| Journal of Neurology Research, ISSN 1923-2845 print, 1923-2853 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Neurol Res and Elmer Press Inc |
| Journal website https://jnr.elmerpub.com |
Original Article
Volume 16, Number 1, March 2026, pages 22-29
Utilization of Stroke Kit Antihypertensives Prior to Thrombolysis in Acute Ischemic Stroke: A Single-Center Retrospective Audit
Delan Pelendage-Pereraa, b, Iouri Banakha, Samuel Shepherda
aMonash Health Pharmacy Department, Clayton, Victoria 3168, Australia
bCorresponding Author: Delan Pelendage-Perera, Monash Health Pharmacy Department, Clayton, Victoria 3168, Australia
Manuscript submitted November 23, 2025, accepted February 23, 2026, published online March 13, 2026
Short title: Utilization of Stroke Kit Antihypertensives
doi: https://doi.org/10.14740/jnr1076
| Abstract | ▴Top |
Background: Current hospital guideline recommends intravenous (IV) hydralazine to reduce blood pressure (BP) to < 185/110 mm Hg in acute ischemic stroke (AIS) patients receiving thrombolysis, with verapamil and metoprolol as alternatives. Emergency department (ED) stroke kits (SK) contain alteplase and these IV antihypertensives. Tenecteplase is a second-line thrombolytic kept separately along with IV labetalol in the resuscitation medication room (RMR). However, other hospitals in Victoria recommend hydralazine, labetalol, and nicardipine for AIS. This study was carried out to determine the frequency of antihypertensives used from the ED SK versus the RMR at a tertiary hospital and to assess the time to reach target BP for thrombolysis.
Methods: A retrospective audit was conducted between 2021 and 2024. The electronic medical records were utilized to identify eligible participants. Inclusion criteria included adult ED hypertensive patients administered either IV hydralazine or labetalol who presented with AIS and indicated for thrombolysis. Patients subsequently administered IV metoprolol or verapamil following hydralazine or labetalol administration were also included.
Results: Out of 349 screened patients, six met the inclusion criteria. Four were treated with alteplase and two with tenecteplase for thrombolysis. One patient (25%) received hydralazine from the SK, the remainder from the RMR. The median time to reach target BP after hydralazine administration was 4.5 min (range: 2–18). No patients received labetalol, metoprolol or verapamil for AIS pre-thrombolysis.
Conclusions: Antihypertensives from the SK were infrequently used, warranting a review of SK contents. Due to a small sample size, the optimal antihypertensive for thrombolysis in AIS could not be determined, suggesting that further research is required.
Keywords: Utilization; Stroke kit; Antihypertensive; Thrombolysis; Acute ischemic stroke; Stroke; Hydralazine; Labetalol
| Introduction | ▴Top |
Stroke is a life-threatening condition and one of the leading causes of death and disability in Australia [1]. Stroke can be categorized as either acute ischemic stroke (AIS) or hemorrhagic stroke (HS). AIS is a blockage in an artery, reducing blood flow to the brain leading to brain tissue necrosis and death [2]. In 2023, approximately 45,785 stroke-related events were recorded in Australia, with approximately 87% being diagnosed as AIS [3]. Both AIS and HS present with similar symptoms, requiring immediate distinction to ensure appropriate therapy. The initial treatment of AIS predominantly requires intravenous (IV) thrombolytic therapy with endovascular clot retrieval [4].
