| Journal of Neurology Research, ISSN 1923-2845 print, 1923-2853 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Neurol Res and Elmer Press Inc |
| Journal website https://jnr.elmerpub.com |
Case Report
Volume 16, Number 2, June 2026, pages 115-121
Transient Akinetic Mutism Following Partial Corpus Callosum Infarction After Rupture of a Distal Anterior Cerebral Artery Microaneurysm
Seigo Kimuraa, c, Daiji Ogawaa, Ryokichi Yagib, Norihito Fukawaa, Keiichi Yamadaa, Hirokatsu Taniguchia, Masahiko Wanibuchib
aKouzenkai Yagi Neurosurgical Hospital, 2-12-13 Higashiimazato, Higashinari-ku, Osaka, Osaka 537-0011, Japan
bOsaka Medical and Pharmaceutical University Hospital, 2-7 Daigakumachi, Takatsuki City, Osaka 569-8686, Japan
cCorresponding Author: Seigo Kimura, Kouzenkai Yagi Neurosurgical Hospital, 2-12-13 Higashiimazato, Higashinari-ku, Osaka, Osaka 537-0011, Japan
Manuscript submitted February 17, 2026, accepted April 9, 2026, published online May 4, 2026
Short title: AM Following CC Infarction After ACA AN Rupture
doi: https://doi.org/10.14740/jnr1106
| Abstract | ▴Top |
Aneurysms located in the distal anterior cerebral artery (ACA) are rare. Cases in which rupture results in subarachnoid hemorrhage (SAH) with a localized medial hemispheric sulcal hematoma and cerebral infarction predominantly involving the corpus callosum (CC) are uncommon. Herein, we report a case of ruptured small ACA aneurysm with SAH accompanied by a localized medial hemispheric sulcal hematoma. A 54-year-old female patient collapsed in the bathroom, and she was rushed to our hospital. Head computed tomography scan revealed an SAH accompanied by a localized medial hemispheric sulcal hematoma. Computed tomography angiography (CTA) failed to identify an aneurysm. Subsequent cerebral angiography revealed a 1.5-mm aneurysm at the junction of the right pericallosal and callosomarginal artery. Clipping was subsequently performed. Diffusion-weighted imaging on magnetic resonance imaging revealed a cerebral infarction predominantly located in the CC. The patient did not move voluntarily; hence, she was diagnosed with akinetic mutism (AM). Her symptoms gradually improved, and she was transferred to a rehabilitation hospital. The patient’s medial callosal artery or CC might have been compressed by the hematoma, causing CC infarction. In cases of CC infarction, symptoms such as AM may develop. However, CC infarction alone could not be identified as the cause. Further, multiple factors, such as functional impairment of the frontal lobe and cingulate gyrus associated with SAH, might have played a role. This case emphasizes that distal ACA microaneurysms may be occult on CTA and that careful interpretation of localized sulcal hematoma distribution is essential.
Keywords: Corpus callosum infarction; Pericallosal artery; Callosomarginal artery; Akinetic mutism; Subarachnoid hemorrhage
| Introduction | ▴Top |
Distal anterior cerebral artery (ACA) aneurysms are rare [1]. Cases in which rupture causes subarachnoid hemorrhage (SAH) with a localized medial hemispheric sulcal hematoma and cerebral infarction predominantly involving the corpus callosum (CC) are uncommon and clinically distinctive. Some reports have shown an association between CC infarction and akinetic mutism (AM) [2, 3]. However, the clinical symptoms of CC infarction vary, and its association with CC infarction and AM remains controversial. Herein, we report a case of ruptured small ACA aneurysm with SAH accompanied by a localized medial hemispheric sulcal hematoma. In this case, clipping was performed, and postoperative magnetic resonance imaging (MRI) revealed a CC infarction.
| Case Report | ▴Top |
Investigations
A 54-year-old female patient complained of not feeling well at around 6 a.m. and went to the bathroom. However, her husband found her collapsed in the bathroom, and she was rushed to our hospital. The patient’s medical history and family history were unremarkable. Upon arrival, her Glasgow Coma Scale score was 8 (E3, V1, M4). Neurological examination revealed left facial nerve palsy, mild dysarthria, left hemiparesis, and left hemispatial neglect. No evident paralysis of the limbs was observed. However, speech was not observed. The patient’s blood pressure and pulse rate were 167/100 mm Hg and 70/min, respectively.
Diagnosis
Head computed tomography (CT) scan revealed an SAH (Fisher group 3) accompanied by a localized medial hemispheric sulcal hematoma (Fig. 1a, b). Head computed tomography angiography (CTA) revealed no aneurysm in the distal ACA. Cerebral angiography revealed a small cerebral aneurysm at the junction of the right pericallosal and callosomarginal artery (P-CA AN) (Fig. 2). The longest diameter of the cerebral aneurysm was 1.5 mm, the shortest diameter was 1.3 mm, and the height was 1.2 mm. Based on the CTA in three directions, the aneurysm was not located at the thickest part of the hematoma but was contained within the hematoma (Figs. 3a–c). Therefore, it was determined to be the ruptured aneurysm. Since the aneurysm was small and not located in the thickest part of the hematoma, clipping was performed to confirm whether it was actually a ruptured aneurysm.
