| Journal of Neurology Research, ISSN 1923-2845 print, 1923-2853 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Neurol Res and Elmer Press Inc |
| Journal website https://jnr.elmerpub.com |
Original Article
Volume 16, Number 2, June 2026, pages 99-114
Can an Antidepressant Improve Motor Recovery After Stroke? A Meta-Analysis of Adjuvant Fluoxetine Reveals Both New Hope and Safety Concerns
I Nyoman Windianaa, c, Luh Putu Lina Kameliab, I Nyoman Gede Narendra Yanakusumac, I Gusti Agung Ayu Pramasinthya Aguseny Yudanaa, Komang Diah Kurnia Kesumaputric
aFaculty of Medicine, Universitas Pendidikan Ganesha, Bali, Indonesia
bDepartment of Neurology, Faculty of Medicine, Universitas Pendidikan Ganesha, Bali, Indonesia
cFaculty of Medicine, Universitas Udayana, Bali, Indonesia
dCorresponding Author: I Nyoman Windiana, Faculty of Medicine, Universitas Pendidikan Ganesha, Bali, Indonesia
Manuscript submitted December 17, 2025, accepted March 5, 2026, published online May 4, 2026
Short title: Fluoxetine for Post-Stroke Motor Recovery
doi: https://doi.org/10.14740/jnr1090
| Abstract | ▴Top |
Background: Stroke is a major cause of long-term disability, with motor impairments posing significant challenges to recovery. In 2011, fluoxetine, a selective serotonin reuptake inhibitor (SSRI), was reported to improve motor recovery in stroke patients by enhancing neuroplasticity and modulating the serotonergic system. However, its efficacy and safety in post-stroke rehabilitation remain controversial. The aim of the study was to evaluate the efficacy and safety of fluoxetine in improving motor recovery and health-related quality of life (HRQoL) after stroke.
Methods: This systematic review and meta-analysis was conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. A comprehensive search of PubMed, Scopus, and Google Scholar was performed for randomized controlled trials (RCTs) evaluating the effects of fluoxetine on motor recovery in post-stroke patients, up to March 2025. Data were analyzed using Review Manager 5.1.0. Motor recovery and HRQoL outcomes were reported as standardized mean differences (SMDs) using a random effects model, while adverse events were assessed as odds ratios (ORs) using a fixed effects model. The review was registered with PROSPERO (ID: CRD420251011077).
Results: Out of 1,173 identified studies, 23 RCTs comprising a total of 12,041 post-stroke patients met inclusion criteria. Fluoxetine significantly improved motor recovery (P = 0.01), particularly with a 90-day treatment duration (P = 0.006) at a daily dose of 20 mg (P = 0.05). HRQoL also improved significantly (P = 0.002). However, fluoxetine use was associated with a higher risk of adverse events (P < 0.00001), notably bone fractures (P < 0.00001), seizures (P = 0.009), and hyponatremia (P = 0.007).
Conclusions: Fluoxetine may offer benefits in enhancing motor recovery and HRQoL in post-stroke patients, especially when administered as a 90-day course at 20 mg/day. However, the increased risk of serious adverse events including bone fractures, seizures, and hyponatremia highlights the importance of careful patient selection and monitoring during treatment.
Keywords: Fluoxetine; Post-stroke; Motor recovery; Health-related quality of life; Adverse events
| Introduction | ▴Top |
Stroke is one of the leading causes of long-term disability worldwide, with motor impairments being among the most common and debilitating consequences [1, 2]. Post-stroke motor dysfunction significantly limits patients’ independence and quality of life, posing a major burden on healthcare systems and caregivers [3]. Despite advances in acute stroke management and rehabilitation techniques, many survivors experience incomplete motor recovery, highlighting the need for adjunctive therapies that can enhance neurorehabilitation outcomes [4].
In recent years, attention has turned to pharmacological agents that may promote neural repair and plasticity. One such agent is fluoxetine, a selective serotonin reuptake inhibitor (SSRI) traditionally used as an antidepressant [5]. Preclinical studies and early clinical trials have suggested that fluoxetine may enhance motor recovery after stroke by modulating neuroplasticity, increasing neurogenesis, and improving synaptic connectivity [6, 7]. The FLAME trial (2011) was one of the first randomized controlled trials (RCTs) to report a significant improvement in motor function with fluoxetine in stroke patients, independent of its antidepressant effects [8].
However, subsequent trials have produced mixed results regarding both the efficacy and safety of fluoxetine in post-stroke rehabilitation [9–11]. Concerns have emerged over adverse effects such as bone fractures, seizures, and hyponatremia, particularly in older adults [12, 13]. Moreover, differences in study designs, populations, and treatment durations have led to uncertainty about its routine clinical use [14].
Given these inconsistencies, a comprehensive synthesis of current evidence is needed to guide clinical decision-making. This systematic review and meta-analysis aims to evaluate the efficacy and safety of fluoxetine in improving motor recovery and health-related quality of life (HRQoL) in post-stroke patients, and to identify optimal treatment parameters for its potential use in stroke rehabilitation.
| Materials and Methods | ▴Top |
This systematic review and meta-analysis was reported in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines and registered in PROSPERO (ID: CRD420251011077). Institutional Review Board (IRB) approval was not required because this study did not involve direct human participants or identifiable personal data; therefore, ethical compliance with human or animal studies was not applicable.
Database and search strategy
A single reviewer (INW) conducted a comprehensive search of the Google Scholar, PubMed, and Scopus databases, covering all articles published from database inception up to March 2025. The search strategy included the following terms: (“Fluoxetine”) AND (“Motor Recovery”) AND (“Stroke”) AND (“Randomized Controlled Trial” OR “RCT” OR “FLAME Trial”). The search was restricted to studies evaluating the efficacy and safety of fluoxetine in improving motor recovery and HRQoL in post-stroke patients.
Study selection criteria
The inclusion criteria for this study were defined as follows: adults aged 18 years and older diagnosed with acute or chronic stroke; patients treated with fluoxetine compared to placebo; studies designed as RCTs, clinical trials, or randomized trials comparing fluoxetine with placebo in post-stroke populations; and studies reporting follow-up data on motor recovery, HRQoL, and treatment-related adverse events. Studies were excluded if they were reviews, case reports, or non-original research articles; lacked a placebo comparison group; or did not report outcomes related to motor recovery, HRQoL, or adverse events. Two authors (INW and INGNY) independently screened titles and abstracts using Microsoft Excel software. Any discrepancies in the selection process were resolved through discussion with a third investigator (IGAAPAY) to reach consensus.
Quality assessment
To evaluate the methodological quality of the included trials, the risk of bias was assessed at the study using the Cochrane Risk of Bias 2 (ROB-2) tool for RCTs. Two independent reviewers (INW and KDKK) screened all articles and independently rated the following domains: bias arising from the randomization process, bias due to deviations from intended interventions, bias due to missing outcome data, bias in measurement of the outcome, and bias in selection of the reported results. Each domain was classified as having a “low risk,” “some concerns,” or “high risk” of bias. Any discrepancies in data extraction or quality assessment were resolved through discussion until consensus was reached.
Data extraction and risk of bias assessment
Two independent authors (INW and IGAAPAY) conducted the data extraction process. A standardized data collection form was used to systematically extract key information from each trial, including the first author, year of publication, study location, sample size, patient diagnosis, study design, outcome measures, and measurement tools. Additionally, detailed information regarding the treatment protocols was recorded, including the intervention and control regimens, duration of treatment, and primary results.
Outcomes
The primary outcome of this study was motor recovery in post-stroke patients, assessed using a range of validated motor function measures. These included the Fugl–Meyer Motor Scale (FMMS), National Institutes of Health Stroke Scale (NIHSS), Rivermead Mobility Index (RMI), modified Rankin Scale (mRS), Vogel-Meyer Motor Scale, pinch strength measured with a dynamometer, number of finger tapping movements, Saltin-Grimby Physical Activity Level Scale (SGPALS), Timed Hand Function Test (THF), and upper extremity changes in the Fugl-Meyer Assessment (FMA-UE). Secondary outcomes included: i) HRQoL, evaluated using various instruments such as the Mental Health Inventory-5 (MHI-5), Barthel Index, Montgomery–Asberg Depression Rating Scale (MADRS), EuroQol 5 Dimensions 5 Levels (EQ-5D-5L), and Patient Health Questionnaire-9 (PHQ-9); 2) incidence of treatment-related adverse events, including bone fractures, seizures, hyperglycemia, hyponatremia, bleeding events, and all-cause mortality.