Prompt administration of IV tissue-type plasminogen activator (tPA), either alteplase or tenecteplase, is required for AIS management [5]. This therapy dissolves the clot responsible for cerebral ischemia, with the most beneficial outcome if administered within 4.5–9 h of symptom onset [6]. The National Institutes of Health Stroke Scale (NIHSS) is a tool that quantifies stroke severity as minor (0–4), moderate (5–15), moderate to severe (16–20) or severe (21–42) [7]. This score assists in determining the effectiveness of tPA post-thrombolysis [7]. While evidence supports tenecteplase non-inferiority, alteplase remains the agent of choice at this hospital [8]. Although tPA is effective in the management of AIS, the risk of intracranial hemorrhage remains high, particularly in the setting of uncontrolled blood pressure (BP). As such, tPA is contraindicated in patients with systolic blood pressure (SBP) exceeding 185 mm Hg and diastolic blood pressure (DBP) over 110 mm Hg [9]. To enable timely administration of tPA, it is crucial to use a fast-acting and effective IV antihypertensive to rapidly achieve the target BP of less than 185 mm Hg SBP and 110 mm Hg DBP, thereby minimizing disabling stroke complications [10]. Moreover, continuous monitoring of BP is essential in maintaining sufficient BP control. The local hospital guideline strives to monitor BP every 15 min for the first 2 h post-administration of tPA, then every 30 min for the next 4 h and then every hour for the next 18 h thereafter to reduce bleeding risk post-tPA [11].
Stroke kits (SK) have been implemented in the emergency department (ED) of the tertiary hospital to facilitate rapid access to time-sensitive medications, such as IV alteplase and antihypertensive, by being conveniently located in a sealed kit inside the resuscitation medication room (RMR). Currently, the SK contain alteplase and several IV antihypertensives: hydralazine, metoprolol, and verapamil. SK are pre-assembled by pharmacy technicians and are intended to streamline acute stroke management. No studies have explored the benefits of SK in reducing the time to administer antihypertensives and tPA. Anecdotally, clinicians may preferentially source antihypertensives and tPA from the ED RMR rather than the SK, raising questions about the utility and resource allocation associated with maintaining these kits. Furthermore, the antihypertensives inside the SK may not reflect contemporary clinical guideline recommendations [12]. At present, hydralazine is the first-line antihypertensive at this hospital, with metoprolol and verapamil as alternatives. There is a lack of consensus from Australian clinical guidelines on the preferred IV antihypertensive in AIS [13]. International guidelines recommend IV labetalol and nicardipine as preferred antihypertensives in AIS, with hydralazine being considered an acceptable substitute [12]. Hydralazine is not considered the preferred agent due to its variable onset and duration of action [14]. Labetalol and nicardipine are favored for their predictable pharmacodynamics and titratability, which are critical in acute stroke settings [15, 16]. Nonetheless, limited evidence exists comparing the effectiveness of these agents in reducing BP and minimizing delays to thrombolysis. Nicardipine, though widely adopted internationally and increasingly used in Melbourne, remains restricted in Australia under the Special Access Scheme (SAS) [17, 18].
This study assessed the frequency of antihypertensives retrieved from the SK versus RMR and the time taken to achieve the target BP for thrombolysis.
| Materials and Methods | ▴Top |
Study design
This retrospective observation cohort study reviewed the use of SK between January 2021 and December 2024. It was carried out at a single site within a large, multi-site, tertiary hospital network. The site’s ED comprises approximately 63 treatment rooms and 75,000 presentations annually [19]. Patients included in the study were adults aged 18 years or older who were diagnosed with AIS and received IV labetalol or hydralazine in ED as antihypertensive treatment pre-thrombolysis. Patients subsequently given IV metoprolol or verapamil were also included. Patients were excluded if they were treated outside the ED, those with AIS not requiring thrombolysis, and those who were administered other IV or oral antihypertensives as part of their emergency treatment.
Data collection
Eligible patients were identified from the electronic medical records (EMR) by generating a medication administration report for those administered either IV labetalol or IV hydralazine in ED during the study period. Patients subsequently administered IV metoprolol and IV verapamil after administration of hydralazine or labetalol were also included to determine if alternative therapies were given as second-line treatment. All patients were individually screened to determine AIS presentation. The following data were collected for included patients: demographics, past medical history, antihypertensives given pre-thrombolysis in ED, total dose of antihypertensives given prior to thrombolysis, tPA agent given, baseline BP, BP after antihypertensives administered (the closest BP times were collected post-administration), time to reach target BP for thrombolysis to occur, location of antihypertensive retrieved, and baseline and post-thrombolysis NIHSS score (Supplementary Material 1, jnr.elmerpub.com).