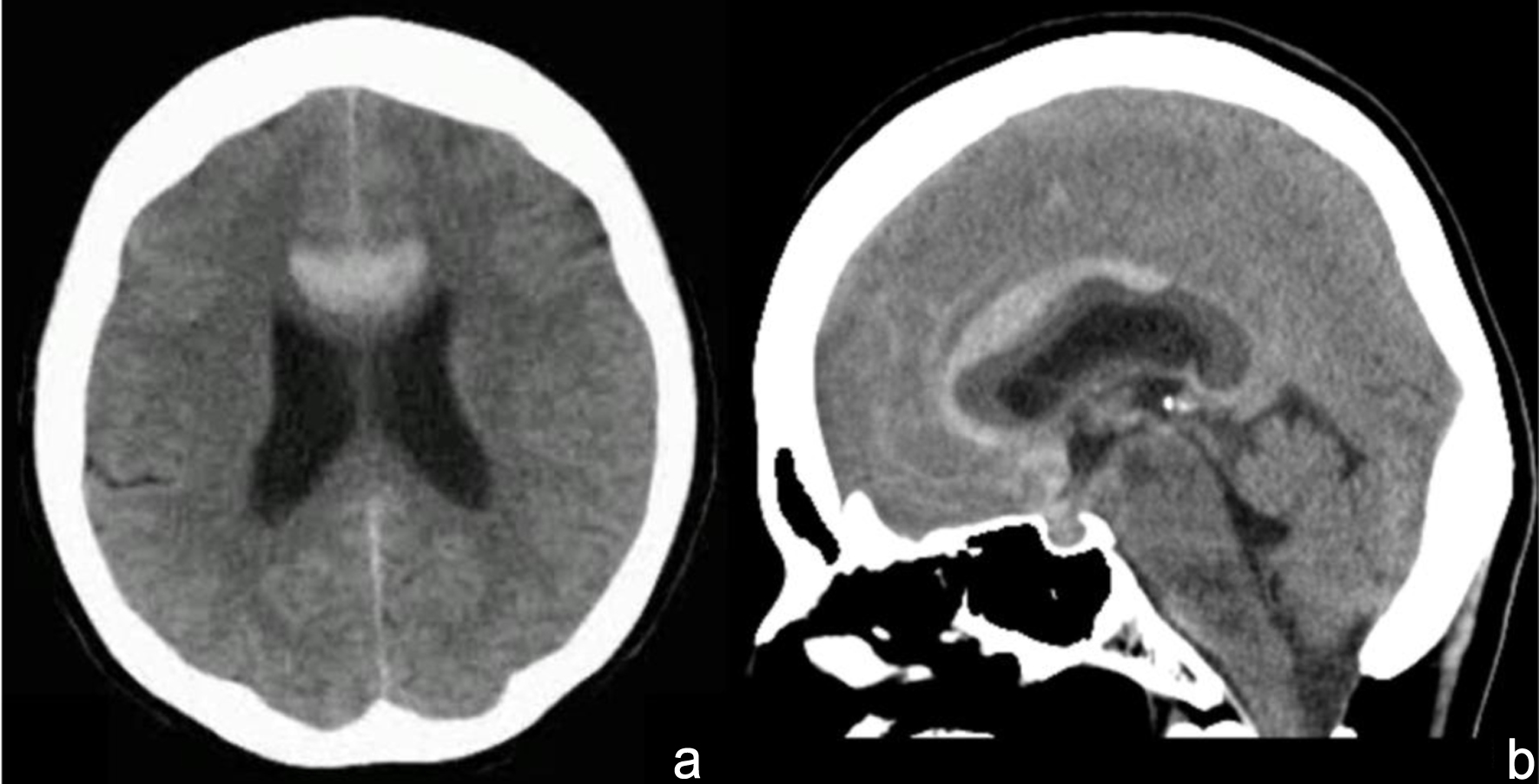 Click for large image | Figure 1. Head computed tomography reveals a subarachnoid hemorrhage accompanied by a localized medial hemispheric sulcal hematoma. (a) Axial image. (b) Sagittal image. |
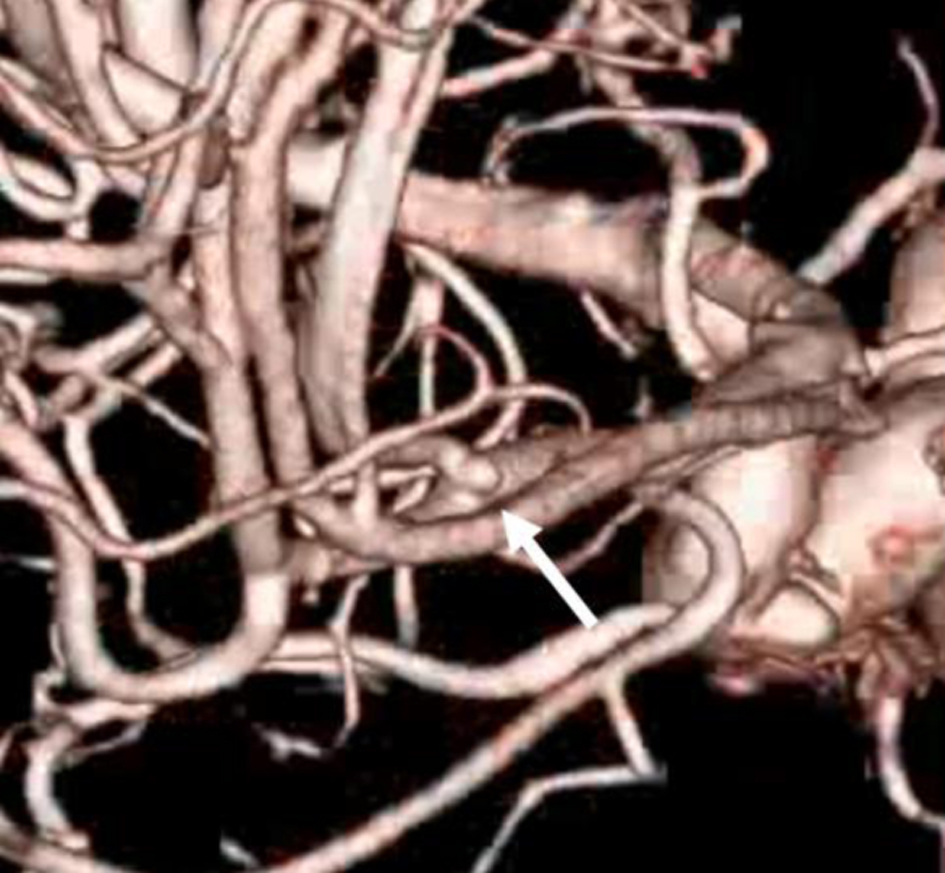 Click for large image | Figure 2. Cerebral angiography reveals a small cerebral aneurysm at the junction of the right pericallosal and the callosomarginal artery (white arrow). |
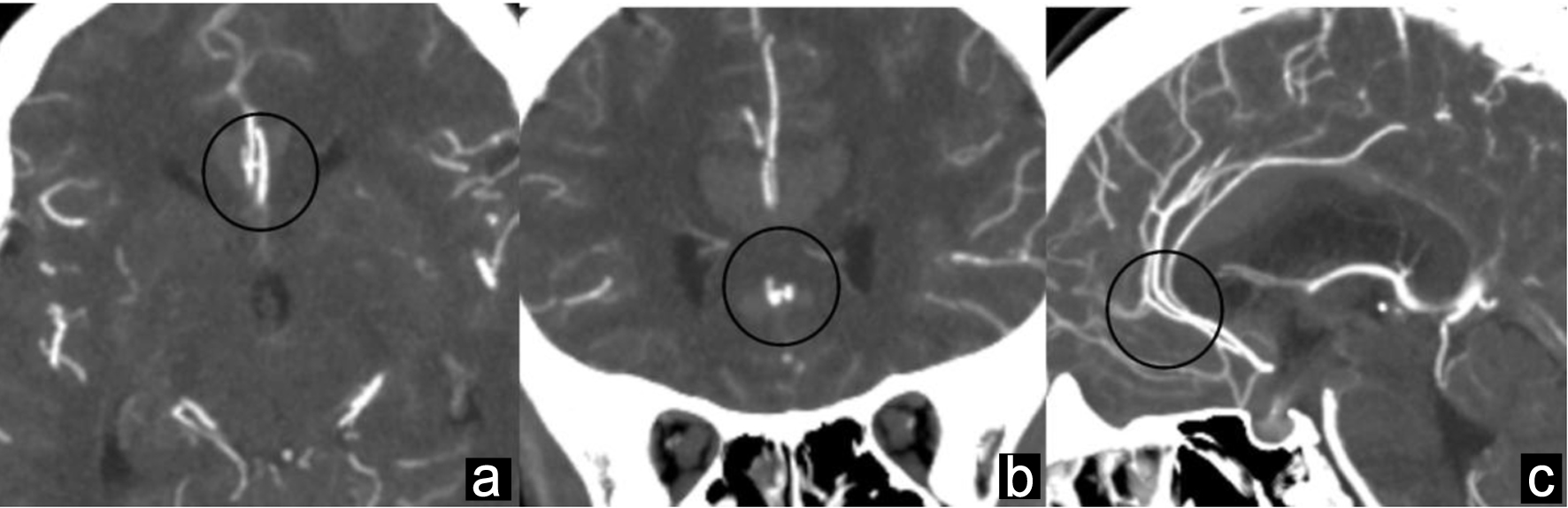 Click for large image | Figure 3. Based on CTA in three directions, the aneurysm is not located at the thickest part of the hematoma but is contained within the hematoma (black circle). (a) Axial CTA image. (b) Coronal CTA image. (c) Sagittal CTA image. CTA: computed tomography angiography. |
Treatment
Under general anesthesia, the patient was placed in the supine position, and a bicoronal skin incision was made. A right unilateral frontal craniotomy across the sagittal suture was performed. Ventricular drainage was conducted via right anterior horn puncture, and cerebrospinal fluid was drained from the ventricle as necessary. A C-shaped dural incision was made at the base of the superior sagittal sinus. The interhemispheric fissure was separated, and the right middle internal frontal artery was traced to reach the right callosomarginal artery. The tissue surrounding the A2 segment proximal to