Statistical analysis
We conducted statistical analyses to evaluate the study outcomes. Motor recovery in post-stroke patients was analyzed based on treatment duration and optimal dosage, using the standardized mean difference (SMD) with a 95% confidence interval (CI), due to the variation in tools used to assess motor function. HRQoL outcomes were also assessed using SMD with 95% CI. For dichotomous outcomes, such as treatment-related adverse events, odds ratios (ORs) with 95% CIs were calculated. Heterogeneity among studies was assessed using the I2 statistic, with values of 25%, 50%, and 75% indicating low, moderate, and high heterogeneity, respectively. A P value of ≤ 0.05 was considered statistically significant for heterogeneity. In the presence of significant heterogeneity, a random-effects model was applied for meta-analysis; otherwise, a fixed-effects model was used. All statistical analyses were performed using Review Manager (RevMan) version 5.1.0 (The Cochrane Collaboration, UK).
| Results | ▴Top |
Article search and selection
A total of 1,173 potential research articles were initially identified. After removing duplicates, screening titles and abstracts, reviewing full texts, and applying the inclusion and exclusion criteria, 23 eligible studies were included in the systematic review and meta-analysis (Fig. 1).
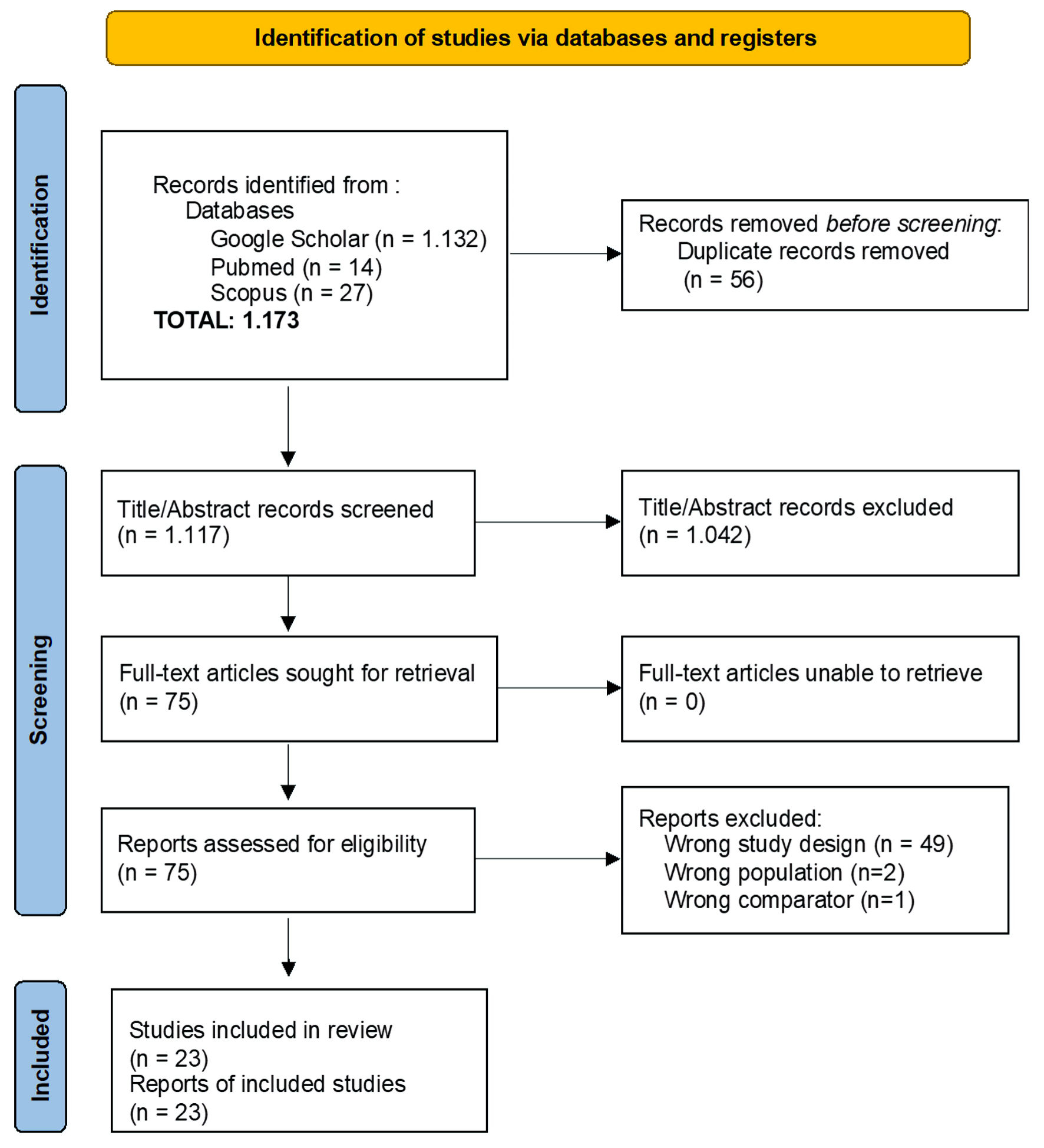 Click for large image | Figure 1. PRISMA flow diagram of study selection process for the systematic review and meta-analysis. PRISMA: Preferred Reporting Items for Systematic Reviews and Meta-Analyses. |
Risk of bias assessment
The quality of the included RCTs was evaluated using the Cochrane ROB-2 tool. Among the 23 included studies, six were identified as having “some concerns” regarding risk of bias, while one study was assessed as having a “high risk” of bias (Fig. 2). Babul et al (2017) [15] demonstrated some concerns due to insufficient reporting of the randomization process and blinding procedures. Although appropriate outcome measures were utilized and an intention-to-treat (ITT) analysis was conducted, missing data were imputed using mean substitution without adequate sensitivity analyses, potentially introducing bias. Dehghani et al (2024) [16] raised concerns primarily due to incomplete reporting on trial completion and missing data handling. The study did not clarify whether all enrolled participants completed the intervention, nor did it describe methods for addressing missing data, which may have introduced attrition bias. The study by Dike et al (2019) [17] was a single-blind trial that did not clearly specify whether participants or outcome assessors were blinded. Additionally, the lack of a placebo in the control group and the potential for unblinded outcome assessment posed a risk of detection and performance biases, especially given the reliance on subjective functional measures such as the FMMS and the mRS. The study by Guo et al (2016) [18] was limited by the absence of blinding among participants and clinicians, as well as by allowing re-randomization during the trial. Furthermore, the methods for handling missing data were not reported, increasing the potential for performance and attrition biases. He et al (2016) [19] exhibited some concerns due to lack of blinding in both patients and clinical staff, which may have influenced participants’ behavior and clinicians’ assessments. The study also did not provide sufficient details regarding allocation concealment, raising the possibility of selection bias. The study by Krishnan et al (2021) [20] lacked blinding in both participants and outcome assessors, which introduces a high risk of performance and detection bias. This is particularly significant given the use of subjective or semi-subjective outcome measures such as motor function scales. The study by Saadat et al (2025) [21] was rated as having a high risk of bias. This was attributed to the absence of transparency in the randomization process, unblinded outcome assessment, and the lack of a pre-registered protocol. These methodological shortcomings increase the likelihood of selective outcome reporting and exaggerated effect estimates.