Dispensing transaction records from the hospital dispensing software (Merlin®) were investigated to determine the total number of SK distributed during the time period and when the antihypertensives were restocked into the SK. These dates were cross-matched with the date the SK were used to determine if the antihypertensive was used from the SK versus the RMR.
The primary outcome of this study was the frequency of antihypertensives utilized from the SK, compared to those from the ED RMR. Secondary outcomes included the time taken to achieve target BP for thrombolysis following antihypertensive administration, in comparison with published data on nicardipine, and the change in NIHSS score following thrombolysis.
Data analysis
The results were analyzed via SPSS version 30 (IBM Corp., Armonk, NY, USA) and represented as frequency tables.
Ethics approval
This study received an exemption from the hospital’s Human and Research Ethics Committee (Reference number: RES-24-0000-009Q).
| Results | ▴Top |
Over the study period, 349 patients received either IV labetalol or IV hydralazine in the ED. There were 43 (12.3%) patients who received IV labetalol and 306 (87.7%) patients who received IV hydralazine. All 43 patients administered labetalol had non-AIS diagnoses, including 15 (34.9%) with aortic dissection, 14 (32.9%) with HS, and six (14.0%) with preeclampsia. Of the 306 patients that were administered hydralazine, 300 patients were excluded (Fig. 1). From those excluded, 186 (60.8%) presented with HS, 37 (12.1%) presented with hypertensive emergency, and 16 (5.2%) had insufficient documentation. No patients were given subsequent doses of IV metoprolol or IV verapamil as alternative therapy post-administration of hydralazine. A total of six patients were included in the final audit. Table 1 shows the patient demographics.
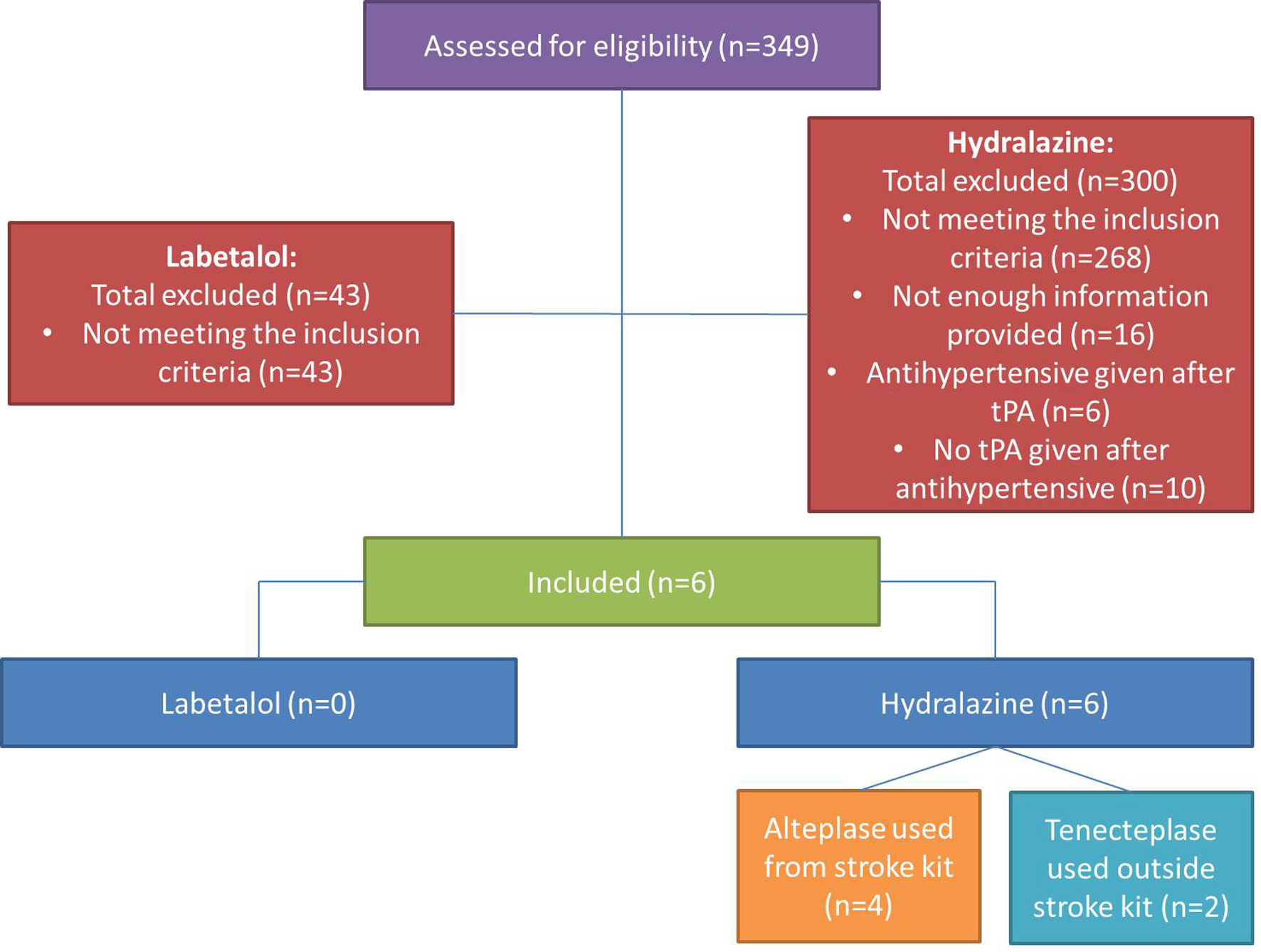 Click for large image | Figure 1. Flow chart representing included participants. |
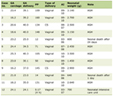 Click to view | Table 1. Patient Demographics (n = 6) |
Of the six patients that received hydralazine, four (66.7%) received alteplase and the remaining two (33.3%) received tenecteplase for thrombolysis. As tenecteplase was not prepacked into the SK, these patients (n = 2) were excluded when determining the location of hydralazine retrieval, but were only included in hydralazine BP lowering analysis. From the four patients that received alteplase, one (25%) patient had hydralazine given from the SK and the remaining three (75%) had their hydralazine retrieved from the RMR. During the study period, 223 SK were supplied to the ED, with hydralazine being used for one patient (0.4%).