the pericallosal-callosomarginal artery junction was resected without touching the aneurysm. While performing hemostasis of the tissues surrounding the A2 segment, aneurysmal pre-rupture was observed. Thus, a temporary clip was applied to the A2 segment to stop the bleeding. In that state, the callosomarginal artery, pericallosal artery, and ruptured aneurysm were exposed. The aneurysm wall was extremely thin, and the black thrombus was visible within it (Fig. 4a). The aneurysm was clipped using a SUGITA straight mini-clip (6 mm) (17-001-81) (Fig. 4b). The temporary clip was removed, and blood flow was restored. The blood flow was stopped for 17 min. Indocyanine green angiography was performed, and the callosomarginal artery, pericallosal artery, right A2 segment, and left pericallosal artery were visualized (Fig. 4c). No contrast agent flowed into the aneurysm, and the microscopic surgery was completed. The wound was closed according to standard procedure, and the surgery was completed.
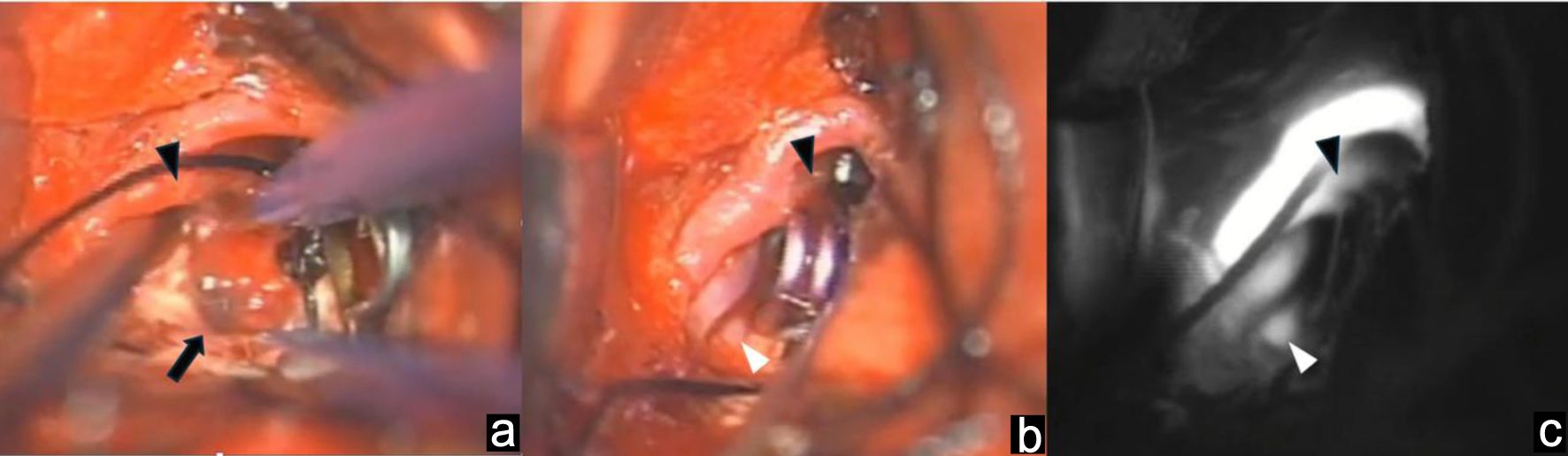 Click for large image | Figure 4. Clipping is performed. (a) The aneurysm wall was extremely thin, and the black thrombus was visible within it. (b) The aneurysm was clipped. (c) Indocyanine green angiography was performed, and the callosomarginal artery, pericallosal artery, right A2 segment, and left pericallosal artery were visualized. Black arrow: aneurysm. Black arrow head: callosomarginal artery. White arrow head: pericallosal artery. |
Head CT scan after surgery revealed no obvious postoperative bleeding. The intubation tube was removed a day after the surgery. Further, diffusion-weighted image of MRI revealed a cerebral infarction predominantly located in the CC (Fig. 5a–c). Magnetic resonance angiography (MRA) revealed no evident occlusion of the main artery. On day 8, cerebral angiography revealed that the aneurysm had been successfully clipped, and no obvious spasm was observed. On day 14, fluid-attenuated inversion recovery of sagittal MRI section revealed a high intensity area in the anterior part of the CC (Fig. 6a–f). This was consistent with the location of the localized medial hemispheric sulcal hematoma on head CT scan upon admission. No spontaneous speech was observed after surgery. The patient did not exhibit any obvious paralysis. However, she did not move voluntarily and was, therefore, diagnosed with AM. On day 17, she walked with parallel bars during rehabilitation. On day 18, she was able to operate a smartphone. On day 22, spontaneous speech was observed, and walking with a walker became possible. There were no obvious symptoms of callosal disconnection syndrome based on the evaluation performed on day 23. Gradually, speech improved, energy levels increased, and facial expressions appeared to be more positive.
