| Journal of Neurology Research, ISSN 1923-2845 print, 1923-2853 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Neurol Res and Elmer Press Inc |
| Journal website https://jnr.elmerpub.com |
Original Article
Volume 16, Number 2, June 2026, pages 77-87
Genotype-Guided Dual Antiplatelet Therapy in CYP2C19 Loss-of-Function Carriers With Stroke or Transient Ischemic Attack: A Meta-Analysis of Ticagrelor–Aspirin Versus Clopidogrel–Aspirin
I Nyoman Windianaa, e, Muhammad Iqhrammullahb, Luh Putu Lina Kameliac, I Nyoman Gede Narendra Yanakusumad, Komang Diah Kurnia Kesumaputrid, I Gusti Agung Ayu Pramasinthya Aguseny Yudanaa
aFaculty of Medicine, Universitas Pendidikan Ganesha, Bali, Indonesia
bUniversitas Muhammadiyah Aceh, Aceh, Indonesia
cDepartment of Neurology, Faculty of Medicine, Universitas Pendidikan Ganesha, Bali, Indonesia
dFaculty of Medicine, Universitas Udayana, Bali, Indonesia
eCorresponding Author: I Nyoman Windiana, Faculty of Medicine, Universitas Pendidikan Ganesha, Bali, Indonesia
Manuscript submitted January 30, 2026, accepted March 18, 2026, published online May 4, 2026
Short title: DAPT in CYP2C19 LOF Carriers With Stroke or TIA
doi: https://doi.org/10.14740/jnr1101
| Abstract | ▴Top |
Background: Individuals presenting with transient ischemic attack (TIA) or minor ischemic stroke face a substantial risk of early stroke recurrence. Although dual antiplatelet therapy (DAPT) lowers the incidence of ischemic events, it is accompanied by an elevated bleeding risk. Moreover, CYP2C19 genetic polymorphisms, particularly loss-of-function (LOF) variants, may impair clopidogrel metabolism and attenuate its antiplatelet efficacy, underscoring the need to evaluate alternative therapeutic strategies. This study aimed to compare the efficacy and safety of ticagrelor plus aspirin versus clopidogrel plus aspirin among CYP2C19 LOF carriers with TIA or minor ischemic stroke.
Methods: A systematic review and meta-analysis was performed following PRISMA recommendations. Electronic databases including PubMed, Scopus, and Google Scholar were systematically screened to identify randomized controlled trials evaluating ticagrelor–aspirin versus clopidogrel–aspirin in patients harboring CYP2C19 LOF alleles. Primary and secondary outcomes comprised 90-day recurrent ischemic or hemorrhagic stroke, composite vascular events, intracranial hemorrhage, and all-cause mortality. Pooled risk ratios (RRs) with corresponding 95% confidence intervals (CIs) were calculated to estimate treatment effects.
Results: Twelve eligible studies encompassing 64,128 CYP2C19 LOF carriers were included in the final analysis. Compared with clopidogrel–aspirin therapy, ticagrelor–aspirin was associated with a significantly lower risk of recurrent stroke at 90 days (P < 0.00001) and composite vascular events (P < 0.00001). Additionally, ticagrelor-based DAPT demonstrated a reduced incidence of intracranial hemorrhage (P = 0.005) and mortality (P < 0.0001).
Conclusion: Among CYP2C19 LOF carriers with TIA or minor ischemic stroke, ticagrelor combined with aspirin appears to offer superior protection against recurrent stroke and vascular complications compared with clopidogrel–aspirin, while also reducing intracranial hemorrhage and mortality. Nevertheless, individualized risk–benefit assessment remains essential, particularly considering the bleeding profile associated with ticagrelor.
Keywords: Ticagrelor; Clopidogrel; Aspirin; Transient ischemic attack; CYP2C19 loss-of-function
| Introduction | ▴Top |
Patients presenting with transient ischemic attack (TIA) or minor ischemic stroke face a substantial risk of early stroke recurrence, particularly within the first weeks following the index event. Dual antiplatelet therapy (DAPT), most commonly clopidogrel combined with aspirin, has been shown to lower the incidence of recurrent ischemic events in both cardiovascular and cerebrovascular settings, although this benefit is accompanied by an increased risk of major bleeding complications [1].
The Platelet-Oriented Inhibition in New TIA and Minor Ischemic Stroke (POINT) trial, a large international randomized, double-blind, placebo-controlled study, evaluated a 90-day regimen of clopidogrel plus aspirin versus aspirin monotherapy in patients with high-risk TIA or minor ischemic stroke. The study demonstrated a significant reduction in major ischemic events with combination therapy; however, this advantage was offset by a modest but statistically significant increase in major hemorrhage [2]. Likewise, the Clopidogrel in High-Risk Patients with Non-disabling Cerebrovascular Events (CHANCE) trial conducted in a Chinese population reported that 21 days of clopidogrel–aspirin therapy reduced recurrent stroke without a significant rise in bleeding events, with the greatest net clinical benefit observed during the early treatment period [3].
Despite these favorable outcomes, the effectiveness of clopidogrel is highly dependent on hepatic bioactivation through the cytochrome P450 system. As a prodrug, clopidogrel requires metabolic conversion to its active form, a process influenced by genetic polymorphisms of the CYP2C19 enzyme. Carriers of CYP2C19 loss-of-function (LOF) alleles estimated to affect approximately 25% of Caucasians and up to 60% of Asian individuals demonstrate reduced formation of the active metabolite, leading to attenuated platelet inhibition and potentially higher rates of recurrent ischemic events following TIA or minor stroke [4].