Effect of hydralazine on BP
The median baseline BP was 183.5/101 mm Hg (range: 180/79–208/131), which decreased to 164.5/90 mm Hg (range: 135/77–178/109) after the first hydralazine dose. The mean dose was 4.58 mg (standard deviation (SD) = 1.02), and the median time to reach target BP was 4.5 min (range: 2–18). SBP decreased from a median of 183.5 to 164.5 mm Hg, and further to a lowest post-administration median of 148 mm Hg. In contrast, DBP showed a reduction from 101 to 90 mm Hg, and to a lowest median of 88.5 mm Hg (Table 2).
 Click to view | Table 2. Hydralazine Blood Pressure Lowering Effect and NIHSS Score |
The time to hydralazine administration and door-to-needle time for thrombolytics were shorter with the use of SK compared to RMR (Table 3). However, the time to administration of thrombolytic after hydralazine did vary significantly.
 Click to view | Table 3. Hydralazine Administration Time and Door to Needle Time |
The mean baseline NIHSS score was 7.75 (n = 4, SD = 5.62), which reduced to a mean score of 3.25 (n = 4, SD = 3.40) post-thrombolysis; however, documentation of NIHSS scores was incomplete in some patients (Table 2).
| Discussion | ▴Top |
The findings of this study revealed that there was an infrequent use of antihypertensives from the SK, with the majority of the antihypertensives being retrieved from the ED RMR. During the study period, 223 SK were supplied to the ED, with hydralazine being obtained from the kit for one patient. This demonstrates that the kits are not being used for the antihypertensive contents, but rather for the alteplase; hence, the contents within the SK may need to be reviewed. Australia reported to have approximately 45,000 stroke events in 2023, with 87% of them being diagnosed as AIS and approximately 68% of these patients having a history of high BP [3, 13]. Given this, the expected number of participants collected from this study was considerably lower than intended. Moreover, the hospital’s stroke team preferentially recommended hydralazine over the other antihypertensives from the SK; therefore, no comparison between the antihypertensives could be made.
