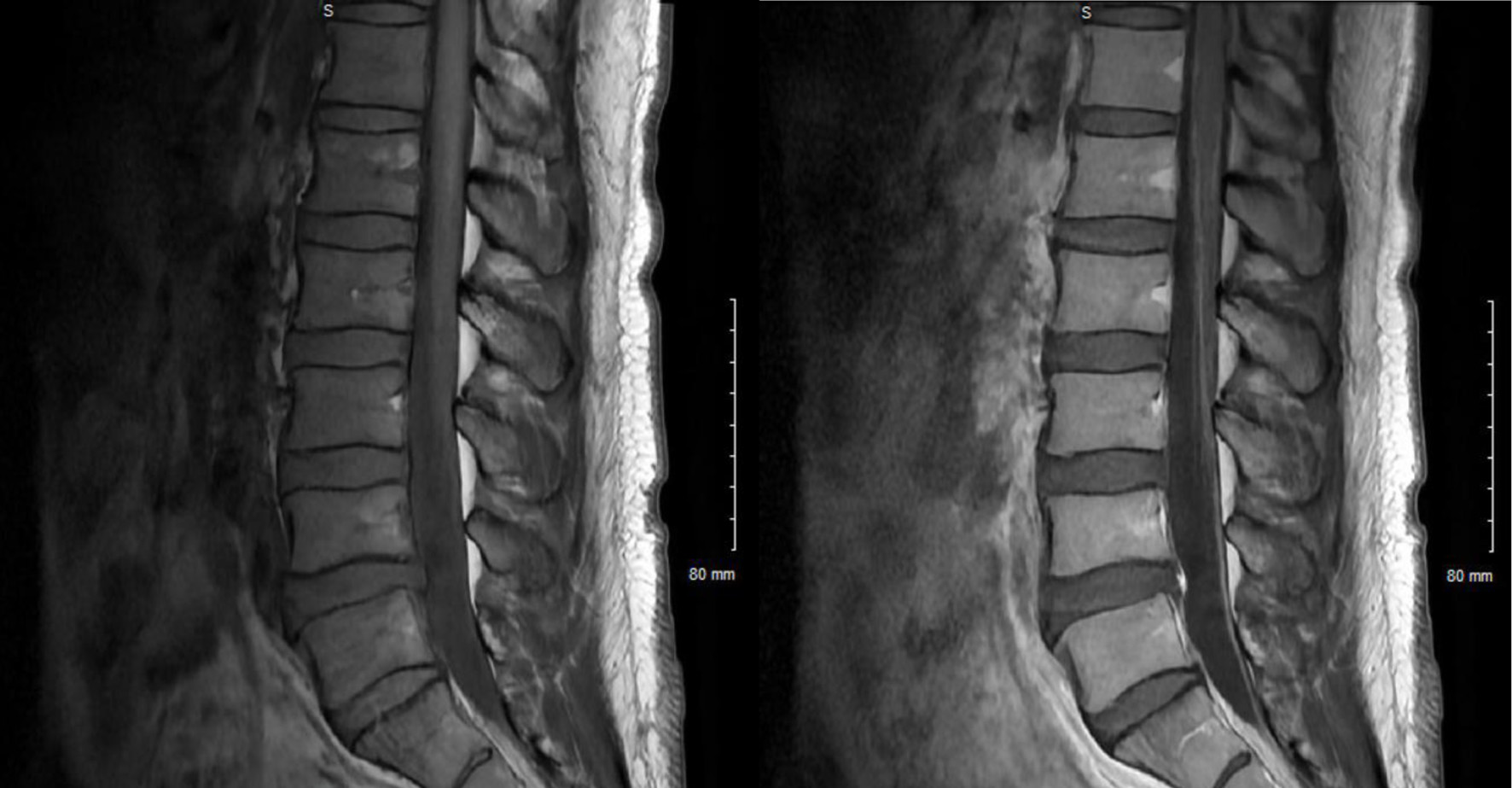 Click for large image | Figure 2. Pre-contrast T1-weighted sagittal images of the L-spine (left) and post-contrast (right) with enhancement of cauda equina nerve roots. |
| Discussion | ▴Top |
VZV is a herpesviridae that can cause a variety of complications including neurological complications due to invasion of the neuroaxis. Patients are more likely to experience CNS involvement if they are immunocompromised, as in the case with HIV. Our patients, with previously unknown HIV, developed meningomyelitis and radiculitis. Myelitis is characterized by inflammation of the spinal cord, and radiculitis is characterized by inflammation of the nerve roots. MRI with contrast of the affected region typically shows hyperintensity on T2-weighted sequences and post contrast enhancement on T1-weighted sequences [17].