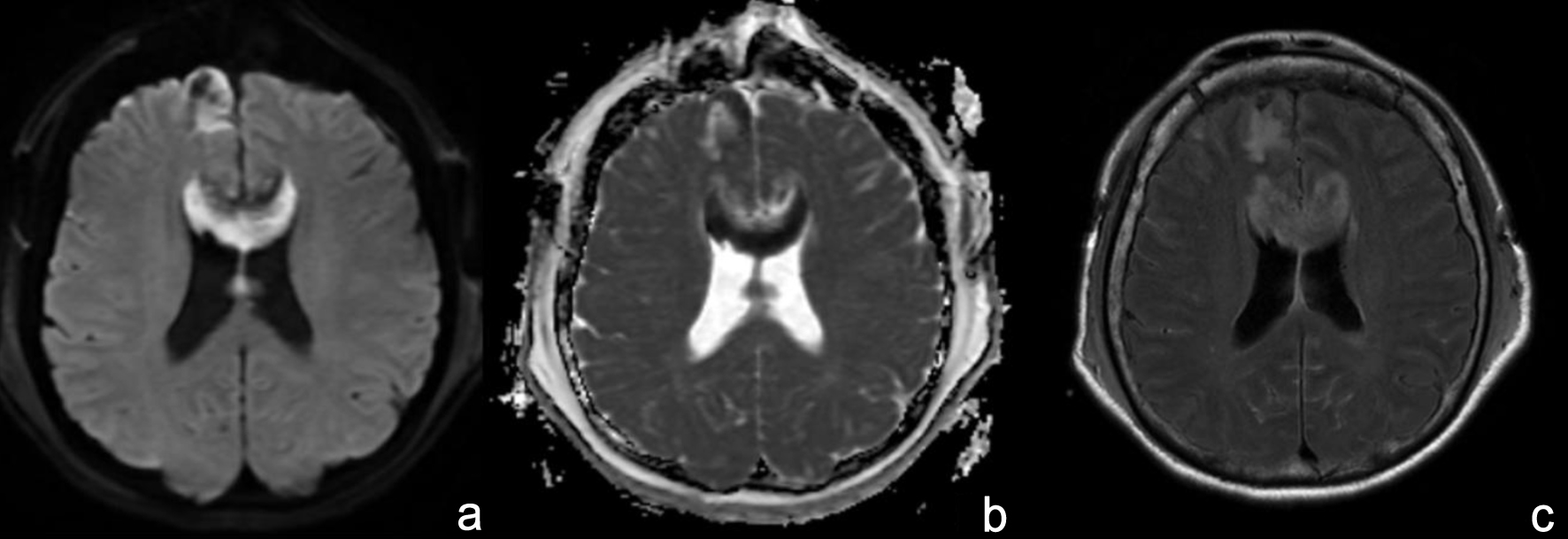 Click for large image | Figure 5. On the day after the surgery, diffusion-weighted imaging on MRI revealed a cerebral infarction predominantly located in the corpus callosum. (a) Axial image of diffusion-weighted image. (b) Axial image of apparent diffusion coefficient map. (c) Axial image of fluid-attenuated inversion recovery. |
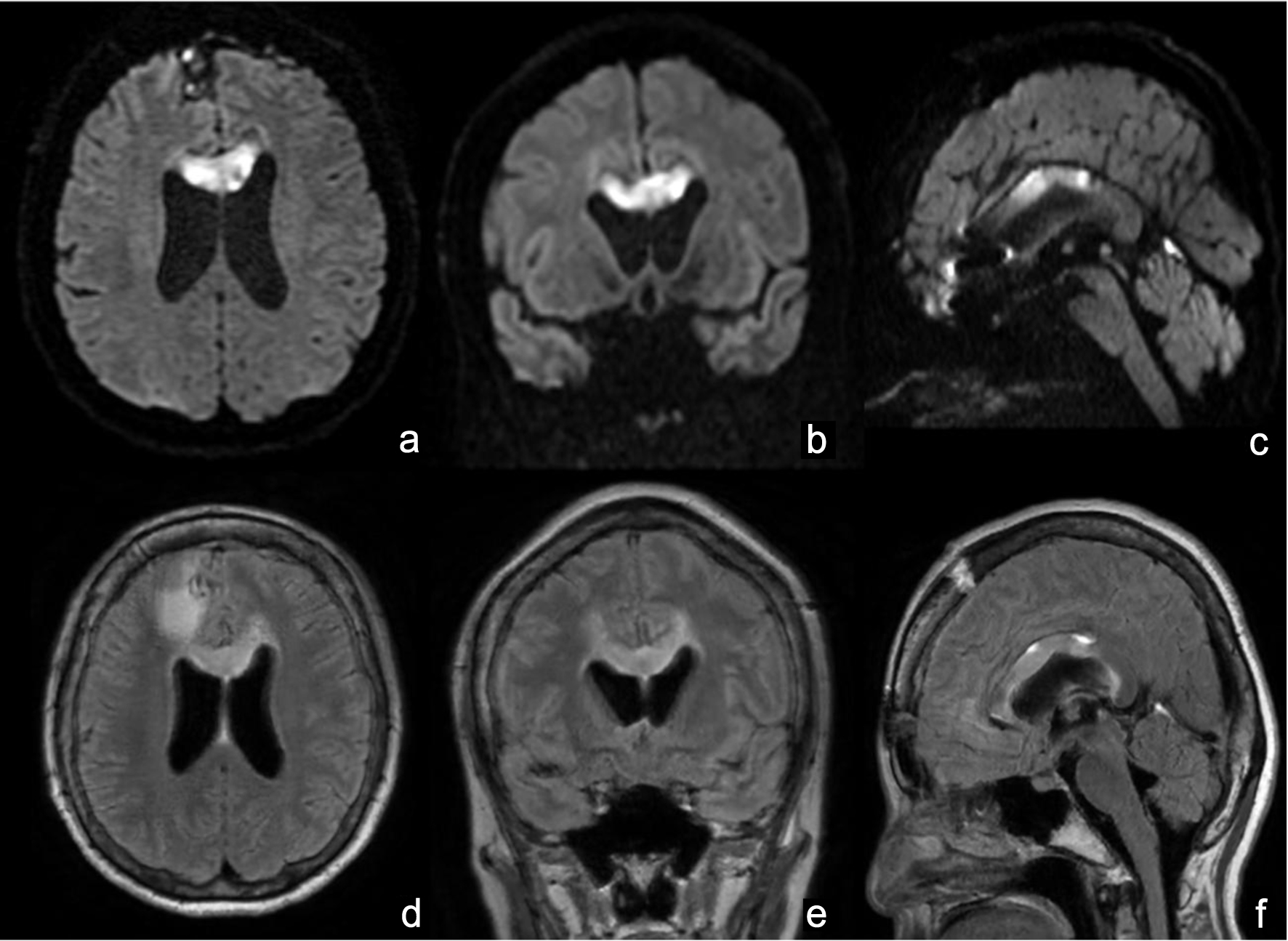 Click for large image | Figure 6. On day 14, diffusion-weighted image and fluid-attenuated inversion recovery on MRI revealed a high intensity area in the anterior part of the corpus callosum. (a) Axial image of diffusion-weighted image. (b) Coronal image of diffusion-weighted image. (c) Sagittal image of diffusion-weighted image. (d) Axial image of fluid-attenuated inversion recovery. (e) Coronal image of fluid-attenuated inversion recovery. (f) Sagittal image of fluid-attenuated inversion recovery. MRI: magnetic resonance imaging. |
Follow-up and outcomes
On day 37, the patient, who had a modified Rankin Scale score of 3, was transferred to a rehabilitation hospital. At the 6-month follow-up, she had improved to a modified Rankin Scale score of 1 and was being followed up as an outpatient.
| Discussion | ▴Top |
Distal ACA aneurysms are rare, accounting for approximately 5% of all cerebral aneurysms, and most of them are referred to as P-CA AN. Distal ACA aneurysms are often small and more likely to rupture before they increase in size. In addition, small aneurysms may be missed [1]. The aneurysm in our patient measured only 1.5 mm at its maximum diameter. Distal ACA aneurysms of approximately 1 mm are rarely described in detail in the literature. Importantly, CTA failed to identify the aneurysm preoperatively. The lesion was partially concealed by the overlapping course of the contralateral ACA, making detection particularly challenging in the acute hemorrhagic setting. In this case, careful assessment of the localized medial sulcal hematoma distribution was crucial in suspecting a distal ACA rupture and prompting diagnostic angiography. Therefore, distal ACA microaneurysms should be suspected in cases of localized medial sulcal hematoma even when initial CTA findings are negative.