Hydralazine from the SK was administered to one patient at 20:03, while three others received it from the RMR doses at 12:04, 17:55, and 01:52. Additional investigations need to be conducted to determine if relevant stroke team staff were available during those times to provide a rationale for preferring the RMR over the SK. Notably, the SK patient received hydralazine and tPA more quickly after ED presentation, suggesting potential for faster access; however, conclusions are limited by the small sample size. The lack of evidence supporting SK benefits in AIS further complicates justification for its use in reducing time to thrombolysis. Educating resuscitation nurses on SK contents may improve antihypertensive use, warranting future research into its impact on treatment timelines.
None of the alternative antihypertensives, IV metoprolol, verapamil or labetalol, were utilized from the SK or RMR in this study. American Heart Association and American Stroke Association guidelines recommend IV antihypertensives such as labetalol and nicardipine with hydralazine as an appropriate substitute [12]. This reinforces the need to optimize the hospital protocol, and then subsequently the SK to ensure that current guidelines are being met.
Another possible solution to address the lack of alternative antihypertensives being administered from the SK may involve removing IV verapamil and metoprolol from the SK. Instead, stocking both IV labetalol and IV nicardipine alongside hydralazine may be more appropriate. This change will also ensure that international guidelines are being upheld by the hospital for AIS management. Nonetheless, considerations such as cost-effectiveness among the three agents need to be investigated. Moreover, due to the restrictive nature of IV nicardipine being an SAS item, immediate access to this agent may not be possible. Another important factor to consider is the awareness of frontline staff utilizing the SK in the first place. This initiative may be a possible solution; however, it relies on multiple factors to be addressed initially to lead to positive clinical and organizational outcomes.
The median time taken for the hydralazine-treated patients to achieve BP target was 4.5 min (n = 6) (Table 2). In contrast, a similar study highlighted that the median time for hydralazine to achieve BP target took longer at 15 min (n = 2) [20]. In addition, a study by Lau et al discovered that the mean time to achieve BP target with hydralazine was 28.5 min (n = 54) [21]. This notable variation in timings emphasizes the unpredictable nature of hydralazine in BP control. However, due to the small number of patients and retrospective study design from both studies, further research is needed to justify the use of hydralazine in AIS therapy. The unpredictable nature and delayed onset of action (5–30 min) are highlighted when benchmarking hydralazine against IV labetalol and nicardipine, which are reported to have an onset of action within 2–5 and 5–15 min, respectively with easier titratability compared to hydralazine [15, 16].
The effectiveness of IV hydralazine on SBP and DBP was investigated (Table 2). Even though SBP and DBP are equally important, the control of SBP is the primary focus in AIS as it is generally higher upon presentation and heavily influences the appropriateness of tPA therapy [22]. In our patients, there was a clinically sufficient reduction in SBP after the administration of hydralazine. In contrast, the lowering effect of hydralazine on the DBP after the first dose of hydralazine was minor. These findings suggest that hydralazine effectively lowers SBP in the acute setting, with minimal impact on DBP. Even though SBP is the major focus, both SBP and DBP still need to be below the target BP for thrombolysis to be recommended. From this study, hydralazine’s median reduction of DBP from baseline was less (11 mm Hg) compared to the median drop of SBP after hydralazine administration (19 mm Hg). However, these results cannot be generalized due to the small sample size, yet they can act as a baseline for upcoming research. Other antihypertensives need to be compared in future projects to ensure that the most effective agent that can reduce both SBP and DBP is given to AIS patients.
To increase the utilization of SK in AIS patients presenting to ED, the adjustment of hospital guidelines and making labetalol available in the SK, an alternative first-line antihypertensive, may be a viable option. In studies by Lau et al [21] and McKay et al [20], the median time for labetalol to reach BP target was 12.9 min (n = 10) and 10.0 min (n = 25), respectively. Yet, a study by El-Ghoroury et al [23] recognized that labetalol took 24.0 min (n = 85) to manage the BP. These results indicate that there is a vast range between the median times after administration of IV labetalol in AIS patients. The average time for IV hydralazine was 18.2 min compared to IV labetalol of 15.6 min among these three studies, emphasizing that IV labetalol takes less time to reach target BP. Even so, this is inconclusive as these results were associated with small sample size studies and the lack of power calculations. Unfortunately, none of our labetalol-treated patients met the inclusion criteria to contribute to these results.