Ticagrelor represents a pharmacologically distinct alternative. As a direct and reversible P2Y12 receptor antagonist, ticagrelor does not require metabolic activation, thereby providing more consistent platelet inhibition across genetic backgrounds. Clinical evidence indicates that ticagrelor in combination with aspirin is more effective than aspirin alone in reducing stroke recurrence and mortality among patients with acute mild-to-moderate ischemic stroke or high-risk TIA [5]. Furthermore, the Platelet Reactivity in Acute Stroke or Transient Ischemic Attack (PRINCE) trial demonstrated lower platelet reactivity with ticagrelor–aspirin compared with clopidogrel–aspirin, particularly among carriers of CYP2C19 LOF variants [6]. These findings raise the possibility that ticagrelor-based DAPT may confer greater protection in genetically susceptible individuals.
Given the differences in pharmacokinetic mechanisms between clopidogrel and ticagrelor, together with the substantial interindividual variability in CYP2C19 activity, a comparative evaluation of their efficacy and safety in CYP2C19 LOF carriers is clinically warranted. The present systematic review and meta-analysis therefore aims to synthesize existing evidence comparing ticagrelor–aspirin and clopidogrel–aspirin in patients with TIA or minor ischemic stroke who harbor CYP2C19 LOF alleles. By examining both recurrent ischemic outcomes and bleeding complications, this study seeks to provide clinically meaningful insights to guide personalized antiplatelet strategies in this high-risk population.
| Materials and Methods | ▴Top |
Study design
The present study employed a systematic review and meta-analysis framework and adhered to the reporting standards established by the PRISMA guidelines.
Data sources and literature search
The literature search was independently performed by two reviewers (INW and INGNY) across PubMed, Scopus, and Google Scholar. All eligible publications from database inception through October 13, 2024, were considered. The search strategy combined the following keywords and Boolean operators: (“ticagrelor”) AND (“clopidogrel”) AND (“stroke” OR “transient ischemic attack” OR “acute ischemic stroke”) AND (“randomized controlled trial” OR “RCT” OR “randomized trial” OR “clinical trial”) AND (“CYP2C19” AND “loss-of-function carrier”).
The search was specifically limited to randomized studies investigating the efficacy and safety of DAPT with ticagrelor plus aspirin compared with clopidogrel plus aspirin in patients with stroke or TIA who carried CYP2C19 LOF alleles. The study selection process is illustrated in Figure 1.
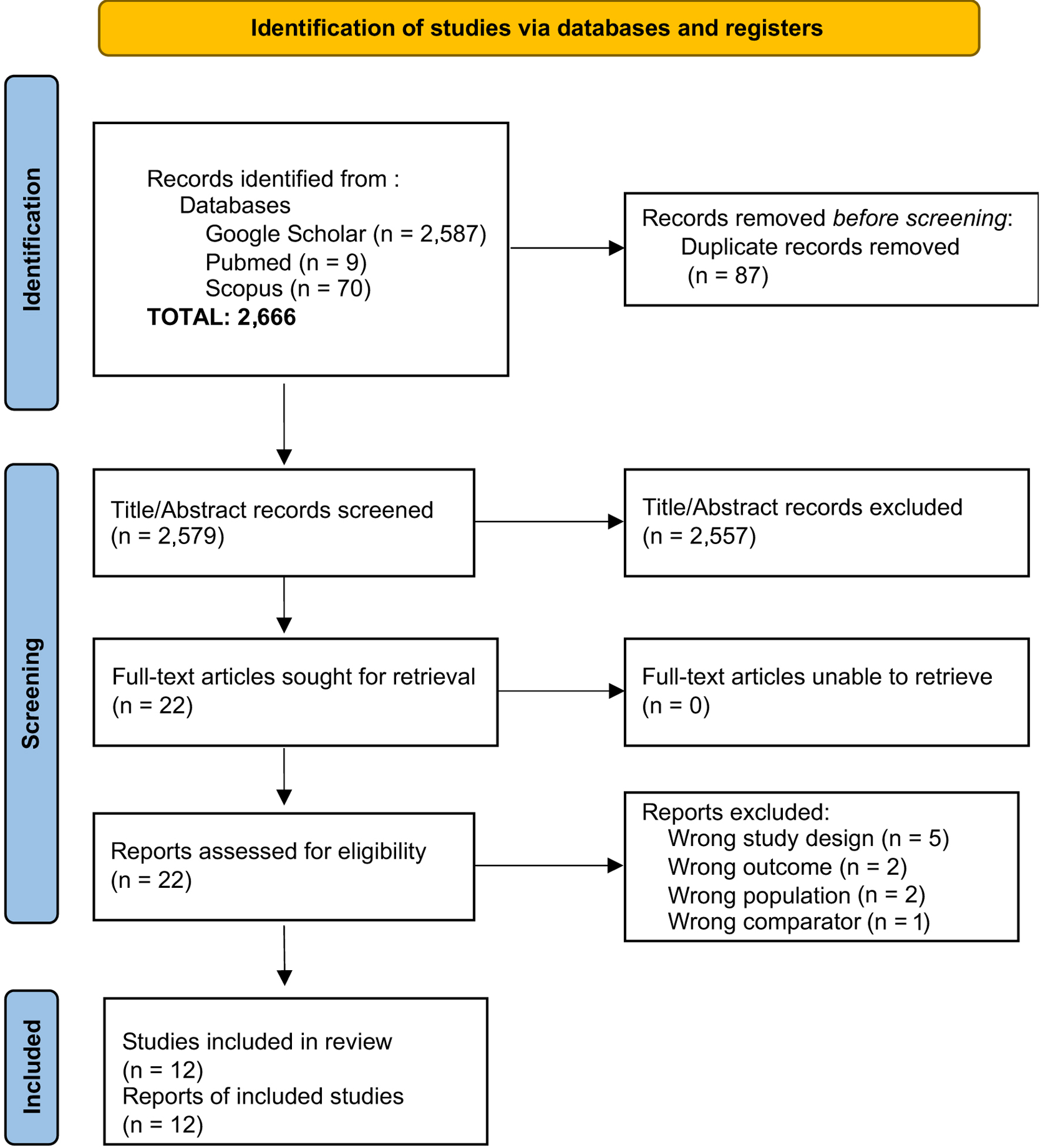 Click for large image | Figure 1. Flow diagram of the study selection process for articles included in the systematic review and meta-analysis. |
Study selection criteria and quality assessment
Study eligibility was defined a priori according to predefined inclusion and exclusion criteria. Eligible studies enrolled adult patients (≥ 18 years) diagnosed with minor ischemic stroke or TIA who received DAPT consisting of either ticagrelor plus aspirin or clopidogrel plus aspirin. Only studies involving CYP2C19 LOF allele carriers, confirmed through genetic testing or documented genotype data, were considered. Furthermore, included articles were required to be randomized controlled trials or clinical trials directly comparing ticagrelor–aspirin with clopidogrel–aspirin in stroke or TIA populations and to report follow-up outcomes related to recurrent stroke, major adverse cardiovascular events (MACEs), or treatment-associated adverse events.