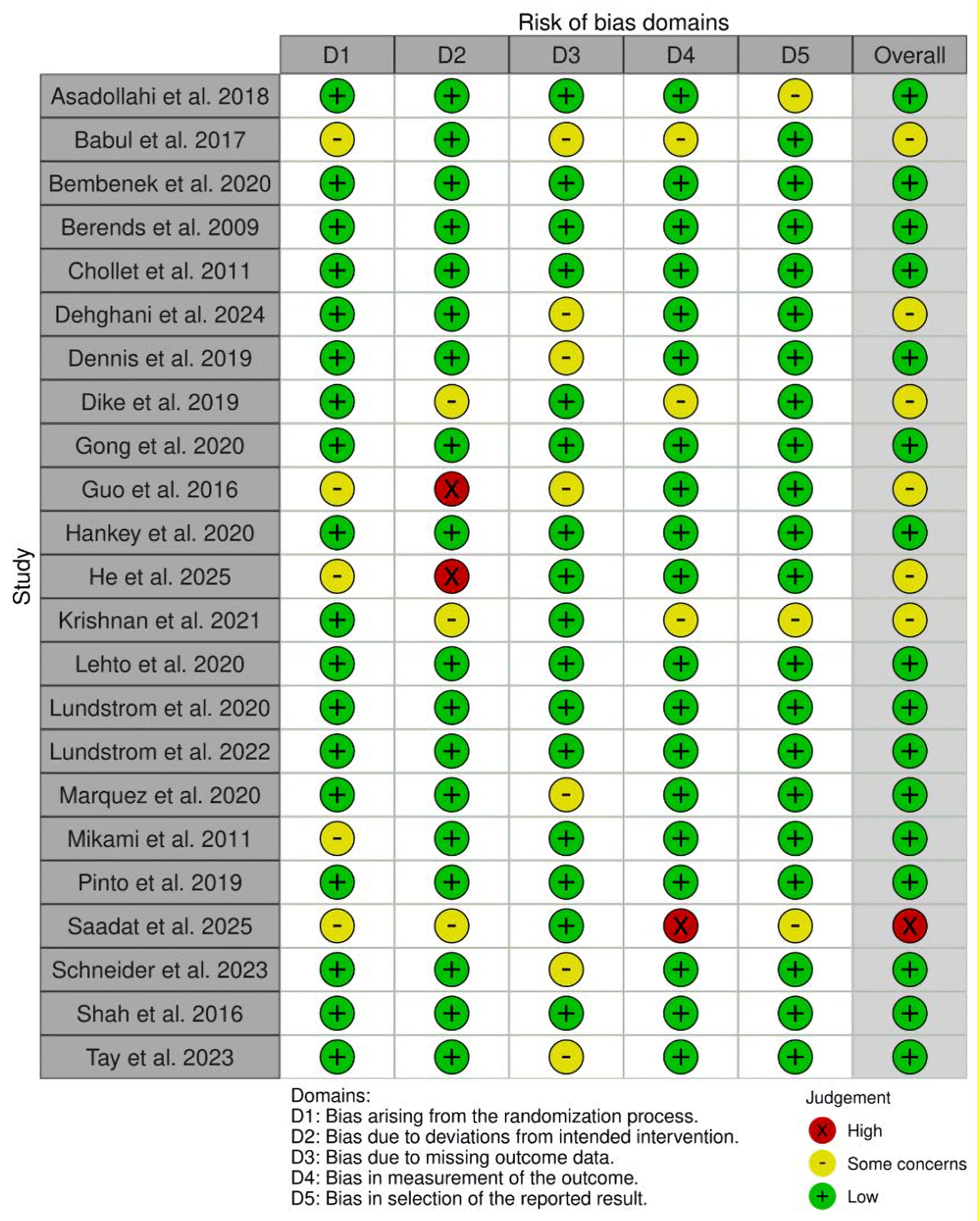 Click for large image | Figure 2. Risk of bias assessment of included studies using the Cochrane Risk of Bias 2 (ROB-2) tool. |
Characteristics of the studies
A comprehensive overview included a total of 23 RCTs involving 12,041 post-stroke patients, with publication dates ranging from 2009 to 2025. Sample sizes varied across studies, from 10 to 3,127 participants. Detailed descriptions of the interventions used in each trial are provided in Tables 1 and 2 [8, 15–36].
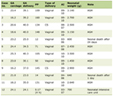 Click to view | Table 1. Characteristics of Included Studies in the Systematic Review and Meta-Analysis |
 Click to view | Table 2. Treatment Regimens Administered in Included Studies |
Meta-analysis results
Motor recovery
Sixteen studies were included in the analysis comparing the effects of fluoxetine versus placebo on motor recovery, categorized by optimal dose (Fig. 3) and treatment duration (Fig. 4). Due to significant heterogeneity among studies (I2 = 96%), SMDs were pooled using a random-effects model. The analysis demonstrated a significant improvement in motor recovery for the fluoxetine group with a treatment duration of 90 days (3 months) (SMD = 0.82; 95% CI, 0.23–1.42; P = 0.006) and an optimal daily dose of 20 mg (SMD = 0.36; 95% CI, 0.00–0.71; P = 0.05).
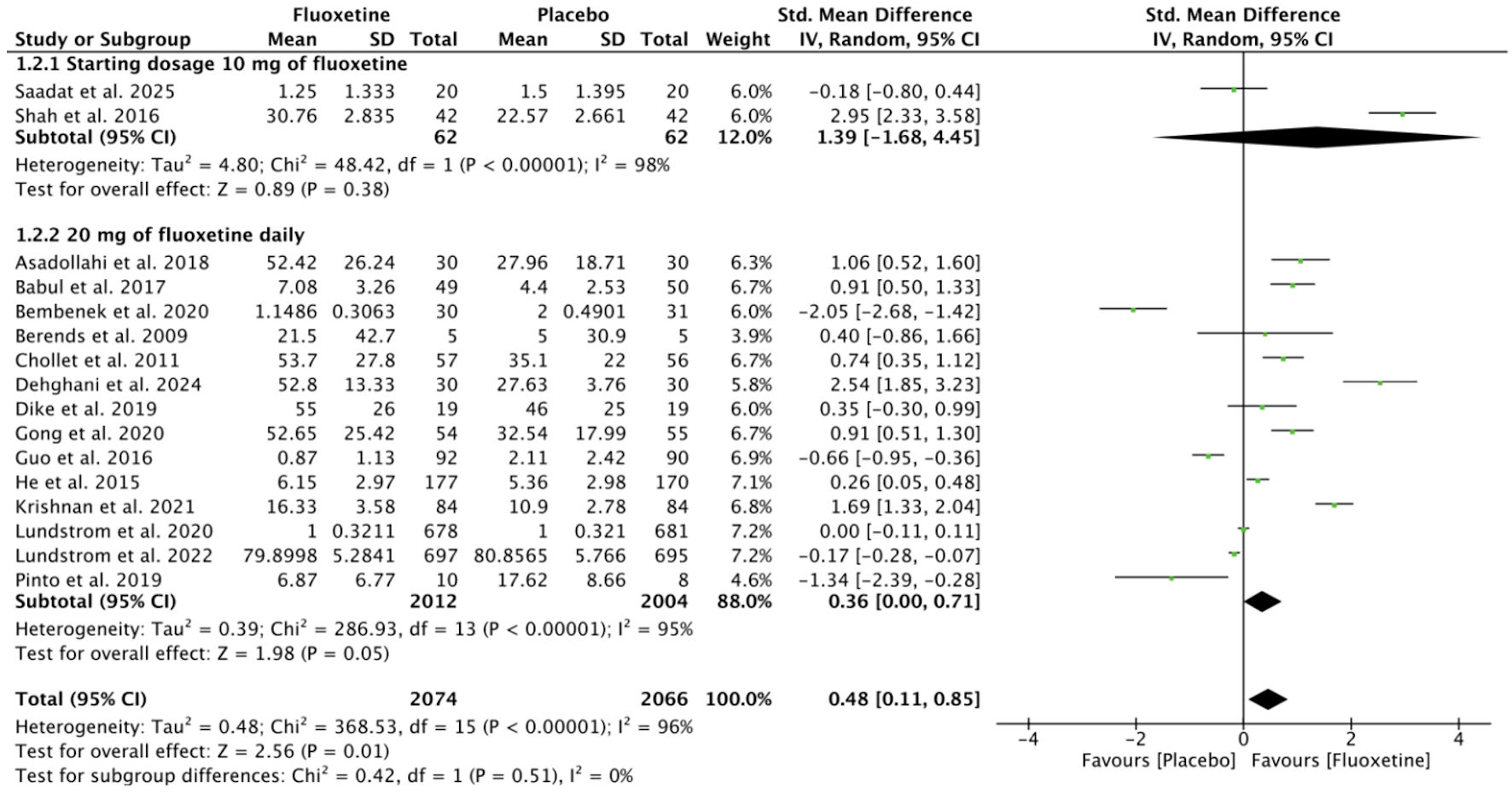 Click for large image | Figure 3. Comparison of fluoxetine dosages in relation to motor recovery outcomes. SD: standard deviation; CI: confidence interval. |