The inner surface of both cerebral hemispheres and most of the CC are perfused by the distal ACA [1]. The CC is perfused by the pericallosal artery in the anterior 4/5 and by the posterior pericallosal artery, a branch of the posterior cerebral artery, in the posterior 1/5 [1, 3, 4]. Due to anastomoses between the two vessels near the splenium, occlusion of either vessel does not necessarily lead to cerebral infarction [5]. The subcallosal artery and the median callosal artery, which branch off from the anterior communicating artery, may primarily supply the CC. Occlusion of these arteries may lead to CC infarction [6]. In our case, CC infarction was highly consistent with the course of the median callosal artery. In addition, temporary clipping of the A2 segment for 17 min may also have contributed to the development of CC infarction. In the acute phase of SAH, hypercoagulability and impaired microcirculation are frequently observed. Under such conditions, prolonged temporary occlusion may reduce perfusion of small perforating arteries such as the median callosal artery, even when major arterial flow appears preserved after clip removal. Therefore, transient ischemia caused by temporary clipping, combined with hematoma-related compression and microvascular vulnerability in the acute SAH phase, may have contributed to the partial CC infarction in this case. Although intraoperative manipulation cannot be completely excluded, no angiographic evidence of major arterial occlusion was observed, supporting a multifactorial rather than purely iatrogenic mechanism.
The symptoms of CC infarction include cognitive impairment, mental disorder, sleep disorder, aphasia, sensory abnormality, central facial paralysis, limb paralysis, hemianopia, ataxia, and alien-hand syndrome. The clinical manifestations of callosal infarction are complex, and there is a lack of specificity because of its own physiological structure and function. Further, adjacent locations are frequently involved after ischemia [7]. Sun et al have reported that the incidence of alien-hand syndrome was extremely low at 0.8%. The symptoms of callosal disconnection syndrome, including alien-hand syndrome, do not necessarily appear in all cases and are more likely to appear in cases of total CC infarction [3]. AM is a condition in which the patient is alert and conscious but remains motionless and completely unable to speak. This condition may be caused by a sudden disruption of communication between the left and right hemispheres of the brain [3]. In our case, the patient presented with SAH due to rupture of an ACA aneurysm, accompanied by CC infarction and AM. Watanabe et al [3] reported the occurrence of AM in a patient with isolated CC infarction. Moreover, another report has shown similar symptoms caused by acute anterior CC damage [2]. By contrast, AM has also been found to occur as a result of bilateral cingulate gyrus damage [3]. Therefore, the mechanism of AM onset in this case cannot be attributed solely to CC infarction. Multiple factors, such as functional impairment of the frontal lobe and cingulate gyrus associated with SAH, may have been involved. The mechanism of AM in this case is likely multifactorial, involving not only partial CC infarction but also transient ischemia of small perforating arteries, mass effect from the localized sulcal clot, and functional disruption of the frontal–cingulate–callosal network following SAH.
Previous reports have found three cases of SAH predominantly accompanied by CC infarction (Table 1). One of the three cases involved aneurysm rupture at the A1, A2 junctions, as described by Takahashi et al [6]. The other two cases were aneurysm ruptures in the distal ACA, as described in the study of Alnaami et al [8] and Abbuehl et al [9]. Two patients with distal ACA aneurysm rupture presented with localized medial hemispheric sulcal hematoma. Among these, Alnaami et al [8] reported a case of CC infarction involving only the body of CC, with no association between CC infarction and spasm. Takahashi et al [6] and Abbuehl et al [9] presented cases of ACA spasm and resulting infarction of the whole CC. In particular, Takahashi et al [6] have reported a case in which spasm-induced blood flow loss in the median callosal artery may have caused infarction of the entire CC. In the case reported by Abbuehl et al [9], callosal disconnection syndrome was observed and was considered to be caused by infarction extending throughout the whole CC. In two cases of distal ACA aneurysm rupture [8, 9], the patients developed AM after SAH onset. In the case reported by Alnaami et al [8], AM improved after 3 months. Meanwhile, in the case reported by Abbuehl et al [9], improvement was observed on day 25. Our case presented with rupture of a distal ACA aneurysm and localized medial hemispheric sulcal hematoma. There was partial infarction of the CC, including the body and genu, with symptoms of AM. The symptoms gradually improved, and no manifestations of callosal disconnection syndrome were observed. The favorable recovery without persistent callosal disconnection suggests that partial involvement of the CC may cause transient AM rather than permanent neurobehavioral deficits. Our patient did not exhibit spasms. Hence, spasm was not considered a factor affecting the onset of CC infarction. There was a thick hematoma in the sulcus. Therefore, the medial callosal artery, which was unclear on cerebral angiography, might have been compressed by the hematoma or the CC itself might have been compressed by the hematoma.