Studies were excluded if they involved patients with prior hemorrhagic stroke or other non-ischemic cerebrovascular conditions, such as hemorrhagic transformation or cerebral venous thrombosis. Additional exclusion criteria comprised participants younger than 18 years or older than 80 years, absence of CYP2C19 genotyping data or lack of specific analysis in LOF carriers, non-randomized or observational study designs, insufficient follow-up duration (< 3 months), incomplete reporting of primary outcomes (e.g., recurrent stroke or major bleeding), and the presence of contraindications to antiplatelet therapy or serious comorbid conditions that would preclude the administration of ticagrelor–aspirin or clopidogrel–aspirin.
The study selection process was conducted independently by two reviewers (INW and INGNY), who screened titles and abstracts for eligibility. Any discrepancies were resolved through discussion, with arbitration by a third reviewer (LPLK) when necessary. Methodological quality and risk of bias were evaluated using the Cochrane Risk of Bias 2 (RoB 2) tool for randomized controlled trials. Disagreements in data extraction or quality assessment were addressed through consensus.
Data extraction
Data extraction was independently performed by two investigators (INW and KDKK) using a standardized data collection form. The following study characteristics were systematically recorded: first author, year of publication, total sample size, study design, clinical diagnosis, details of the intervention regimen, and comparator treatment.
Outcomes
The primary endpoint of this meta-analysis was the incidence of recurrent ischemic or hemorrhagic stroke within 90 days. Secondary endpoints included composite vascular events defined as stroke, TIA, myocardial infarction, or vascular death as well as intracranial hemorrhage and all-cause mortality.
Statistical analysis
Quantitative synthesis was conducted through meta-analytic techniques to pool effect estimates across eligible studies. Treatment effects were calculated as risk ratios (RRs) with corresponding 95% confidence intervals (CIs) for all predefined outcomes. Between-study heterogeneity was assessed using the I2 statistic, with values of approximately 25%, 50%, and 75% interpreted as low, moderate, and high heterogeneity, respectively. Statistical significance was defined as a two-sided P-value ≤ 0.05.
A random-effects model was applied in the presence of moderate to substantial heterogeneity, whereas a fixed-effects model was used when heterogeneity was minimal. All statistical analyses were conducted using Review Manager (RevMan) version 5.1.0 (Cochrane Collaboration, UK). Publication bias was explored through visual inspection of funnel plots when at least 10 studies were available for a given outcome.
Research ethics
This study was conducted in accordance with the ethical guidelines applicable to systematic reviews and meta-analyses. We followed the PRISMA guidelines to ensure transparency and accuracy in our reporting. As this study is a systematic review and does not involve the collection of primary data from human subjects, ethical approval from a review board was not necessary. Additionally, this study has been registered with PROSPERO under registration number CRD420251002523, demonstrating our commitment to transparency and methodological integrity.
| Results | ▴Top |
Study selection
The initial database search yielded 2,666 records. After removing duplicate entries and screening titles and abstracts, potentially eligible articles underwent full-text evaluation. Following the application of predefined inclusion and exclusion criteria, 12 studies met the eligibility requirements and were incorporated into the final systematic review and meta-analysis (Fig. 1).
Risk of bias
Methodological quality was appraised using the Cochrane RoB 2 tool. Among the included studies, two trials were judged to have some concerns regarding potential bias specifically those conducted by Yang et al (2020) [7] and Yang et al (2020) [8]. The detailed risk-of-bias assessment across all domains is summarized in Figure 2 [7–18].