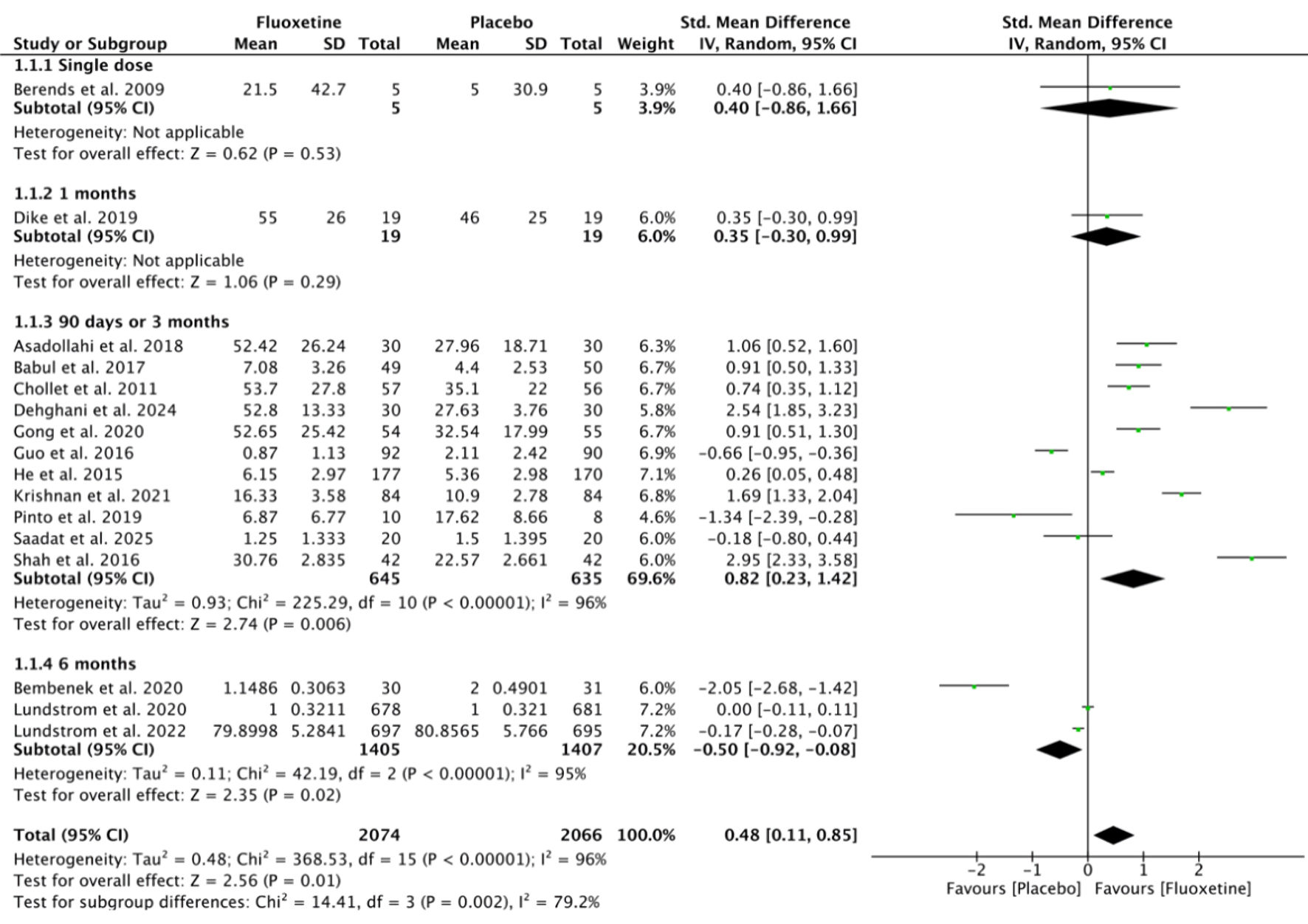 Click for large image | Figure 4. Effect of fluoxetine treatment duration on motor recovery in post-stroke patients. SD: standard deviation; CI: confidence interval. |
HRQoL
A total of 17 studies were included in the analysis comparing HRQoL between the fluoxetine and placebo groups (Fig. 5). Due to substantial heterogeneity among the studies (I2 = 98%), SMDs were pooled using a random-effects model. The results demonstrated a statistically significant improvement in HRQoL in the fluoxetine group compared to the placebo group (SMD = 0.37; 95% CI, 0.13–0.60; P = 0.002).
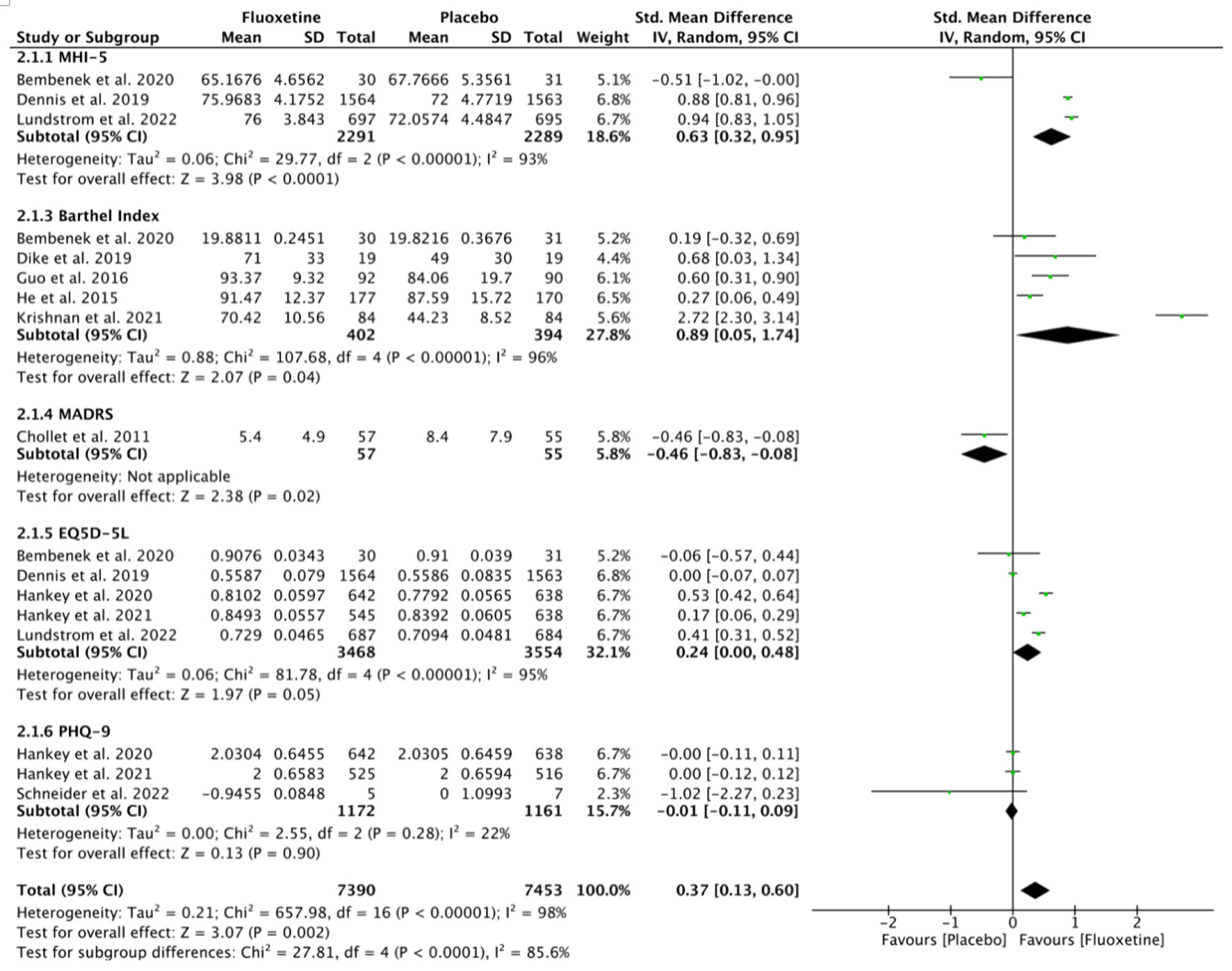 Click for large image | Figure 5. Effect of fluoxetine on health-related quality of life in post-stroke patients. SD: standard deviation; CI: confidence interval. |
New treatment-related adverse events
A total of eight studies were included in the analysis comparing the incidence of new treatment-related adverse events between the fluoxetine and placebo groups (Fig. 6). Based on the heterogeneity test results (I2 = 56.5%), ORs were pooled using a fixed-effect model. The analysis indicated that the fluoxetine group had a significantly higher risk of new adverse events compared to the placebo group (OR = 1.55; 95% CI, 1.33–1.80; P < 0.001), particularly bone fractures (OR = 2.30; 95% CI, 1.65–3.20; P < 0.001), seizures (OR = 1.48; 95% CI, 1.11–1.99; P = 0.009), and hyponatremia (OR = 2.11; 95% CI, 1.22–3.66; P = 0.007).