While both cases have similar diagnoses and treatments, these patients had remarkably different presentations. In case 1, the patient presented with symptoms of cauda equina with saddle anesthesia, bladder/bowel incontinence, and lower extremity numbness/weakness. With the MRI findings of extensive spinal cord involvement, this case presentation is most consistent with VZV meningomyelitis.
While this patient presentation has several overlapping features of Elsberg syndrome (a neuroinflammatory reaction resulting in bilateral lumbosacral radiculitis with or without myelitis), the extensive spinal cord findings would be more aptly described as VZV meningomyelitis [15]. In one retrospective review discussing MRI findings in patients with Elsberg syndrome, about 60% of patients had lower cord T2 hyperintense abnormalities and 43% had cord gadolinium enhancement [15]. However, only 20% of patients had MRI findings that expanded rostrally to T9–T10, which is a red flag per proposed clinical criteria [15]. Our patient had cord enhancement from the thoracic spine to C7 with enhancement of the cauda equina roots. This aligns with other case reports of VZV reactivation in immunocompromised individuals, as it more typically presents with widespread involvement of the spinal cord and brain compared to HSV-induced Elsberg syndrome including encephalitis, polyneuropathy, and ventriculitis [15, 18]. While this patient was only treated with IV ganciclovir (in the setting of an acyclovir shortage), corticosteroids have also been used for treatment but data are limited [17].
In contrast to the first case, the second case demonstrates a distinctly different presentation of VZV-associated radiculitis. This patient exhibited active VZV skin lesions along with unilateral lower-leg symptoms. Because his involvement was limited to radiculitis—supported by MRI findings showing enhancement of the cauda equina roots—his examination was notable for hyporeflexia. As a result, the discussion about potential treatment is different. While the data are limited in this setting, one article recommended treating VZV radiculitis without obvious meningitis in immunocompetent hosts with only oral acyclovir [19]. One prospective observational cohort study of the Danish Group of Infections of the Brain database showed that in VZV meningitis regardless of immunocompetence early administration of antiviral treatment was not associated with improved outcomes 30 days after discharge [20]. Steroids were generally not indicated.
Regarding the concomitant infections of both VZV and HIV, in both case 1 and case 2, patients were newly diagnosed with HIV via screening laboratory tests completed in the emergency department. There are a few prior case reports that discuss VZV reactivation—Elsberg syndrome and disseminated zoster—as an initial presentation of HIV [16, 21]. However, most of the current literature documents VZV reactivation in HIV patients before widespread ART or of chronic untreated HIV+ individuals. This report emphasizes the diagnostic and clinical significance of recognizing such manifestations, which may serve as sentinel events for underlying immunodeficiency.
Acknowledgments
The authors have no acknowledgments.
Financial Disclosure
This research was unfunded.
Conflict of Interest
There are no financial disclosures or conflict of interest.
Informed Consent
Informed consent was obtained from all patients in compliance with the Declaration of Helsinki.
Author Contributions
AK performed the literature review and wrote the report. CB conceptualized and wrote the report. KM conceptualized, supervised, and critically reviewed the report.
Data Availability
The authors declare that data supporting the findings of this study are available within the article.
Abbreviations
ARS: acute retroviral syndrome; cART: combined antiretroviral therapy; CSF: cerebrospinal fluid; HIV: human immunodeficiency virus; HSV: herpes simplex virus; VZV: varicella-zoster virus
| References | ▴Top |
- CDC. Fast facts: HIV in the United States. HIV. July 1, 2024. Accessed April 14, 2025. https://www.cdc.gov/hiv/data-research/facts-stats/index.html.
- Valcour V, Chalermchai T, Sailasuta N, Marovich M, Lerdlum S, Suttichom D, Suwanwela NC, et al. Central nervous system viral invasion and inflammation during acute HIV infection. J Infect Dis. 2012;206(2):275-282.
doi pubmed - Braun DL, Kouyos RD, Balmer B, Grube C, Weber R, Gunthard HF. Frequency and spectrum of unexpected clinical manifestations of primary HIV-1 infection. Clin Infect Dis. 2015;61(6):1013-1021.
doi pubmed - Holroyd KB, Vishnevetsky A, Srinivasan M, Saylor D. Neurologic complications of acute HIV infection. Curr Treat Options Infect Dis. 2020;12(3):227-242.