Direct comparisons of IV nicardipine, labetalol, and hydralazine have been explored in the literature, but such evidence remains limited. From previous studies by McKay et al [20] and El-Ghoroury et al [23], the median time to achieve BP target in AIS patients after receiving nicardipine was 22 min (n = 2) and 40 min (n = 3,008), respectively. Conversely, a study by Hao et al [24] argued that nicardipine’s effect on BP reduction was “significantly superior to that of labetalol”. Overseeing the previous research and this study, nicardipine seems to take longer to achieve BP goal compared to hydralazine and labetalol. Despite this, the onsets of action of labetalol, hydralazine, and nicardipine have been reported to be 2–5, 5–30, and 5–15 min, respectively from their respective medication profiles [14–16]. These discrepancies make it difficult to draw a definite conclusion on which antihypertensive is the most optimal for AIS management.
The NIHSS scores declined after thrombolysis, highlighting the importance of thrombolytics and antihypertensives in reducing the likelihood of disabling complications post-AIS. However, due to insufficient documentation on the EMR, only two patients were given a NIHSS score post-thrombolysis. This small sample size cannot accurately interpret NIHSS scores due to the lack of power analysis; therefore, conclusions cannot be made. Instead, these data can be used in addition to upcoming research. Lau et al [21] also measured mean NIHSS score at baseline of 7.5 (n = 54), which is comparable to our study; however, an NIHSS score post-thrombolysis was not determined. Local hospital guideline employs NIHSS score to assess acute neurological impairment, which is beneficial in the acute setting. The modified Rankin Scale (mRS) is another quantitative tool that can measure the level of disability that a patient may experience pre- and post-stroke, especially favorable in gauging long-term management goals [25]. The implementation of the mRS in the local hospital guideline, in conjunction with the existing NIHSS, may assist in providing a holistic care approach to AIS patients and may provide a more meaningful opportunity to assess the long-term effectiveness of tPA.
Limitations
The main limitations of this study are that no AIS patients were treated with labetalol, preventing a direct comparison of BP control with hydralazine. Additionally, the small sample size of six patients in the hydralazine group who met the inclusion criteria does not allow for generalization to the broader AIS population. However, it can serve as a foundation for further research. Initially during the data collection period, authors perceived difference in NIHSS score pre- and post-thrombolysis as the only technique to verify the effectiveness of thrombolysis therapy. However, the mRS is another valuable tool that can help with assessing the long-term effectiveness of tPA therapy [25]. As a result, only the short-term effectiveness of thrombolytic therapy could be determined. Lastly, due to the retrospective nature of this study, there were cases of insufficient documentation that led to difficulties in analyzing key information such as NIHSS scores and BP lowering effect. In hydralazine-treated patients, documentation of BP was not every 15 min for the first 2 h post-tPA as per the hospital protocol. Hence, there were discrepancies in the BP readings that were analyzed. The median time to reach target BP may not reflect what was truly recorded for these patients.
In conclusion, the infrequent usage of antihypertensives from the ED SK compared to the RMR at the hospital warrants a review of its contents. Further research is still required to identify the most optimal antihypertensive in reducing BP for pre-thrombolysis AIS patients. Inconsistent recording of NIHSS scores may indicate the need for practice updates to standardize documentation across the hospital, including the addition of mRS score recording to enable a more comprehensive assessment of post-stroke outcomes. Considerations such as increasing clinician usage of SK through education and upholding AIS clinical guidelines are necessary to appropriately manage AIS patients.
| Supplementary Material | ▴Top |
Suppl 1. Data collection tool template
Acknowledgments
The authors would like to thank members of the hospital’s Pharmacy Informatics team who assisted with extracting patients from the electronic medical records, and Dr Jason Vuong and Rjlyne Villaflor who assisted with data interpretation.