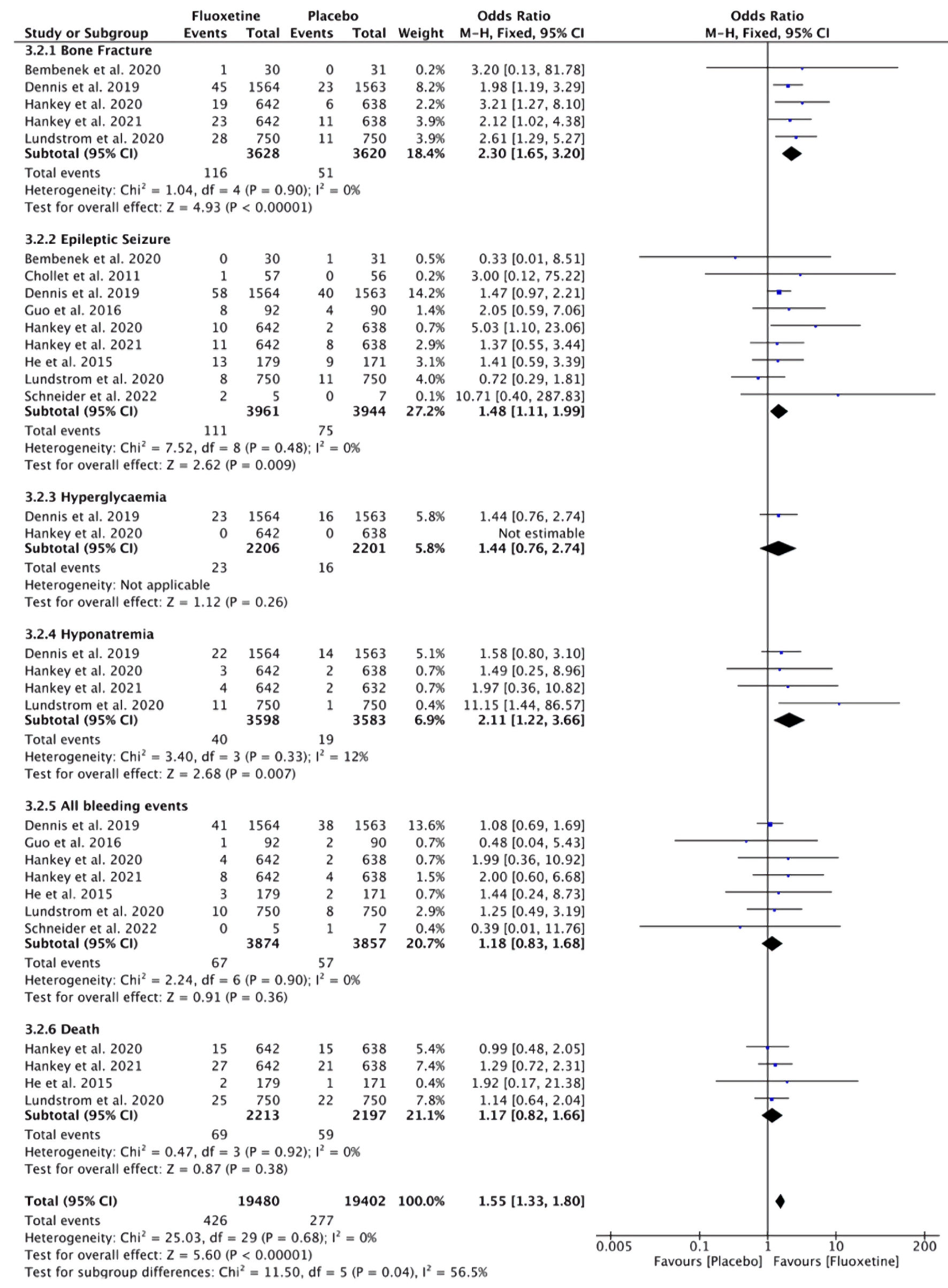 Click for large image | Figure 6. Incidence of new adverse events related to fluoxetine treatment in post-stroke patients. CI: confidence interval. |
Publication bias
To assess the presence of publication bias in the meta-analysis of motor recovery, HRQoL, and new incidence of adverse events related to treatment, the “metabias” function from Review Manager 5.1.0 (Cochrane, UK) was utilized. Upon examination of the funnel plot, it is evident that the distribution of studies on both the left and right sides of the plot is equal, suggesting a symmetrical distribution and no indication of publication bias (Fig.7).
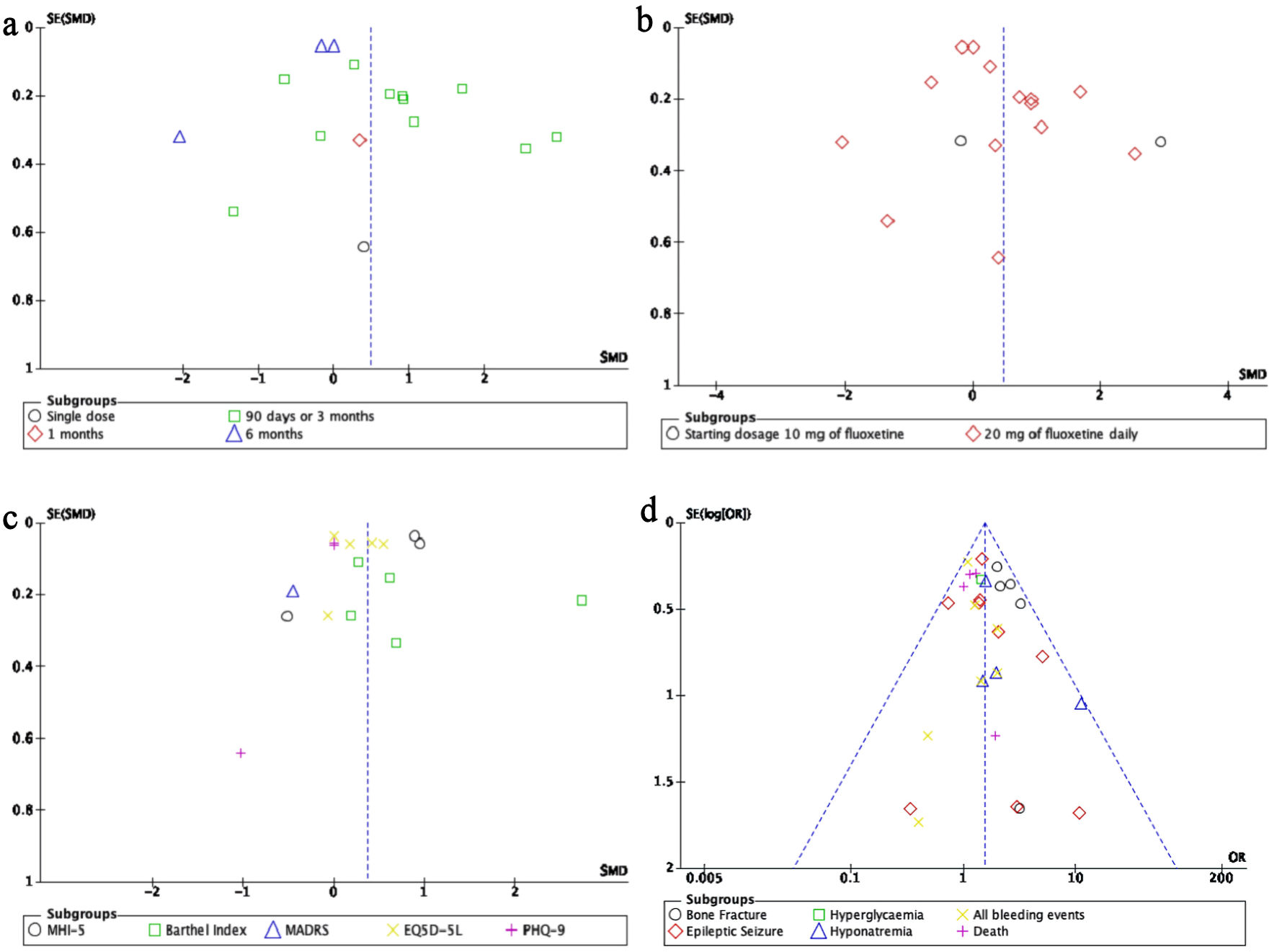 Click for large image | Figure 7. Publication bias was assessed using funnel plots for each outcome, including (a) duration of treatment on motor recovery, (b) dosage of treatment on motor recovery, (c) health-related quality of life, and (d) treatment-related adverse events. MHI-5: Mental Health Inventory-5; MADRS: Montgomery–Asberg Depression Rating Scale; EQ-5D-5L: EuroQol 5 Dimensions 5 Levels; PHQ-9: Patient Health Questionnaire-9. |
| Discussion | ▴Top |
The results of this analysis demonstrate that fluoxetine significantly improves motor recovery when administered at a daily dose of 20 mg for a duration of 3 months (90 days) and also enhances HRQoL. However, fluoxetine is associated with a significant increase in treatment-related adverse events, including bone fractures, seizures, and hyponatremia. These findings suggest that while fluoxetine offers benefits in motor recovery and quality of life, careful consideration must be given to the dosage, duration of treatment, and thorough screening for risk factors to minimize potential side effects.