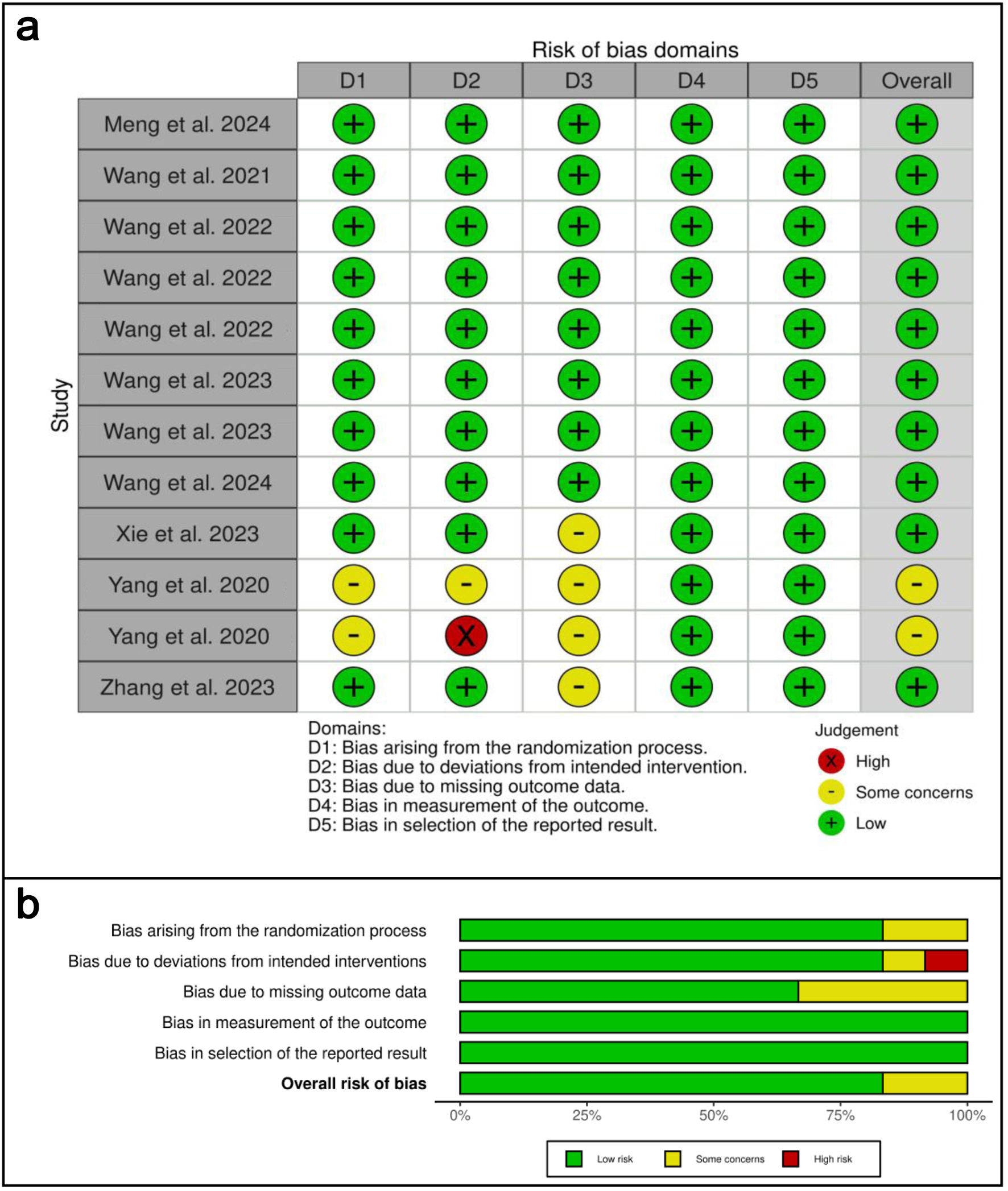 Click for large image | Figure 2. Risk of bias analysis. (a) Traffic-light plot of the risk of bias assessment using RoB 2, illustrating the bias risk results across each domain. (b) Overall risk of bias conclusion using RoB 2, summarizing the risk of bias. |
Study characteristics
The baseline characteristics of the included trials are summarized in Table 1 [7–18]. Overall, 12 studies published between 2020 and 2024 were analyzed, comprising a total of 64,128 patients with TIA who carried CYP2C19 LOF alleles. Individual study sample sizes ranged from 339 to 6,412 participants, reflecting variability in study scale and population characteristics.
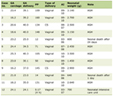 Click to view | Table 1. Characteristics of Study Data |
Meta-analysis results
Primary efficacy outcome: recurrent stroke within 90 days
Twelve trials contributed data to the analysis of 90-day recurrent stroke (Fig. 3) [7–18]. Moderate between-study heterogeneity was observed (I2 = 45%); therefore, a random-effects model was applied. Pooled analysis demonstrated that ticagrelor combined with aspirin was associated with a significantly lower risk of recurrent stroke compared with clopidogrel–aspirin (RR 0.72; 95% CI 0.65–0.80; P < 0.00001).