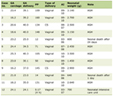 Click to view | Table 1. Subarachnoid Hemorrhage Predominantly Accompanied by CC Infarction |
This case provides an important educational message for neurosurgeons: even very small distal ACA aneurysms may rupture and present with atypical neurobehavioral symptoms, and careful interpretation of sulcal hematoma distribution and network-level dysfunction is essential. The treatment options for cerebral aneurysms include clipping and coil embolization. This case involved an aneurysm of the ACA, and both clipping and coil embolization were considered possible treatment options for the location of the aneurysm. However, the aneurysm was small, and coil embolization was challenging to perform because extremely small coils would be required. In addition, clipping was performed to assess whether the hematoma had actually ruptured, as it was not detected in the thickest part of the localized medial hemispheric sulcal hematoma. During surgery, pre-rupture was observed, and the aneurysm was considered to have actually ruptured because a brownish-red hematoma was found through the aneurysm wall. Clipping was considered useful for confirming extremely small cerebral aneurysms and the actual rupture site.
Conclusion
We encountered a case of SAH caused by a ruptured small ACA aneurysm accompanied by a localized medial hemispheric sulcal hematoma. The aneurysm was extremely small and somewhat difficult to identify. However, it was identified by cautiously reviewing the images. The medial callosal artery or the CC might have been compressed by the hematoma, leading to CC infarction. In cases of CC infarction, symptoms such as AM may develop. However, in this case, CC infarction alone could not be determined as the cause. Further, multiple factors, such as functional impairment of the frontal lobe and cingulate gyrus associated with SAH, might have played a role.
Learning points
Distal ACA microaneurysms can rupture and cause localized medial sulcal hematoma with CTA-negative findings, requiring careful hematoma pattern recognition and diagnostic angiography. Partial CC infarction after rupture may induce transient AM through multifactorial network dysfunction, and favorable recovery is achievable with appropriate management.
Acknowledgments
None to declare.
Financial Disclosure
None to declare.
Conflict of Interest
The authors declare no conflict of interest.
Informed Consent
Informed consent has been obtained from the patient for publication.
Author Contributions
Conceptualization, design, definition of intellectual content, literature search, clinical studies, data acquisition, data analysis, manuscript preparation, manuscript editing, and manuscript review: Seigo Kimura, Daiji Ogawa, Ryokichi Yagi, Norihito Fukawa, Keiichi Yamada, Hirokatsu Taniguchi, and Masahiko Wanibuchi. Guarantor: Seigo Kimura.
Data Availability
All data generated or analyzed during this study are included in this published article.
Abbreviations
ACA: anterior cerebral artery; AM: akinetic mutism; CC: corpus callosum; CT: computed tomography; CTA: computed tomography angiography; MRA: magnetic resonance angiography; MRI: magnetic resonance imaging; mRS: modified Rankin Scale; P-CA AN: pericallosal and callosomarginal artery aneurysm; SAH: subarachnoid hemorrhage
| References | ▴Top |
- Inci S, Erbengi A, Ozgen T. Aneurysms of the distal anterior cerebral artery: report of 14 cases and a review of the literature. Surg Neurol. 1998;50(2):130-139; discussion 139-140.
doi pubmed - Kumar K, Strbian D, Sundararajan S. Acute cerebral infarction presenting with weakness in both legs and one arm. Stroke. 2015;46(6):e134-136.
doi pubmed - Watanabe O, Miyano S, Sugishita M, Kamikubo T, Sugawara M. Rehabilitation of a case with almost complete infarction of the corpus callosum. Japanese Journal of Rehabilitation Medicine. 2001;38(6):465-470.
- Liu C, Wang H, Xie B, Tian S, Ding Y. Clinical analysis of Marchiafava-Bignami disease. BMC Neurol. 2024;24(1):389.
doi pubmed - Li S, Sun X, Bai YM, Qin HM, Wu XM, Zhang X, Jolkkonen J, et al. Infarction of the corpus callosum: a retrospective clinical investigation. PLoS One. 2015;10(3):e0120409.
doi pubmed - Takahashi S, Ohnaka K, Ishiwada T, Kino T, Arai T. Infarction of the entire corpus callosum as a complication in subarachnoid hemorrhage: a case report. Interdisciplinary Neurosurgery. 2017;7:53-55.
- Sun X, Li J, Fan C, Zhang H, Si Y, Fang X, Guo Y, et al. Clinical, neuroimaging and prognostic study of 127 cases with infarction of the corpus callosum. Eur J Neurol. 2019;26(8):1075-1081.
doi pubmed - Alnaami I, Alqahtani SA. Full recovery of Akinetic Mutism after corpus callosum infarction in post subarachnoid hemorrhage of ruptured distal anterior cerebral artery aneurysm. Neurosciences (Riyadh). 2020;25(5):412-415.
doi pubmed - Abbuehl LS, Lippert J, Hakim A. Split-brain syndrome after subarachnoid haemorrhage. BMJ Case Rep. 2024;17(1):e258538.
doi pubmed
This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (CC BY 4.0), which permits unrestricted use, distribution, and reproduction in any medium, including commercial use, provided the original work is properly cited.
Journal of Neurology Research is published by Elmer Press Inc.