Our findings align with those of Liu et al (2021) [37], who reported that fluoxetine may improve motor recovery but does not significantly enhance overall functional recovery. Similarly, Mead et al (2020) [38] found that fluoxetine is associated with better neurological and depression scores, as well as fewer diagnoses of depression, but observed no significant difference in the proportion of patients achieving independence or disability outcomes, while noting an increased risk of seizures. Earlier meta-analyses by Yi et al (2010) [39], Gu et al (2018) [40], and Mead et al (2012) [41] suggested that fluoxetine might reduce dependency and disability if administered early after stroke.
Importantly, the significant associations observed for seizures and hyponatremia were largely driven by a limited number of large-scale trials, particularly Hankey et al (2020) [22] and Lundstrom et al (2020) [23], which contributed the highest statistical weight in the pooled analysis. Although the pooled results demonstrated statistical significance, the relatively small number of studies reporting these specific adverse events warrants cautious interpretation. The heavy influence of a few large multicenter trials may have disproportionately shaped the pooled estimates. Therefore, these findings should be confirmed in future well-powered randomized trials specifically designed to evaluate safety outcomes.
Fluoxetine, primarily indicated for the treatment of depression and other psychiatric disorders, has recently garnered interest for its potential efficacy in promoting motor recovery post-stroke. Preclinical investigations in animal models have elucidated several mechanistic pathways, through which fluoxetine may exert neurorestorative effects [24, 25]. Notably, fluoxetine, as a SSRI, enhances neurogenesis within canonical neurogenic regions such as the hippocampus and the subventricular zone [26–28]. This effect is mediated, in part, by the upregulation of neurotrophic factors, which are critical for neuronal survival, synaptic plasticity, and the reorganization of neural circuits following ischemic injury [29–32, 42].
Moreover, fluoxetine exhibits anti-inflammatory properties by attenuating microglial activation and neutrophil infiltration, thereby reducing the release of pro-inflammatory cytokines that exacerbate neuronal injury [33–35]. This immunomodulatory capacity may contribute to the preservation of neural tissue and facilitate functional recovery. Additionally, fluoxetine appears to influence cerebrovascular regulation via the induction of heme oxygenase-1 (HO-1) and hypoxia-inducible factor-1 alpha (HIF-1α), proteins implicated in vascular homeostasis and adaptation to ischemic stress [36, 43]. The upregulation of β-1 adrenergic receptors observed in ischemic brain regions following SSRI treatment further suggests a potential role in enhancing neural repair processes, although the precise molecular mechanisms warrant further elucidation [44, 45].
Despite these compelling findings from animal studies, the clinical applicability of these mechanisms in human stroke patients remains to be conclusively demonstrated. The RCTs analyzed herein did not assess neurogenic or regenerative endpoints directly, likely due to the protracted timeline required for such processes and the relatively short duration of follow-up. While our meta-analysis supports the therapeutic benefits of fluoxetine in improving motor function and HRQoL, it concurrently underscores an increased incidence of adverse events, notably bone fractures, seizures, and hyponatremia. These safety concerns necessitate vigilant risk assessment and monitoring during clinical use.
Given the elevated risk of bone fractures associated with fluoxetine use, adjunctive strategies such as vitamin D supplementation may be considered to mitigate this adverse effect. Vitamin D plays a pivotal role in calcium homeostasis and bone metabolism, and its supplementation has been shown to reduce fracture risk in at-risk populations. Similarly, to address the increased incidence of seizures and hyponatremia, comprehensive monitoring of electrolyte levels and neurological status is essential. The potential role of adjunctive strategies, such as magnesium supplementation or seizure prophylaxis, remains uncertain and may warrant investigation in future studies, as current evidence is insufficient to support routine clinical implementation, although current evidence remains limited and warrants further clinical investigation.
Although preclinical evidence provides a robust biological rationale supporting fluoxetine’s neurorestorative potential, further rigorous clinical investigations are essential to validate these mechanisms in human populations, optimize dosing regimens, and balance therapeutic efficacy against patient safety. The incorporation of adjunctive interventions, such as vitamin D supplementation and careful electrolyte management, may mitigate adverse events and improve the overall safety profile of fluoxetine in post-stroke rehabilitation.
Despite these promising findings, this meta-analysis is subject to several important limitations that warrant careful consideration. A primary contributor to heterogeneity is the considerable variation in assessment tools employed to measure motor recovery across included studies. While some trials utilized comprehensive and validated instruments like the FMMS, others relied on alternative measures such as the NIHSS, RMI, or finger tapping counts. These instruments differ markedly in sensitivity, scope, and focus, which likely contributed to the moderate to high heterogeneity observed in the primary outcomes (I2 = 96%), thereby limiting direct comparability and generalizability of results. Moreover, variability in study design, sample size, and patient demographic characteristics may have further amplified this heterogeneity.
Although fluoxetine demonstrated statistically significant improvements in motor recovery and HRQoL, the associated increase in treatment-related adverse events including bone fractures, seizures, and hyponatremia raises critical safety concerns that may restrict its broad clinical applicability. Additional heterogeneity arose from differences in treatment duration, dosage regimens, follow-up periods, and outcome assessment methodologies, further complicating the interpretation and external validity of the findings.
To contextualize the observed safety signals, it is important to consider whether these adverse events are specific to the post-stroke population or represent established pharmacological effects of fluoxetine. In psychiatric populations, SSRIs, including fluoxetine, have been associated with an increased risk of falls, bone fractures, hyponatremia, and, in rare cases, seizures. Several large observational studies in patients treated for depression have demonstrated a higher incidence of fracture and fall-related injuries, potentially mediated by SSRI-induced reductions in bone mineral density, impaired platelet function, or effects on balance and psychomotor performance [45–48].
These findings suggest that at least part of the increased risk observed in our meta-analysis may reflect class-related adverse effects rather than stroke-specific mechanisms alone. However, patients recovering from stroke may be particularly vulnerable due to pre-existing motor impairment, frailty, advanced age, and polypharmacy. Such factors could amplify the baseline pharmacological risks associated with fluoxetine, thereby contributing to the higher incidence of adverse outcomes observed in this population. Distinguishing between drug-intrinsic effects and stroke-related susceptibility remains challenging and warrants further investigation through comparative safety analyses across different patient populations.