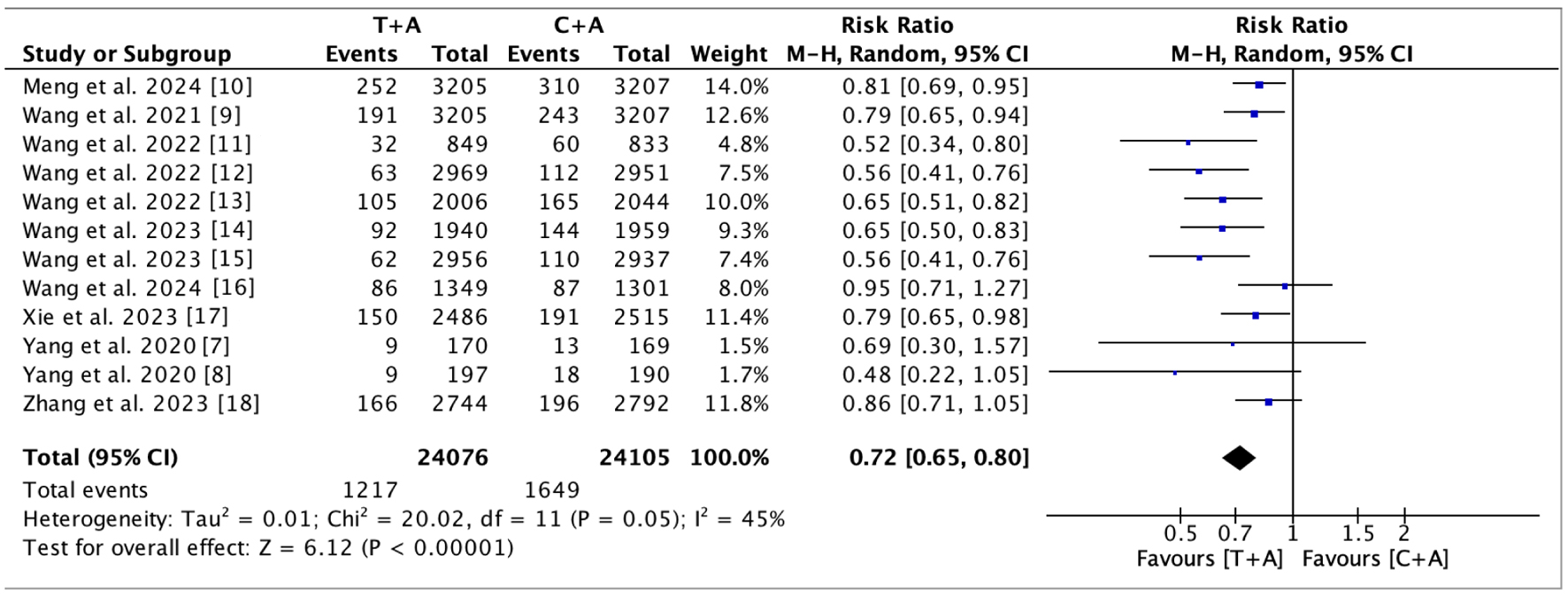 Click for large image | Figure 3. Analysis of primary efficacy outcome. The forest plot demonstrates that the administration of the ticagrelor–aspirin combination significantly reduced the incidence of recurrent stroke within 90 days compared to the clopidogrel–aspirin combination (RR, 0.72; 95% CI, 0.65–0.80; P < 0.00001). CI: confidence interval; RR: risk ratio. |
Composite vascular events
The incidence of composite vascular outcomes was examined across 12 studies (Fig. 4a) [7–18]. No significant heterogeneity was detected (I2 = 0%), and a fixed-effects model was consequently used. The pooled estimate indicated a significant reduction in composite vascular events with ticagrelor–aspirin relative to clopidogrel–aspirin (RR 0.74; 95% CI 0.70–0.79; P < 0.00001).
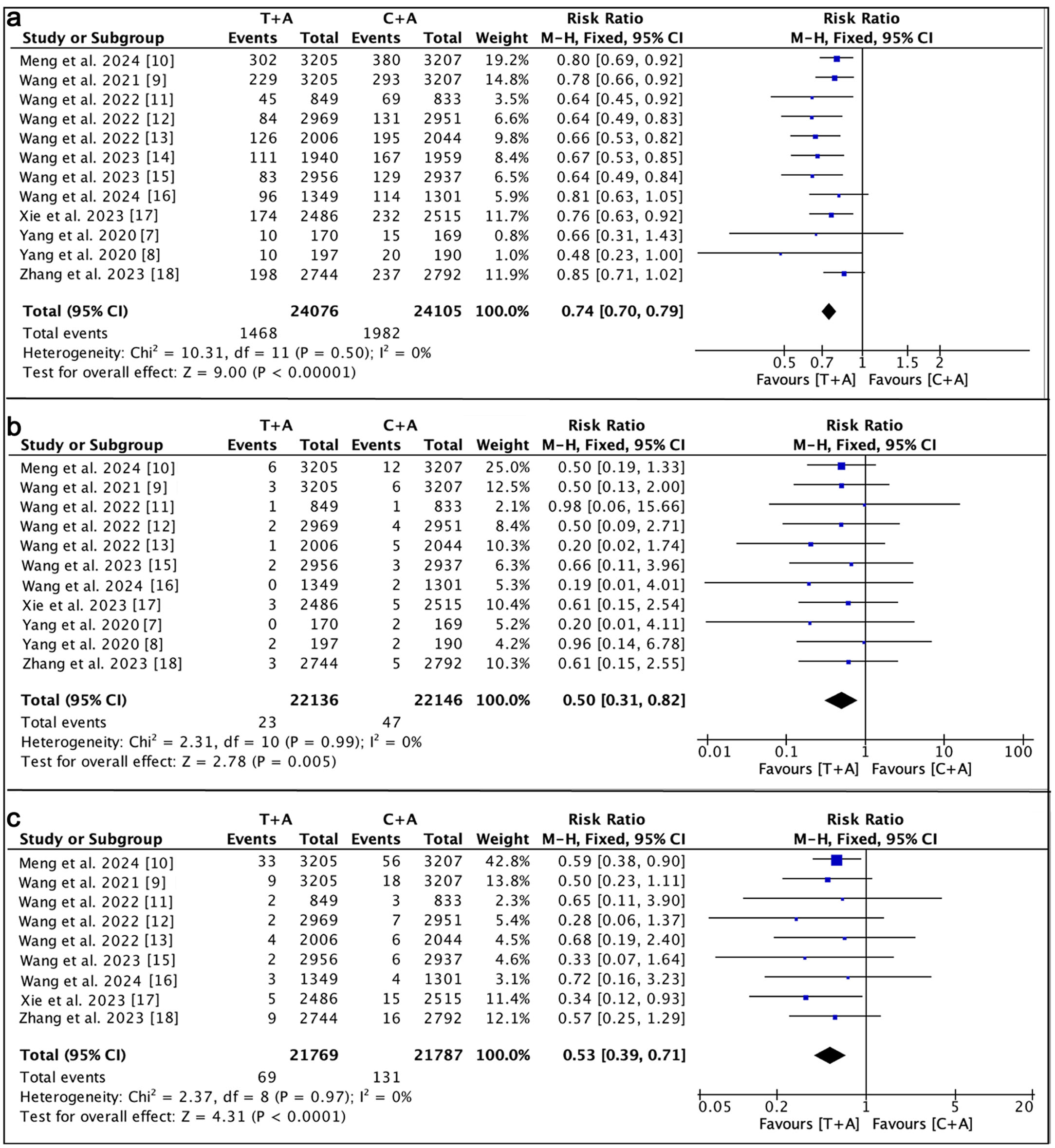 Click for large image | Figure 4. Analysis of secondary safety outcome. (a) The forest plot demonstrates that the administration of the ticagrelor–aspirin combination significantly reduced the incidence of composite vascular events compared to the clopidogrel–aspirin combination (RR, 0.74; 95% CI, 0.70–0.79; P < 0.00001). (b) The forest plot demonstrates that the administration of the ticagrelor–aspirin combination significantly reduced the incidence of intracranial hemorrhage compared to the clopidogrel–aspirin combination (RR, 0.50; 95% CI, 0.31–0.82; P = 0.005). (c) The forest plot demonstrates that the administration of the ticagrelor–aspirin combination significantly reduced the incidence of mortality compared to the clopidogrel–aspirin combination (RR, 0.53; 95% CI, 0.39–0.71; P < 0.0001). CI: confidence interval; RR: risk ratio. |
Intracranial hemorrhage
Eleven studies reported data on intracranial hemorrhage (Fig. 4b). Given the absence of statistical heterogeneity (I2 = 0%), effect estimates were calculated using a fixed-effects model. Ticagrelor–aspirin therapy was associated with a significantly lower incidence of intracranial hemorrhage compared with clopidogrel–aspirin (RR 0.50; 95% CI 0.31–0.82; P = 0.005).
Mortality
Nine studies provided data regarding all-cause mortality (Fig. 4c). With no evidence of heterogeneity (I2 = 0%), a fixed-effects model was applied. The pooled results showed that treatment with ticagrelor–aspirin significantly reduced mortality compared with clopidogrel–aspirin (RR 0.53; 95% CI 0.39–0.71; P < 0.0001).
Publication bias
Between-study heterogeneity was moderate for the primary endpoint (I2 = 45%) and negligible for the secondary outcomes (I2 = 0%). The moderate heterogeneity observed in the primary analysis suggests some variability across studies, potentially arising from differences in study design, patient characteristics, treatment protocols, or outcome assessment methods.
To explore the possibility of publication bias, funnel plots were generated using the meta-bias function in Review Manager version 5.1.0 (Cochrane Collaboration, UK). Visual inspection of the plots demonstrated a largely symmetrical distribution of studies around the pooled effect estimate, indicating no evident publication bias for either the primary or secondary outcomes.