doi pubmed - Kolson D. Neurologic complications in persons With HIV infection in the era of antiretroviral therapy. Top Antivir Med. 2017;25(3):97-101.
pubmed - Tan IL, Smith BR, von Geldern G, Mateen FJ, McArthur JC. HIV-associated opportunistic infections of the CNS. Lancet Neurol. 2012;11(7):605-617.
doi pubmed - Bowen LN, Smith B, Reich D, Quezado M, Nath A. HIV-associated opportunistic CNS infections: pathophysiology, diagnosis and treatment. Nat Rev Neurol. 2016;12(11):662-674.
doi pubmed - Birlea M, Arendt G, Orhan E, Schmid DS, Bellini WJ, Schmidt C, Gilden D, et al. Subclinical reactivation of varicella zoster virus in all stages of HIV infection. J Neurol Sci. 2011;304(1-2):22-24.
doi pubmed - Pomerantz HS, Xu X, White J, Sunil TS, Deiss RG, Ganesan A, Agan BK, et al. Association between quantitative varicella-zoster virus antibody levels and zoster reactivation in HIV-infected persons. AIDS Res Ther. 2018;15(1):25.
doi pubmed - De La Blanchardiere A, Rozenberg F, Caumes E, Picard O, Lionnet F, Livartowski J, Coste J, et al. Neurological complications of varicella-zoster virus infection in adults with human immunodeficiency virus infection. Scand J Infect Dis. 2000;32(3):263-269.
doi pubmed - Kennedy PGE, Gershon AA. Clinical features of varicella-zoster virus infection. Viruses. 2018;10(11):609.
doi pubmed - Marais G, Naidoo M, McMullen K, Stanley A, Bryer A, van der Westhuizen D, Bateman K, et al. Varicella-zoster virus reactivation is frequently detected in HIV-infected individuals presenting with stroke. J Med Virol. 2022;94(6):2675-2683.
doi pubmed - Gutierrez J, Ortiz G. HIV/AIDS patients with HIV vasculopathy and VZV vasculitis: a case series. Clin Neuroradiol. 2011;21(3):145-151.
doi pubmed - Steiner I, Kennedy PG, Pachner AR. The neurotropic herpes viruses: herpes simplex and varicella-zoster. Lancet Neurol. 2007;6(11):1015-1028.
doi pubmed - Savoldi F, Kaufmann TJ, Flanagan EP, Toledano M, Weinshenker BG. Elsberg syndrome: A rarely recognized cause of cauda equina syndrome and lower thoracic myelitis. Neurol Neuroimmunol Neuroinflamm. 2017;4(4):e355.
doi pubmed - Santos DH, Carneiro de Oliveira RM, Junior WRF, Olivetti BC. Myelorradiculitis due to Varicella Zoster (Elsberg syndrome) as the first symptom of HIV in a patient with Crohn's disease in use of Infliximab. Mult Scler Relat Disord. 2021;47:102643.
doi pubmed - Lameiras C, Patrocinio de Jesus R, Flor-de-Lima B, Silva J, Pacheco P. A case of varicella-zoster virus meningomyelitis in an HIV-1-infected patient: facing the challenges related to its management and prognosis. Cureus. 2022;14(8):e27652.
doi pubmed - Hung CH, Chang KH, Kuo HC, Huang CC, Liao MF, Tsai YT, Ro LS. Features of varicella zoster virus myelitis and dependence on immune status. J Neurol Sci. 2012;318(1-2):19-24.
doi pubmed - Luisier V, Weber L, Fishman D, Praz G, Ghika JA, Genoud D, Chabwine JN. Definition and management of varicella zoster virus-associated meningoradiculitis: a case report. BMC Res Notes. 2016;9(1):451.
doi pubmed - Petersen PT, Bodilsen J, Jepsen MPG, Larsen L, Storgaard M, Hansen BR, Helweg-Larsen J, et al. Clinical features and prognostic factors in adults with viral meningitis. Brain. 2023;146(9):3816-3825.
doi pubmed - Zeng L, Feng S, Yao L, Zhao J, Zhang G. Disseminated herpes zoster with decreased CD4 counts in a HIV-infected patient. Clin Cosmet Investig Dermatol. 2023;16:3165-3170.
doi pubmed
This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (CC BY 4.0), which permits unrestricted use, distribution, and reproduction in any medium, including commercial use, provided the original work is properly cited.
Journal of Neurology Research is published by Elmer Press Inc.