Demographic characteristics represent an important yet underexplored factor in the interpretation of safety outcomes. The observed increase in bone fracture risk, for instance, may carry differential clinical implications across patient subgroups, particularly among postmenopausal women, who have a higher baseline prevalence of osteoporosis. However, most included trials did not consistently report stratified analyses by sex or age, thereby limiting our ability to evaluate potential effect modification across demographic subgroups. The absence of detailed demographic reporting restricts meaningful risk stratification and may obscure clinically relevant heterogeneity in treatment response and adverse event susceptibility.
Similarly, prior exposure to fluoxetine or other SSRIs represents a potentially influential yet insufficiently documented variable. The majority of included trials did not specify whether participants were fluoxetine-naive at the time of enrollment. Previous antidepressant exposure may plausibly influence both therapeutic responsiveness and tolerability through alterations in neuroplasticity pathways or receptor sensitivity, thereby affecting clinical outcomes following stroke. Future investigations should explicitly document and stratify patients according to prior SSRI exposure to clarify its role in modulating recovery trajectories.
Taken together, these considerations underscore the need for rigorously designed, adequately powered multicenter RCTs employing standardized outcome measures and comprehensive baseline characterization. Future studies should extend follow-up beyond the commonly evaluated 90-day treatment window, delineate patient subpopulations most likely to derive net clinical benefit, and directly compare fluoxetine with alternative pharmacological and non-pharmacological rehabilitation strategies. In parallel, mechanistic studies incorporating advanced neuroimaging or biomarker analyses may further elucidate the biological substrates underlying observed clinical effects. Such efforts are essential to refining clinical guidelines and optimizing the risk-benefit profile of fluoxetine in post-stroke rehabilitation.
Conclusions
This systematic review and meta-analysis demonstrates that fluoxetine, administered at a dose of 20 mg/day for 90 days, may significantly enhance motor recovery and improve HRQoL in post-stroke patients. However, these therapeutic benefits are tempered by an increased risk of serious adverse events, notably bone fractures, seizures, and hyponatremia. Given these safety concerns and variability among studies, fluoxetine should be prescribed with caution, limited to carefully selected patients under vigilant clinical supervision. To mitigate the risk of adverse events, adjunctive strategies such as vitamin D supplementation to reduce fracture risk, along with appropriate electrolyte monitoring and seizure prophylaxis, should be considered. Future high-quality, large-scale RCTs are warranted to further elucidate the long-term safety profile of fluoxetine, identify patient subgroups most likely to benefit, and compare its efficacy with alternative rehabilitation interventions, thereby guiding evidence-based clinical practice.
Acknowledgments
The authors would like to express their sincere gratitude to the Faculty of Medicine, Universitas Pendidikan Ganesha, for their guidance and academic support during the preparation of this manuscript.
Financial Disclosure
The authors received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The authors declare that there is no conflict of interest regarding the publication of this article.
Informed Consent
Informed consent was not applicable for this study, as it was a systematic review and meta-analysis based exclusively on previously published data, with no direct involvement of human participants.
Author Contributions
I Nyoman Windiana conceived and designed the study, developed the research protocol, conducted literature search and study selection, performed data extraction and statistical analysis, and drafted the manuscript. Luh Putu Lina Kamelia contributed to literature search, study selection, data extraction, and critical revision of the manuscript for important intellectual content. I Nyoman Gede Narendra Yanakusuma participated in data extraction, quality assessment of included studies, and statistical analysis. I Gusti Agung Ayu Pramasinthya Aguseny Yudana contributed to data interpretation, manuscript editing, and critical review of the final version. Komang Diah Kurnia Kesumaputri participated in data extraction, quality assessment of included studies, and statistical analysis. All authors read and approved the final manuscript.
Data Availability
The authors declare that data supporting the findings of this study are available within the article.
| References | ▴Top |
- Jambi LK, Hamad A, Salah H, Sulieman A. Stroke and disability: Incidence, risk factors, management, and impact. Journal of Disability Research. 2024;3(7):20240094.
- Ghadirpour R, Unit NN, Ghadirpour R. The aftermath of ischemic stroke: inflammation, comorbidity, and disability. Eur J Neurodegenerative Dis. 2023;12:35-40.
- Tiwari S, Joshi A, Rai N, Satpathy P. Impact of stroke on quality of life of stroke survivors and their caregivers: a qualitative study from India. J Neurosci Rural Pract. 2021;12(4):680-688.
doi pubmed - Li X, He Y, Wang D, Rezaei MJ. Stroke rehabilitation: from diagnosis to therapy. Front Neurol. 2024;15:1402729.
doi pubmed - Zelek-Molik A, Litwa E. Trends in research on novel antidepressant treatments. Front Pharmacol. 2025;16:1544795.
doi pubmed - Rogalewski A, Schabitz WR. Stroke recovery enhancing therapies: lessons from recent clinical trials. Neural Regen Res. 2022;17(4):717-720.
doi pubmed - Rosas-Sanchez GU, German-Ponciano LJ, Guillen-Ruiz G, Cueto-Escobedo J, Limon-Vazquez AK, Rodriguez-Landa JF, Soria-Fregozo C. Neuroplasticity and mechanisms of action of acute and chronic treatment with antidepressants in preclinical studies. Biomedicines. 2024;12(12):2744.
doi pubmed - Bembenek JP, Niewada M, Klysz B, Mazur A, Kurczych K, Gluszkiewicz M, Czlonkowska A. Fluoxetine for stroke recovery improvement - the doubleblind, randomised placebo-controlled FOCUS-Poland trial. Neurol Neurochir Pol. 2020;54(6):544-551.
doi pubmed - Su N, Wen C, Guo S, Yu Y, Wang C. The efficacy and tolerability of selective serotonin reuptake inhibitors for motor recovery in non-depressed patients after acute stroke: a meta-analysis. Front Neurol. 2021;12:749322.
doi pubmed - Kalbouneh HM, Toubasi AA, Albustanji FH, Obaid YY, Al-Harasis LM. Safety and efficacy of SSRIs in improving poststroke recovery: a systematic review and meta-analysis. J Am Heart Assoc. 2022;11(13):e025868.
doi pubmed - Elsnhory A, Hasan MT, Hagrass AI, Hanbal A, Fathy A, Ahmed E, Ouerdane Y, et al. Recovery in stroke patients treated with fluoxetine versus placebo: a pooled analysis of 7,165 patients. Neurologist. 2023;28(2):104-116.
doi pubmed - Pannu A, Goyal RK. From evidence to practice: a comprehensive analysis of side effects in synthetic anti-depressant therapy. Curr Drug Saf. 2025;20(2):120-147.
doi pubmed - Anagha K, Shihabudheen P, Uvais NA. Side effect profiles of selective serotonin reuptake inhibitors: a cross-sectional study in a naturalistic setting. Prim Care Companion CNS Disord. 2021;23(4).
doi pubmed - Su D, Zhang Y, Wang A, Shao D, Xu F, Dong M, Zhao L, et al. Efficacy and tolerability of selective serotonin reuptake inhibitors on promoting motor recovery after stroke:meta-analysis of randomized controlled trials. Expert Rev Neurother. 2021;21(10):1179-1189.
doi pubmed - Babul M, Hassanuzzaman, Ahammed Z, Kibria A, Faruk G, Azam A, Rahman M. Fluoxetine for Motor recovery after acute ischemic stroke: a randomized placebo-controlled trial. Neuroscience. 2017;5(3):31-36.
- Dehghani V, Masoud SA, Noorbakhsh M, Akbari H, Rezapoor Kafteroodi B, Masoud M. Investigating the effect of citalopram and fluoxetine on motor function after stroke in patients with acute cerebral stroke. Neurology Letters. 2024;3(2):43-49.
- Dike FO, Ekeh BC, Ogun AS. Pharmacological enhancement of motor function recovery in patients with acute ischaemic stroke: a trial of fluoxetine. J Neurol Stroke. 2019;9(1):47-51.