| Discussion | ▴Top |
This systematic review and meta-analysis evaluated the comparative efficacy and safety of ticagrelor–aspirin versus clopidogrel–aspirin in patients with TIA carrying CYP2C19 LOF alleles. The pooled findings demonstrate that ticagrelor-based DAPT is associated with significant reductions in recurrent stroke, composite vascular events, intracranial hemorrhage, and mortality compared with clopidogrel–aspirin. These results support the hypothesis that ticagrelor may provide enhanced vascular protection in genetically susceptible individuals.
The observed differences between the two regimens are biologically plausible. Clopidogrel is a prodrug requiring hepatic activation via the CYP2C19 pathway, and LOF alleles impair the formation of its active metabolite, resulting in reduced platelet inhibition and higher residual thrombotic risk. In contrast, ticagrelor directly and reversibly inhibits the P2Y12 receptor independently of CYP2C19 metabolism, ensuring more consistent antiplatelet effects across genotypes. This pharmacological distinction likely explains the superior outcomes observed among CYP2C19 LOF carriers, particularly in high-risk TIA and minor ischemic stroke populations [9, 10, 19, 20].
Genotype-guided antiplatelet strategies have been explored in cardiovascular settings, with mixed results. While the PHARMCLO trial demonstrated reduced MACEs with genotype-directed therapy in acute coronary syndrome patients carrying CYP2C19 LOF alleles [11], other trials such as POPular Genetics and TAILOR-PCI have reported less consistent findings. In this context, the present analysis provides stroke-specific evidence supporting the potential role of ticagrelor–aspirin in secondary prevention among genetically high-risk TIA patients [12].
Additional pathophysiological mechanisms may further contribute to these findings. Elevated homocysteine levels, which are associated with endothelial dysfunction, oxidative stress, and enhanced platelet activation, may amplify thrombotic risk in susceptible individuals [13–15]. In patients with impaired clopidogrel metabolism, these mechanisms may remain insufficiently controlled, whereas ticagrelor, through its potent and genotype-independent platelet inhibition, may more effectively counteract this prothrombotic state [16]. Beyond platelet inhibition, ticagrelor has also been shown to exert pleiotropic effects, including modulation of vascular tone, inhibition of smooth muscle proliferation, and attenuation of inflammatory responses, which may collectively contribute to improved vascular outcomes [17, 21, 22].
Despite these promising results, clinical application should be guided by an individualized risk–benefit approach. Although ticagrelor offers more potent platelet inhibition, it has been associated with an increased risk of bleeding in certain populations. Therefore, treatment decisions should incorporate not only genetic factors but also patient-specific characteristics, including bleeding risk, comorbidities, age, and concomitant therapies. These considerations are essential to optimize therapeutic outcomes while minimizing adverse events in clinical practice.