Financial Disclosure
This research received no grant from any funding agency in the public, commercial, or not-for-profit area.
Conflict of Interest
The authors declare they have no conflicts of interest.
Informed Consent
Not applicable.
Author Contributions
Conceptualization: Delan Pelendage-Perera (equal), Iouri Banakh (equal), Samuel Shepherd (equal); research question: Delan Pelendage-Perera (equal), Iouri Banakh (equal), Samuel Shepherd (equal); methodology: Delan Pelendage-Perera (equal), Iouri Banakh (equal); data extraction: Delan Pelendage-Perera (lead); data management: Delan Pelendage-Perera (lead); data synthesis: Delan Pelendage-Perera (equal), Iouri Banakh (equal); data analysis: Delan Pelendage-Perera (equal), Iouri Banakh (equal); writing – original draft preparation: Delan Pelendage-Perera (lead); reviewing and editing manuscript: Delan Pelendage-Perera (equal), Iouri Banakh (equal), Samuel Shepherd (equal).
Data Availability
The data that supports the finding of this study are available from the corresponding author upon request.
Abbreviations
AIS: acute ischemic stroke; BP: blood pressure; BPH: benign prostate hyperplasia; DBP: diastolic blood pressure; ED: emergency department; EMR: electronic medical records; HS: hemorrhagic stroke; HTN: hypertension; IV: intravenous; MGUS: monoclonal gammopathy of undetermined significance; mRS: modified Rankin Scale; NIHSS: National Institutes of Health Stroke Scale; RMR: resuscitation medication room; SAS: Special Access Scheme; SBP: systolic blood pressure; SD: standard deviation; SK: stroke kits; TIA: transient ischemic attack; tPA: tissue-type plasminogen activator
| References | ▴Top |
- Brain Foundation. Stroke: symptoms, treatments, and statistics in Australia. Brain Foundation. 2024. Available from: https://brainfoundation.org.au/disorders/stroke/. Accessed October 20, 2024.
- American Stroke Association. Ischemic Strokes (Clots). American Stroke Association. American Heart Association; 2024. Available from: https://www.stroke.org/en/about-stroke/types-of-stroke/ischemic-stroke-clots. Accessed June 2, 2025.
- Australian Institute of Health and Welfare. Heart, stroke and vascular disease: Australian facts, Stroke. Australian Institute of Health and Welfare. 2023. Available from: https://www.aihw.gov.au/reports/heart-stroke-vascular-diseases/hsvd-facts/contents/heart-stroke-and-vascular-disease-subtypes/stroke. Accessed March 6, 2025.
- Agency for Clinical Innovation. Endovascular clot retrieval for clot stroke; 2020. Available from: https://www.seslhd.health.nsw.gov.au/sites/default/files/groups/Prince_of_Wales_Hospital/Telestroke/ACI-Telestroke-ECR-patient-information.pdf. Accessed June 6, 2025.
- Jilani TN, Siddiqui AH. Tissue plasminogen activator(Archived). In: StatPearls. Treasure Island (FL) ineligible companies. 2025.
pubmed - Stroke Foundation. Australian and New Zealand Living Clinical Guidelines for Stroke Management - Chapter 3 of 8: Acute medical and surgical management. Reperfusion therapy; 2024. Available from: https://app.magicapp.org/#/guideline/QnoKGn/section/EQvlDj. Accessed October 14, 2024.
- Brott T, Adams HP, Jr., Olinger CP, Marler JR, Barsan WG, Biller J, Spilker J, et al. Measurements of acute cerebral infarction: a clinical examination scale. Stroke. 1989;20(7):864-870.
doi pubmed - Meng X, Li S, Dai H, Lu G, Wang W, Che F, Geng Y, et al. Tenecteplase vs alteplase for patients with acute ischemic stroke: the ORIGINAL randomized clinical trial. JAMA. 2024;332(17):1437-1445.
doi pubmed - Aiyagari V, Gorelick PB. Management of blood pressure for acute and recurrent stroke. Stroke. 2009;40(6):2251-2256.
doi pubmed - Hacke W, Kaste M, Bluhmki E, Brozman M, Davalos A, Guidetti D, Larrue V, et al. Thrombolysis with alteplase 3 to 4.5 hours after acute ischemic stroke. N Engl J Med. 2008;359(13):1317-1329.
doi pubmed - Management of Adult Transient Ischaemic Attack and Stroke Clinical Guideline, Monash Health. Version 7; Published March 2024. Accessed on July 20, 2024.