- Guo Y, He Y, Tang B, Ma K, Cai Z, Zeng S, Zhang Y, et al. Effect of using fluoxetine at different time windows on neurological functional prognosis after ischemic stroke. Restor Neurol Neurosci. 2016;34(2):177-187.
doi pubmed - He YT, Tang BS, Cai ZL, Zeng SL, Jiang X, Guo Y. Effects of fluoxetine on neural functional prognosis after ischemic stroke: a randomized controlled study in China. J Stroke Cerebrovasc Dis. 2016;25(4):761-770.
doi pubmed - Krishnan K, K M, K N, Teja YD, Reddy VS, Raju NS, Rathinam KK. Role of fluoxetine in pharmacological enhancement of motor functions in stroke patients: A randomized, placebo-controlled, single-blind trial. Contemp Clin Trials Commun. 2021;23:100800.
doi pubmed - Saadat P, Hamzehpour R, Karimi F, Ebrahimi P, Haji Ahmadi M. Fluoxetine versus citalopram in improving post-stroke motor function: a comparative single-blind clinical trial. Caspian Journal of Neurological Sciences. 2025;11(2):132-139.
- Affinity Trial Collaboration. Safety and efficacy of fluoxetine on functional outcome after acute stroke (AFFINITY): a randomised, double-blind, placebo-controlled trial. Lancet Neurol. 2020;19(8):651-660.
doi pubmed - Collaboration ET. Safety and efficacy of fluoxetine on functional recovery after acute stroke (EFFECTS): a randomised, double-blind, placebo-controlled trial. Lancet Neurol. 2020;19(8):661-669.
doi pubmed - Asadollahi M, Ramezani M, Khanmoradi Z, Karimialavijeh E. The efficacy comparison of citalopram, fluoxetine, and placebo on motor recovery after ischemic stroke: a double-blind placebo-controlled randomized controlled trial. Clin Rehabil. 2018;32(8):1069-1075.
doi pubmed - Berends HI, Nijlant J, van Putten M, Movig KL, MJ IJ. Single dose of fluoxetine increases muscle activation in chronic stroke patients. Clin Neuropharmacol. 2009;32(1):1-5.
pubmed - Chollet F, Tardy J, Albucher JF, Thalamas C, Berard E, Lamy C, Bejot Y, et al. Fluoxetine for motor recovery after acute ischaemic stroke (FLAME): a randomised placebo-controlled trial. Lancet Neurol. 2011;10(2):123-130.
doi pubmed - Collaboration FT. Effects of fluoxetine on functional outcomes after acute stroke (FOCUS): a pragmatic, double-blind, randomised, controlled trial. Lancet. 2019;393(10168):265-274.
doi pubmed - Gong L, Yang X, Feng Y, Fei Z, Wang M, Qin B, Wang Q, Pan W. The efficacy of integrative anti-depressive therapy on motor recovery after ischemic stroke–A randomized clinical trial. European Journal of Integrative Medicine. 2020;35:101102.
- Lehto E. Can fluoxetine increase the level ofphysical activity after stroke? An exploratory analysis of EFFECTS, a randomized controlled trial.
- Lundstrom E, Isaksson E, Greilert Norin N, Nasman P, Wester P, Martensson B, Norrving B, et al. Effects of Fluoxetine on Outcomes at 12 Months After Acute Stroke: Results From EFFECTS, a Randomized Controlled Trial. Stroke. 2021;52(10):3082-3087.
doi pubmed - Marquez-Romero JM, Reyes-Martinez M, Huerta-Franco MR, Ruiz-Franco A, Silos H, Arauz A. Fluoxetine for motor recovery after acute intracerebral hemorrhage, the FMRICH trial. Clin Neurol Neurosurg. 2020;190:105656.
doi pubmed - Mikami K, Jorge RE, Adams HP, Jr., Davis PH, Leira EC, Jang M, Robinson RG. Effect of antidepressants on the course of disability following stroke. Am J Geriatr Psychiatry. 2011;19(12):1007-1015.
doi pubmed - Bonin Pinto C, Morales-Quezada L, de Toledo Piza PV, Zeng D, Saleh Velez FG, Ferreira IS, Lucena PH, et al. Combining fluoxetine and rTMS in poststroke motor recovery: a placebo-controlled double-blind randomized phase 2 clinical trial. Neurorehabil Neural Repair. 2019;33(8):643-655.
doi pubmed - Schneider CL, Prentiss EK, Busza A, Williams ZR, Mahon BZ, Sahin B. FLUORESCE: a pilot randomized clinical trial of fluoxetine for vision recovery after acute ischemic stroke. J Neuroophthalmol. 2023;43(2):237-242.
doi pubmed - Shah IA, Asimi RP, Kawoos Y, Wani MA, Wani MA, Dar MA. Effect of fluoxetine on motor recovery after acute haemorrhagic stroke: a randomized trial. J Neurol Neurophysiol. 2016;7(2):364.
- Tay J, Collaboration ET, Martensson B, Markus HS, Lundstrom E. Does fluoxetine reduce apathetic and depressive symptoms after stroke? An analysis of the Efficacy oF Fluoxetine-a randomized Controlled Trial in Stroke trial data set. Int J Stroke. 2023;18(3):285-295.
doi pubmed - Liu G, Yang X, Xue T, Chen S, Wu X, Yan Z, Wang Z, et al. Is Fluoxetine Good for Subacute Stroke? A meta-analysis evidenced from randomized controlled trials. Front Neurol. 2021;12:633781.
doi pubmed - Mead GE, Legg L, Tilney R, Hsieh CF, Wu S, Lundstrom E, Rudberg AS, et al. Fluoxetine for stroke recovery: Meta-analysis of randomized controlled trials. Int J Stroke. 2020;15(4):365-376.
doi pubmed - Yi ZM, Liu F, Zhai SD. Fluoxetine for the prophylaxis of poststroke depression in patients with stroke: a meta-analysis. Int J Clin Pract. 2010;64(9):1310-1317.
doi pubmed - Gu SC, Wang CD. Early selective serotonin reuptake inhibitors for recovery after stroke: a meta-analysis and trial sequential analysis. J Stroke Cerebrovasc Dis. 2018;27(5):1178-1189.
doi pubmed - Mead GE, Hsieh CF, Lee R, Kutlubaev MA, Claxton A, Hankey GJ, Hackett ML. Selective serotonin reuptake inhibitors (SSRIs) for stroke recovery. Cochrane Database Syst Rev. 2012;11(11):CD009286.
doi pubmed - Hankey GJ, Hackett ML, Almeida OP, Flicker L, Mead GE, Dennis MS, Etherton-Beer C, et al. Twelve-month outcomes of the AFFINITY trial of fluoxetine for functional recovery after acute stroke: AFFINITY trial steering committee on behalf of the AFFINITY trial collaboration. Stroke. 2021;52(8):2502-2509.
doi pubmed - Zhang J, Zhang N, Lei J, Jing B, Li M, Tian H, Xue B, et al. Fluoxetine shows neuroprotective effects against LPS-induced neuroinflammation via the Notch signaling pathway. Int Immunopharmacol. 2022;113(Pt A):109417.
doi pubmed - Olivas-Cano I, Rodriguez-Andreu JM, Blasco-Ibanez JM, Crespo C, Nacher J, Varea E. Fluoxetine increased adult neurogenesis is mediated by 5-HT3 receptor. Neurosci Lett. 2023;795:137027.
doi pubmed - Filipovic D, Turck CW. Prefrontal cortex molecular signatures of chronically socially isolated rats and their response to fluoxetine treatment. Mol Neurobiol. 2025;62(11):14544-14559.
doi pubmed - Xu Y, Ku B, Tie L, Yao H, Jiang W, Ma X, et al. Neuroprotective effects of fluoxetine against microglial activation and neuronal apoptosis in cerebral ischemia. Neuroscience. 2008;152(3):753-761.
- Hu Q, Liu L, Zhou L, Lu H, Wang J, Chen X, Wang Q. Effect of fluoxetine on HIF-1alpha- Netrin/VEGF cascade, angiogenesis and neuroprotection in a rat model of transient middle cerebral artery occlusion. Exp Neurol. 2020;329:113312.
doi pubmed - Palvimaki EP, Laakso A, Kuoppamaki M, Syvalahti E, Hietala J. Up-regulation of beta 1-adrenergic receptors in rat brain after chronic citalopram and fluoxetine treatments. Psychopharmacology (Berl). 1994;115(4):543-546.
doi pubmed
This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (CC BY 4.0), which permits unrestricted use, distribution, and reproduction in any medium, including commercial use, provided the original work is properly cited.
Journal of Neurology Research is published by Elmer Press Inc.