Limitations
Despite these encouraging findings, several limitations should be acknowledged. Variability in study design, sample size, baseline patient characteristics, and follow-up durations across the included trials may have contributed to the moderate heterogeneity observed in the primary outcome (I2 = 45%). Although the pooled estimates consistently favored ticagrelor–aspirin, caution is warranted when extrapolating these results to specific clinical subgroups.
Furthermore, while intracranial hemorrhage was reduced in the pooled analysis, ticagrelor remains associated with an increased bleeding tendency in broader clinical practice, emphasizing the importance of careful patient selection and individualized risk–benefit assessment. Clinical decision-making should therefore incorporate not only genetic status but also patient-specific factors, including baseline bleeding risk, comorbidities, age, and concomitant medications.
In addition, sex-related differences may influence stroke characteristics, treatment response, and clinical outcomes through biological and clinical mechanisms, including hormonal effects, platelet reactivity, and variations in comorbidity profiles. However, the lack of sex-stratified data in the included studies precluded subgroup analyses based on gender, limiting further exploration of these differences.
Moreover, the lack of detailed reporting on cause-specific mortality (neurological versus non-neurological) across the included studies limited our ability to comprehensively evaluate the safety profile of the interventions. Finally, the absence of patient-level data restricted more granular analyses, including stratification by stroke subtype and other clinically relevant variables.
Future research
Future research should prioritize large, adequately powered randomized controlled trials specifically designed to evaluate genotype-guided antiplatelet therapy in patients with TIA and minor ischemic stroke. In particular, studies incorporating stratified analyses based on sex, stroke subtype (e.g., lacunar versus non-lacunar infarction), and baseline clinical risk factors are needed to better define patient subgroups that derive the greatest benefit from ticagrelor-based therapy. This is especially relevant given the distinct pathophysiological mechanisms, clinical characteristics, and prognostic trajectories associated with lacunar versus non-lacunar infarction, which may differentially influence treatment response and outcomes.
In addition, future investigations should include detailed reporting of cause-specific mortality to improve understanding of the safety profile of different antiplatelet regimens. Integration of genetic, clinical, and imaging parameters may further enhance precision medicine approaches in secondary stroke prevention.
Moreover, exploration of additional biological modifiers, including pathways related to homocysteine metabolism and inflammatory biomarkers, may provide further insights into individualized therapeutic strategies and optimize long-term vascular outcomes in high-risk populations.
Conclusion
This systematic review and meta-analysis indicates that ticagrelor combined with aspirin may offer superior efficacy and overall safety compared with clopidogrel–aspirin among patients with TIA who carry CYP2C19 LOF alleles. Ticagrelor-based DAPT was associated with significant reductions in 90-day recurrent stroke, composite vascular events, intracranial hemorrhage, and mortality. These findings highlight the growing importance of pharmacogenetic considerations in optimizing antiplatelet strategies, particularly in individuals with impaired clopidogrel metabolism. While the results suggest that ticagrelor–aspirin may represent a more effective approach for this genetically high-risk population, individualized evaluation of bleeding risk remains crucial in clinical practice. Future large-scale, genotype-guided randomized controlled trials are needed to confirm these observations and to further refine precision-based antiplatelet therapy in patients with TIA.
Acknowledgments
The authors would like to express their sincere gratitude to the Faculty of Medicine, Universitas Pendidikan Ganesha, for their guidance and academic support during the preparation of this manuscript.
Financial Disclosure
The authors received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The authors declare that there is no conflict of interest regarding the publication of this article.
Informed Consent
Not applicable. This study is a systematic review and meta-analysis based on previously published data and did not involve direct interaction with human participants.
Author Contributions
I Nyoman Windiana conceptualized and designed the study, developed the research protocol, conducted the literature search and study selection, performed data extraction and statistical analyses, and drafted the manuscript. Muhammad Iqhrammullah contributed to data interpretation, manuscript editing, and critical revision of the final version. Luh Putu Lina Kamelia assisted in the literature search, study selection, data extraction, and critically revised the manuscript for important intellectual content. I Nyoman Gede Narendra Yanakusuma, Komang Diah Kurnia Kesumaputri, and I Gusti Agung Ayu Pramasinthya Aguseny Yudana contributed to data extraction, quality assessment of the included studies, and statistical analysis. All authors reviewed and approved the final manuscript.
Data Availability
The authors declare that data supporting the findings of this study are available within the article.
| References | ▴Top |
- Johnston SC, Easton JD, Farrant M, Barsan W, Conwit RA, Elm JJ, Kim AS, et al. Clopidogrel and aspirin in acute ischemic stroke and high-risk TIA. N Engl J Med. 2018;379(3):215-225.
doi pubmed - Powers WJ, Rabinstein AA, Ackerson T, Adeoye OM, Bambakidis NC, Becker K, Biller J, et al. Guidelines for the early management of patients with acute ischemic stroke: 2019 update to the 2018 guidelines for the early management of acute ischemic stroke: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke. 2019;50(12):e344-e418.
doi pubmed - Pan Y, Jing J, Chen W, Meng X, Li H, Zhao X, Liu L, et al. Risks and benefits of clopidogrel-aspirin in minor stroke or TIA: Time course analysis of CHANCE. Neurology. 2017;88(20):1906-1911.
doi pubmed - Brown SA, Pereira N. Pharmacogenomic impact of CYP2C19 variation on clopidogrel therapy in precision cardiovascular medicine. J Pers Med. 2018;8(1):8.
doi pubmed - Navarese EP, Khan SU, Kolodziejczak M, Kubica J, Buccheri S, Cannon CP, Gurbel PA, et al. Comparative efficacy and safety of oral P2Y(12) inhibitors in acute coronary syndrome: network meta-analysis of 52 816 patients from 12 randomized trials. Circulation. 2020;142(2):150-160.