- Powers WJ, Rabinstein AA, Ackerson T, Adeoye OM, Bambakidis NC, Becker K, Biller J, et al. Guidelines for the early management of patients with acute ischemic stroke: 2019 update to the 2018 Guidelines for the Early Management of Acute Ischemic Stroke: A Guideline for Healthcare Professionals From the American Heart Association/American Stroke Association. Stroke. 2019;50(12):e344-e418.
doi pubmed - Stroke Foundation. Australian and New Zealand living clinical guidelines for stroke management - chapter 3 of 8: acute medical and surgical management. Acute blood pressure lowering therapy; 2025. Available from: https://app.magicapp.org/#/guideline/QnoKGn/section/EQvlDj. Accessed June 1, 2025.
- Whelton PK, Carey RM, Aronow WS, Casey DE, Jr., Collins KJ, Dennison Himmelfarb C, DePalma SM, et al. 2017 ACC/AHA/AAPA/ABC/ACPM/AGS/APhA/ASH/ASPC/NMA/PCNA guideline for the prevention, detection, evaluation, and management of high blood pressure in adults: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Hypertension. 2018;71(6):e13-e115.
doi pubmed - Miller M, Maani CV. Labetalol. In: StatPearls. Treasure Island (FL) ineligible companies. 2025.
pubmed - Marik PE, Varon J. Perioperative hypertension: a review of current and emerging therapeutic agents. J Clin Anesth. 2009;21(3):220-229.
doi pubmed - Nicardipine (intravenous). Stroke Medication Guideline, Royal Melbourne Hospital. Version 1; Published November 2017. Accessed on July 12, 2023.
- Nicardipine. Australian injectable drugs handbook 9th edition. SHPA. 2023. Available from: https://aidh.hcn.com.au/browse/n/nicardipine. Accessed July 12, 2023.
- Buntine P, Aldridge ES, Craig S, Crellin D, Stella J, Gill SD, Wright B, et al. Behavioural drivers influencing emergency department attendance in Victoria during the 2020 COVID-19 pandemic: A mixed methods investigation. Emerg Med Australas. 2022;34(5):758-768.
doi pubmed - McKay C, Hall AB, Cortes J. Time to blood pressure control before thrombolytic therapy in patients with acute ischemic stroke: comparison of labetalol, nicardipine, and hydralazine. J Neurosci Nurs. 2015;47(6):327-332.
doi pubmed - Lau TY, Oates G, Banakh I, Cam SL. Hydralazine versus labetalol for pre-thrombolysis hypertension for ischaemic stroke: a single centre retrospective study. [Oral presentation at the Medicines Management 2024 Conference]. November 14-16, 2024; Adelaide, Australia.
- Lindenstrom E, Boysen G, Nyboe J. Influence of systolic and diastolic blood pressure on stroke risk: a prospective observational study. Am J Epidemiol. 1995;142(12):1279-1290.
doi pubmed - El-Ghoroury H, Sudekum DM, Hecht JP. Blood pressure control in acute stroke: labetalol or nicardipine? J Stroke Cerebrovasc Dis. 2021;30(9):105959.
doi pubmed - Hao F, Yin S, Tang L, Zhang X, Zhang S. Nicardipine versus labetalol for hypertension during acute stroke: a systematic review and meta-analysis. Neurol India. 2022;70(5):1793-1799.
doi pubmed - Banks JL, Marotta CA. Outcomes validity and reliability of the modified Rankin scale: implications for stroke clinical trials: a literature review and synthesis. Stroke. 2007;38(3):1091-1096.
doi pubmed
This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (CC BY 4.0), which permits unrestricted use, distribution, and reproduction in any medium, including commercial use, provided the original work is properly cited.
Journal of Neurology Research is published by Elmer Press Inc.