doi pubmed - Li M, Yang Q, Shi J, Zhang X, Lin H, Ge F. Ticagrelor plus aspirin in patients with minor ischemic stroke and transient ischemic attack: a network meta-analysis. BMC Neurol. 2023;23(1):303.
doi pubmed - Yang Y, Chen W, Pan Y, Yan H, Meng X, Liu L, Wang Y, et al. Effect of ticagrelor versus clopidogrel on platelet reactivity measured by thrombelastography in patients with minor stroke or TIA. Aging (Albany NY). 2020;12(20):20085-20094.
doi pubmed - Yang Y, Chen W, Pan Y, Yan H, Meng X, Liu L, Wang Y, et al. Ticagrelor is superior to clopidogrel in inhibiting platelet reactivity in patients with minor stroke or TIA. Front Neurol. 2020;11:534.
doi pubmed - Wang Y, Meng X, Wang A, Xie X, Pan Y, Johnston SC, Li H, et al. Ticagrelor versus clopidogrel in CYP2C19 loss-of-function carriers with stroke or TIA. N Engl J Med. 2021;385(27):2520-2530.
doi pubmed - Meng X, Wang A, Tian X, Johnston C, Li H, Bath PM, Xu Q, et al. One-year outcomes of early therapy with ticagrelor vs clopidogrel in CYP2C19 loss-of-function carriers with stroke or TIA trial. Neurology. 2024;102(3):e207809.
doi pubmed - Wang A, Meng X, Tian X, Johnston SC, Li H, Bath PM, Zuo Y, et al. Effect of hypertension on efficacy and safety of ticagrelor-aspirin versus clopidogrel-aspirin in minor stroke or transient ischemic attack. Stroke. 2022;53(9):2799-2808.
doi pubmed - Wang C, Jing J, Jia W, Meng X, Wang A, Xu Q, Zhang X, et al. Efficacy and safety of ticagrelor versus clopidogrel in acute minor stroke or transient ischemic attack patients with different intracranial artery stenosis statuses: a subgroup analysis of CHANCE-2, a randomized, double-blind, controlled trial. SSRN [Preprint]. 2022. Available from: https://doi.org/10.2139/ssrn.4054464.
- Wang A, Xie X, Tian X, Johnston SC, Li H, Bath PM, Zuo Y, et al. Ticagrelor-aspirin versus clopidogrel-aspirin among CYP2C19 loss-of-function carriers with minor stroke or transient ischemic attack in relation to renal function: a post hoc analysis of the CHANCE-2 trial. Ann Intern Med. 2022;175(11):1534-1542.
doi pubmed - Wang A, Meng X, Tian X, Zuo Y, Bath PM, Li H, Xie X, et al. Ticagrelor aspirin vs clopidogrel aspirin in CYP2C19 loss-of-function carriers with minor stroke or TIA stratified by risk profile. Neurology. 2023;100(5):e497-e504.
doi pubmed - Wang C, Jia W, Jing J, Meng X, Wang A, Xu Q, Zhang X, et al. Ticagrelor versus clopidogrel in minor stroke or transient ischemic attack with intracranial artery stenosis: a post hoc analysis of CHANCE-2. J Am Heart Assoc. 2023;12(21):e031611.
doi pubmed - Wang A, Tian X, Xie X, Li H, Bath PM, Jing J, Lin J, et al. Differential effect of ticagrelor versus clopidogrel by homocysteine levels on risk of recurrent stroke: a post hoc analysis of the CHANCE-2 trial. CMAJ. 2024;196(5):E149-E156.
doi pubmed - Xie X, Johnston SC, Wang A, Xu Q, Bath PM, Pan Y, Li H, et al. Association of CYP2C19 loss-of-function metabolizer status with stroke risk among Chinese patients treated with ticagrelor-aspirin vs clopidogrel-aspirin: a prespecified secondary analysis of a randomized clinical trial. JAMA Netw Open. 2023;6(6):e2317037.
doi pubmed - Zhang J, Wang A, Tian X, Meng X, Xie X, Jing J, Lin J, et al. Impact of body mass index on efficacy and safety of ticagrelor versus clopidogrel in patients with minor stroke or transient ischemic attack. CMAJ. 2023;195(26):E897-E904.
doi pubmed - Jiang XL, Samant S, Lesko LJ, Schmidt S. Clinical pharmacokinetics and pharmacodynamics of clopidogrel. Clin Pharmacokinet. 2015;54(2):147-166.
doi pubmed - Johnston SC, Elm JJ, Easton JD, Farrant M, Barsan WG, Kim AS, Lindblad AS, et al. Time course for benefit and risk of clopidogrel and aspirin after acute transient ischemic attack and minor ischemic stroke. Circulation. 2019;140(8):658-664.
doi pubmed - Triska J, Maitra N, Deshotels MR, Haddadin F, Angiolillo DJ, Vilahur G, Jneid H, et al. A comprehensive review of the pleiotropic effects of ticagrelor. Cardiovasc Drugs Ther. 2024;38(4):775-797.
doi pubmed - Bhatia K, Jain V, Aggarwal D, Vaduganathan M, Arora S, Hussain Z, Uberoi G, et al. Dual antiplatelet therapy versus aspirin in patients with stroke or transient ischemic attack: meta-analysis of randomized controlled trials. Stroke. 2021;52(6):e217-e223.
doi pubmed
This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (CC BY 4.0), which permits unrestricted use, distribution, and reproduction in any medium, including commercial use, provided the original work is properly cited.
Journal of Neurology Research is published by Elmer Press Inc.